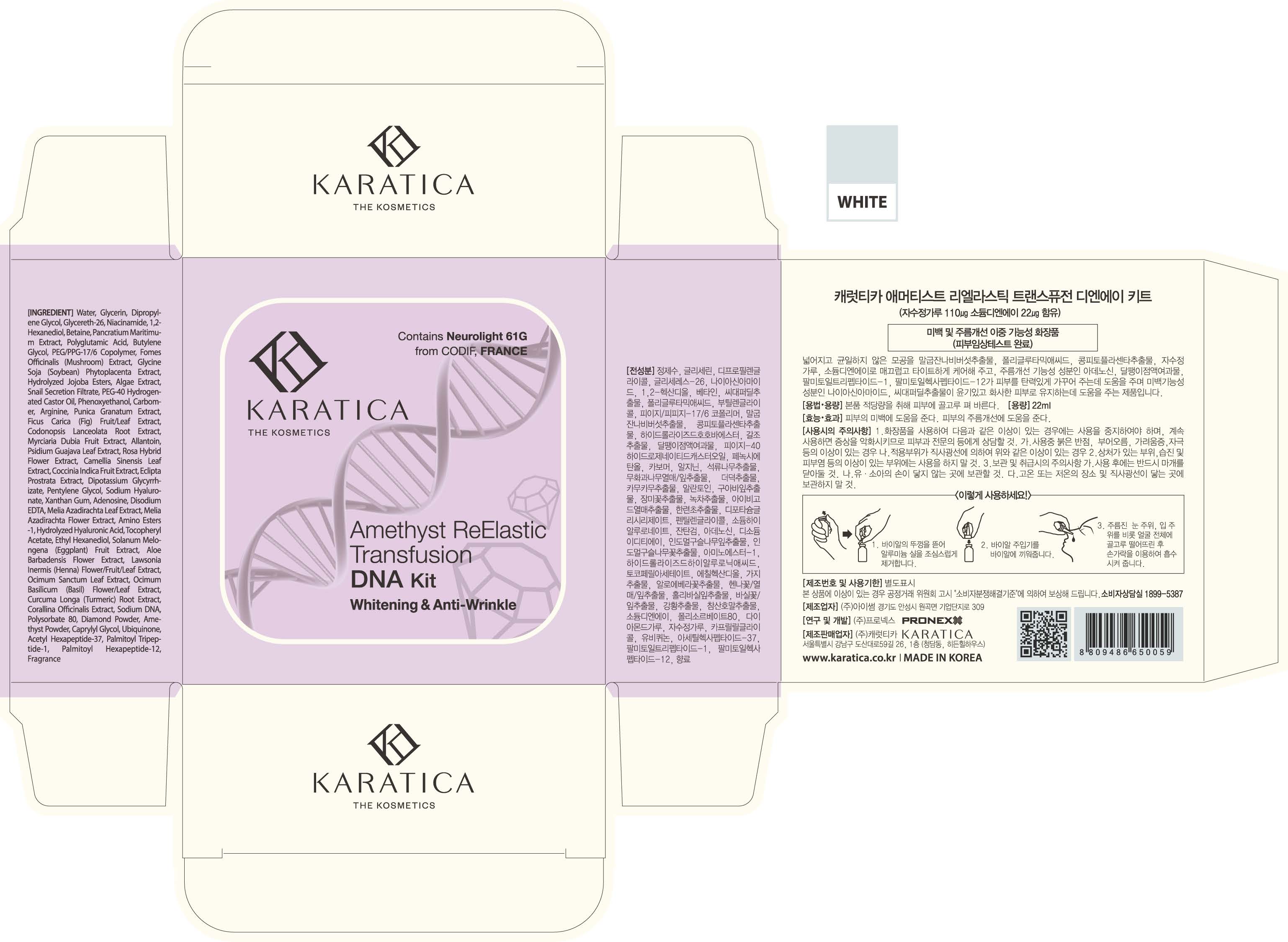
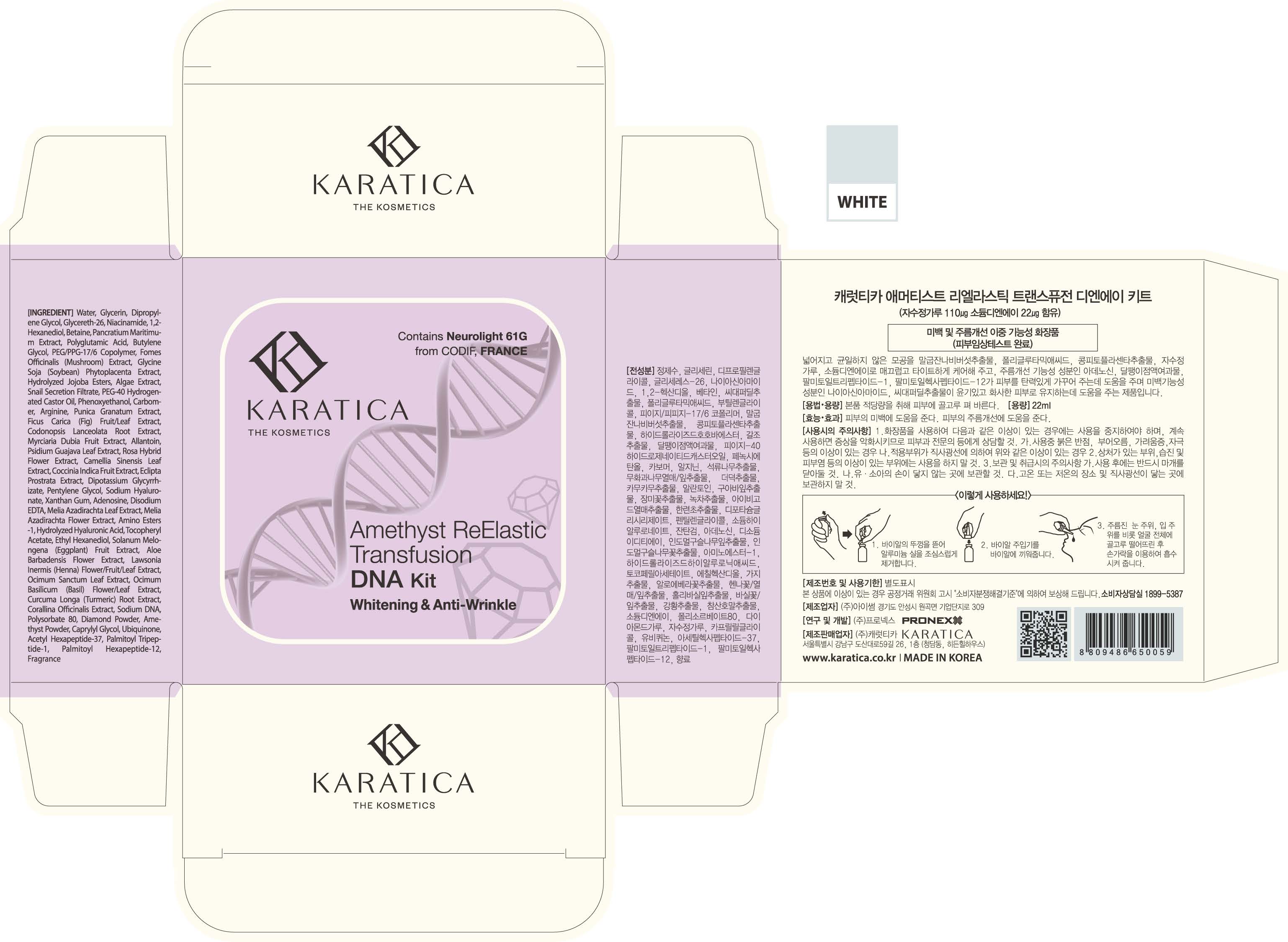
Label: AMETHYST REELASTIC TRANSFUSION DNA KIT- glycerin liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 70514-0003-1 - Packager: Karatica Co., Ltd
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 5, 2017
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- INACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
-
WARNINGS
If you experience following symptoms after using the cosmetics, you should immediately stop using the cosmetics. If you continue to use them, the symptoms may worsen. Consult with your dermatologist.
A) When you experience red spots, swelling, itchiness and irritation while applying the product.
B) When the applied area experience symptoms while it is exposed to direct sunlight
2. Do not use the cosmetics on the areas where you have wounds, eczema or dermatitis
3. Precautions when storing and using the product
A) Keep the lid closed after using the product
B) Store it out of reach of children
C) Do not store it in places with high or low temperature or where it is exposed to the sunlight directly. - DOSAGE & ADMINISTRATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
AMETHYST REELASTIC TRANSFUSION DNA KIT
glycerin liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70514-0003 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GLYCERIN (UNII: PDC6A3C0OX) (GLYCERIN - UNII:PDC6A3C0OX) GLYCERIN 5 g in 100 mL Inactive Ingredients Ingredient Name Strength NIACINAMIDE (UNII: 25X51I8RD4) ADENOSINE (UNII: K72T3FS567) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70514-0003-1 22 mL in 1 AMPULE; Type 0: Not a Combination Product 12/05/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part347 12/05/2017 Labeler - Karatica Co., Ltd (689605545) Registrant - Karatica Co., Ltd (689605545) Establishment Name Address ID/FEI Business Operations Karatica Co., Ltd 689605545 manufacture(70514-0003)