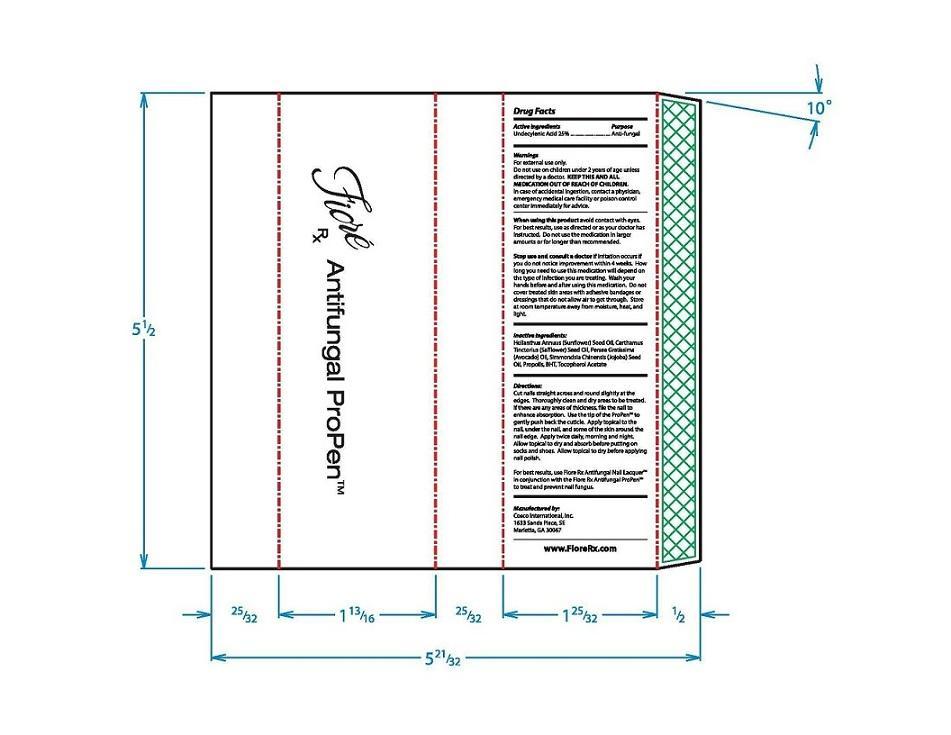
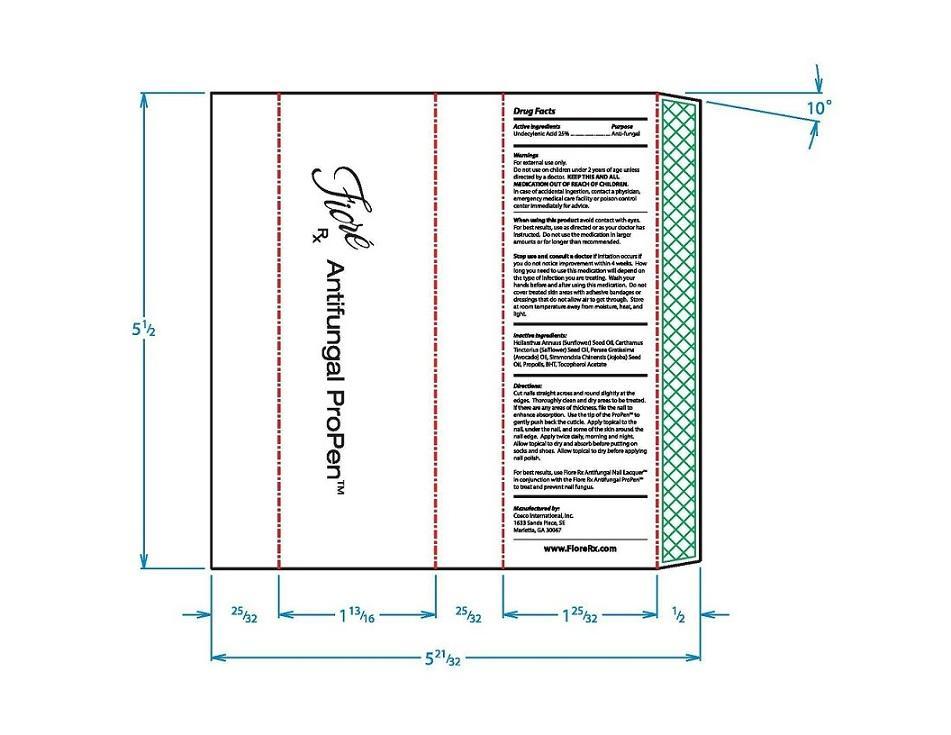
Label: FIORE RX ANTIFUNGAL PROPEN- undecylenic acid liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 52261-2500-0, 52261-2500-1 - Packager: Cosco International, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated September 19, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- WARNINGS
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
-
Directions:
Cut nail straight across and round slightly at the edges. Throughly clean and dry areas to be treated. If there are any ares of thickness, file the nail to enhance absorption. Use the tip of the ProPen™ to gently push back the cuticle. Apply topical to the nail, under the nail, and some of the skin around the nail edge. Apply twice daily, morning and night. Allow topical to dry and absorb before putting on socks and shoes. Allow topical to dry before applying nail polish.
For best results, use Fiore Rx Antifungal Nail Lacquer™ in conjuction with the Fiore Rx Antifungal ProPen™ to treat and prevent nail fungus.
- Inactive ingredients:
- Outer Package PLPDP
-
INGREDIENTS AND APPEARANCE
FIORE RX ANTIFUNGAL PROPEN
undecylenic acid liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:52261-2500 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Undecylenic Acid (UNII: K3D86KJ24N) (Undecylenic Acid - UNII:K3D86KJ24N) Undecylenic Acid 0.5 g in 2 g Inactive Ingredients Ingredient Name Strength SUNFLOWER SEED (UNII: R9N3379M4Z) 1.4368 g in 2 g SAFFLOWER OIL (UNII: 65UEH262IS) 0.03 g in 2 g AVOCADO OIL (UNII: 6VNO72PFC1) 0.02 g in 2 g SIMMONDSIA CHINENSIS SEED (UNII: D24K2Q1F6H) 0.01 g in 2 g PROPOLIS WAX (UNII: 6Y8XYV2NOF) 0.002 g in 2 g BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) 0.001 g in 2 g .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) 0.0002 g in 2 g Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:52261-2500-1 1 in 1 BOX 1 NDC:52261-2500-0 2 g in 1 TUBE, WITH APPLICATOR Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333C 05/21/2013 Labeler - Cosco International, Inc. (016433141) Registrant - Cosco International, Inc. (016433141) Establishment Name Address ID/FEI Business Operations Cosco International, Inc. 016433141 manufacture(52261-2500) , label(52261-2500) , pack(52261-2500)