Label: MULTI-PURPOSE SKIN PROTECTANT- a and d ointment ointment
- NDC Code(s): 59779-841-01, 59779-841-02, 59779-841-03, 59779-841-04
- Packager: CVS Pharmacy
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated February 23, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
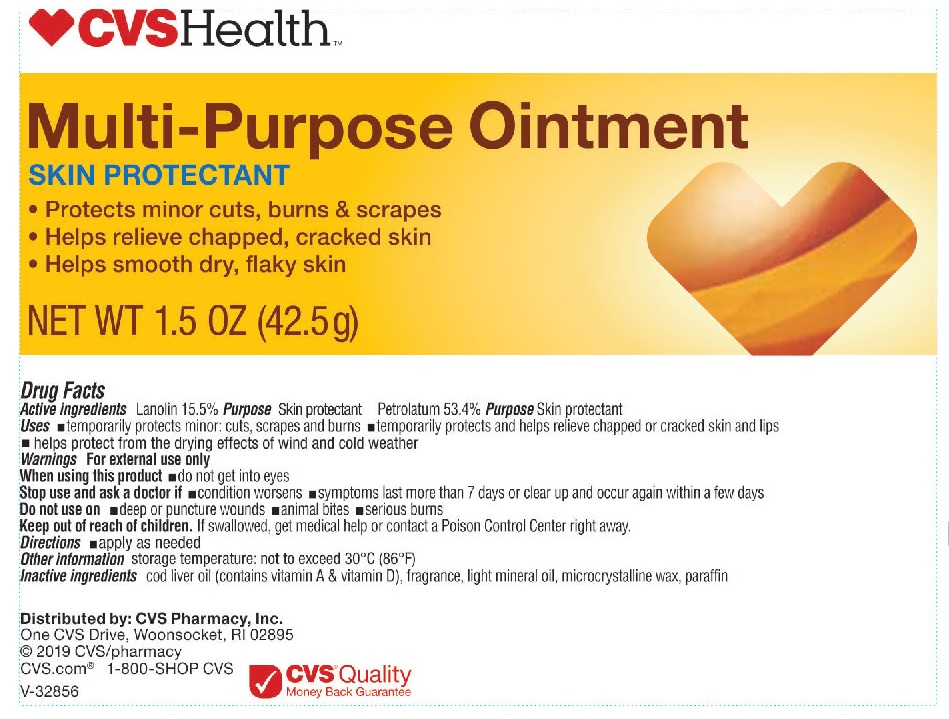
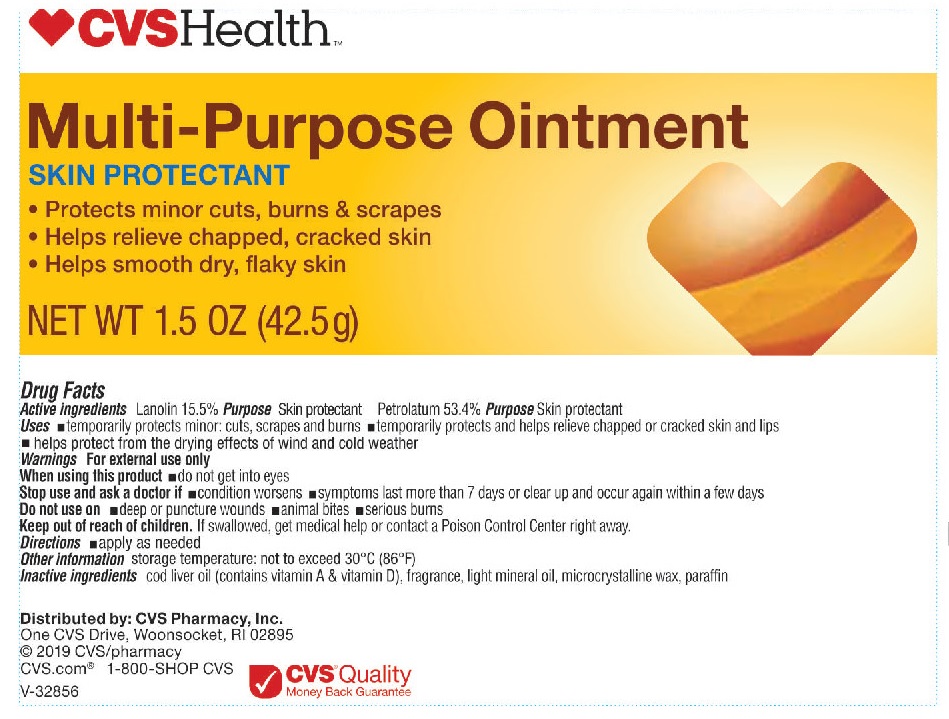
- Active Ingredients
- Uses
- Warnings
- When using this product
- Stop use and ask a doctor if
- Do not use on
- Keep out of reach of children.
- Directions
- Other information
- Inactive ingredients
- Questions?
- Carton Label
- Tube Label
- Carton Label
- Tube Label
-
INGREDIENTS AND APPEARANCE
MULTI-PURPOSE SKIN PROTECTANT
a and d ointment ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:59779-841 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PETROLATUM (UNII: 4T6H12BN9U) (PETROLATUM - UNII:4T6H12BN9U) PETROLATUM 53.4 g in 100 g LANOLIN (UNII: 7EV65EAW6H) (LANOLIN - UNII:7EV65EAW6H) LANOLIN 15.5 g in 100 g Inactive Ingredients Ingredient Name Strength MICROCRYSTALLINE WAX (UNII: XOF597Q3KY) PARAFFIN (UNII: I9O0E3H2ZE) COD LIVER OIL (UNII: BBL281NWFG) MINERAL OIL (UNII: T5L8T28FGP) CARROT SEED OIL (UNII: 595AO13F11) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59779-841-03 1 in 1 CARTON 07/20/2016 1 NDC:59779-841-01 42.5 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:59779-841-04 1 in 1 CARTON 07/20/2016 2 NDC:59779-841-02 42.5 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M016 07/20/2016 Labeler - CVS Pharmacy (062312574) Registrant - Unipack LLC (116015769) Establishment Name Address ID/FEI Business Operations Unipack LLC 009248480 manufacture(59779-841)