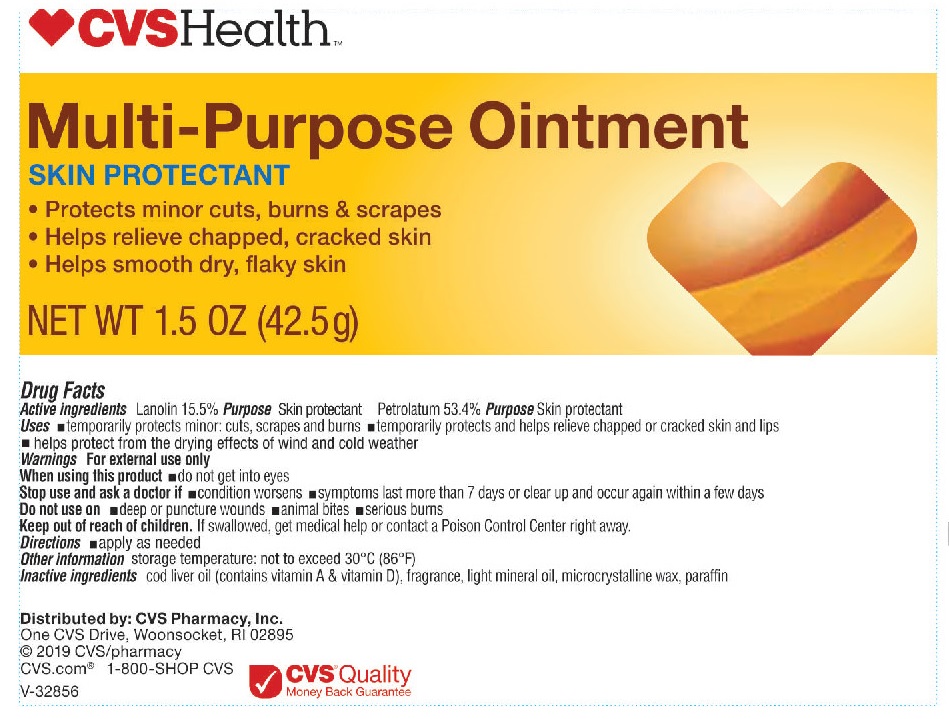
Uses
helps treat and prevent diaper rash, temporarily protects minor: cuts, scrapes and burns, temporarily protects and helps relieve chapped, chafed or cracked skin and lips, protects chafed skin due to diaper rash and helps seal out wetness
Stop use and ask a doctor if
condition worsens, symptoms last more than 7 days or clear up and occur again within a few days.
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Directions
for skin protectant use: apply as needed
for diaper rash use: change wet and soiled diapers promptly, cleanse the diaper area and allow to dry, apply ointment liberally as often as necessary, with each diaper change, especially at bedtime or anytime when exposure to wet diapers may be prolonged