Label: EXTRA PAIN RELIEF- acetaminophen, aspirin and caffeine tablet, film coated
-
NDC Code(s):
49483-370-00,
49483-370-01,
49483-370-20,
49483-370-31, view more49483-370-42
- Packager: TIME CAP LABS
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated March 15, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- DRUG FACTS
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings
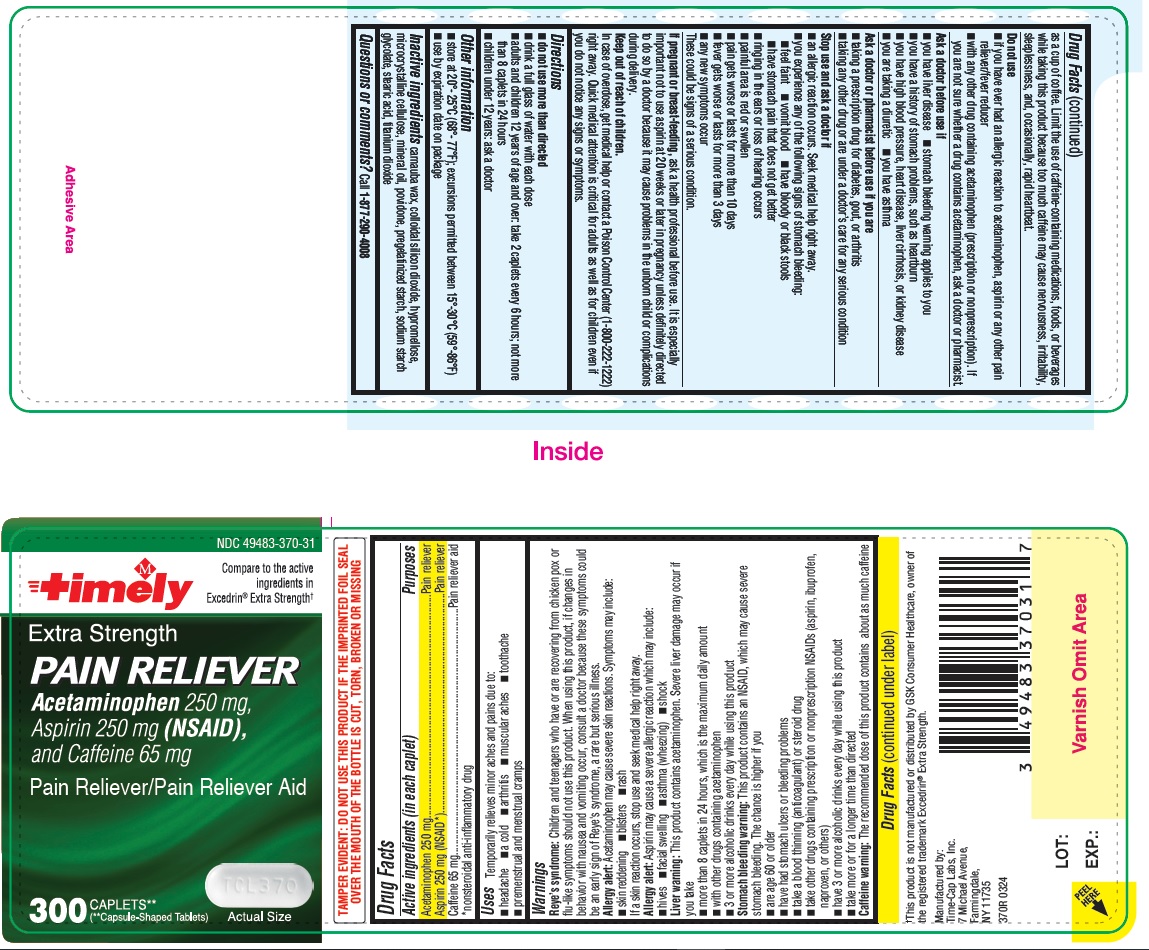
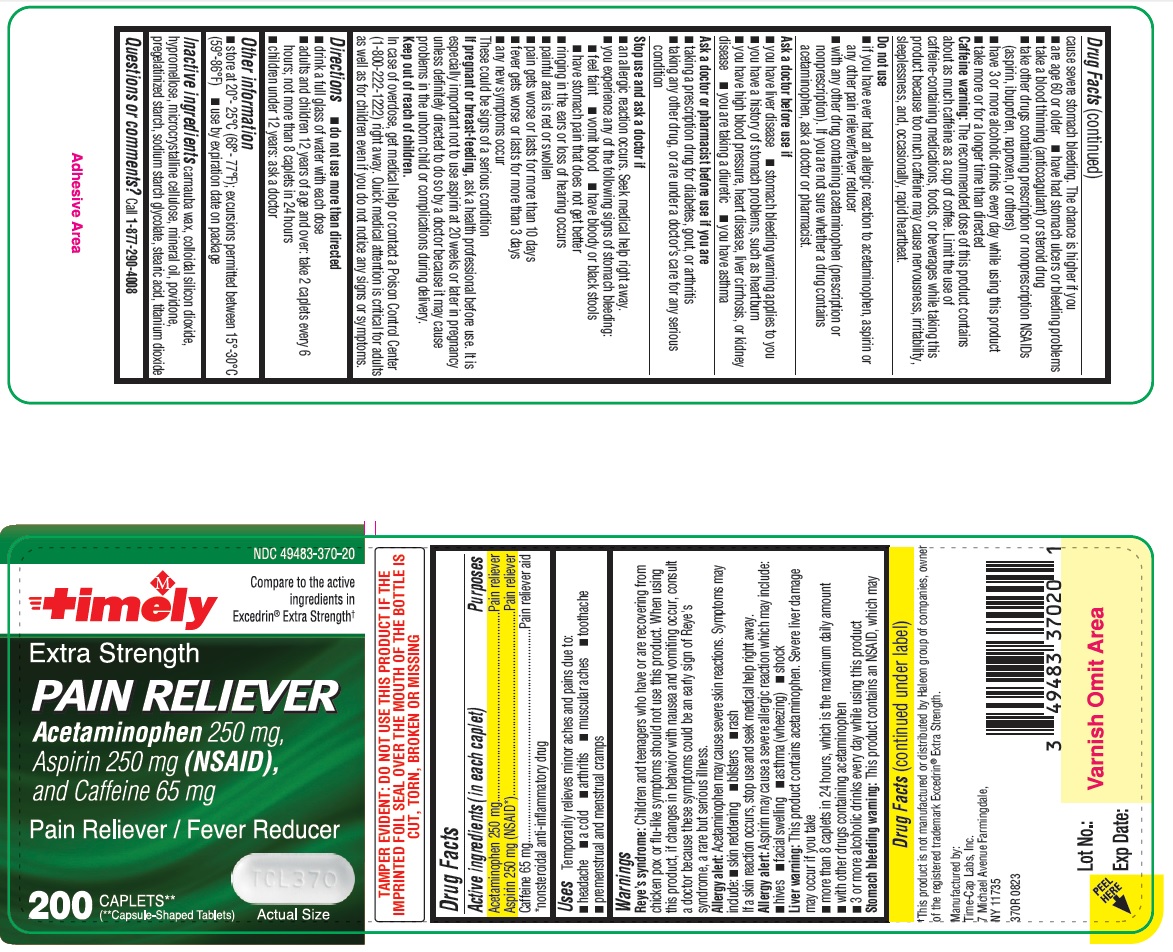
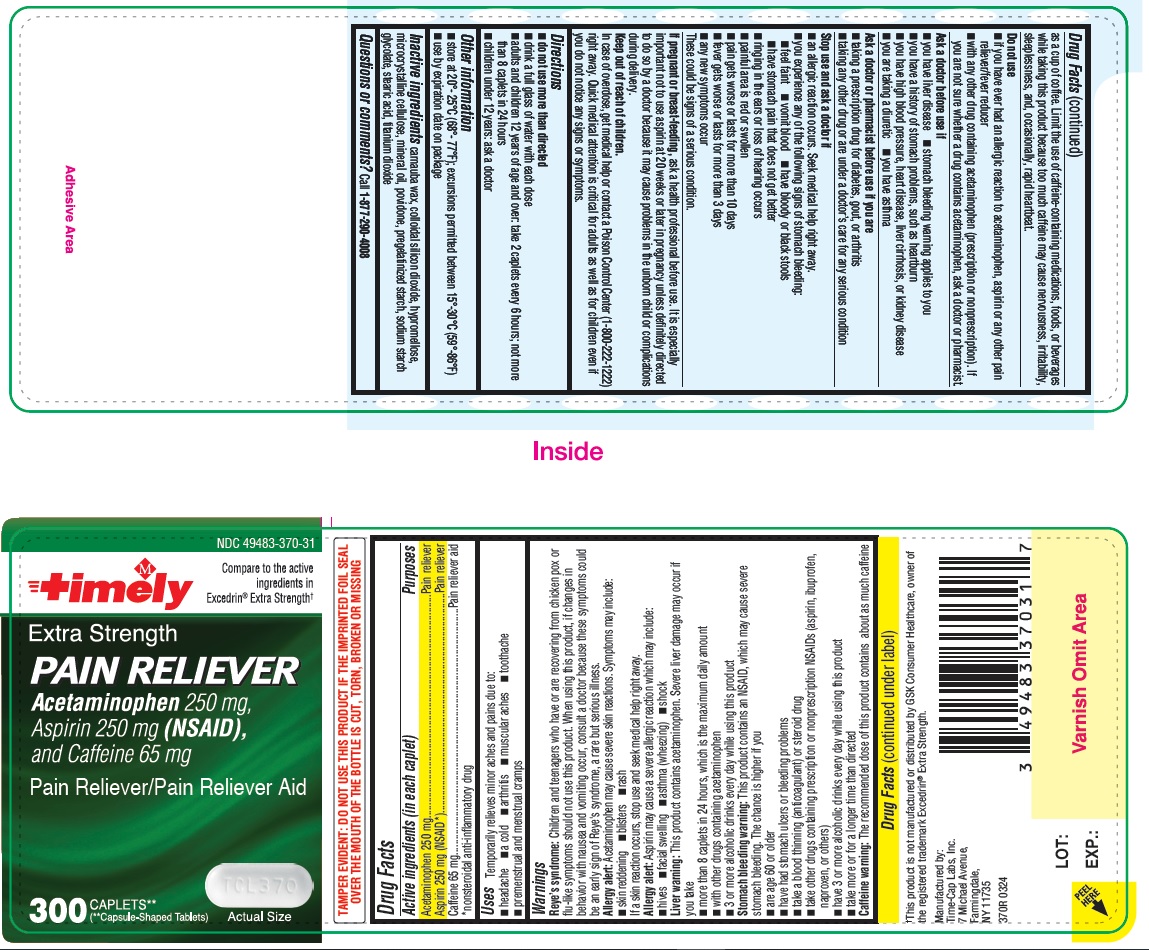
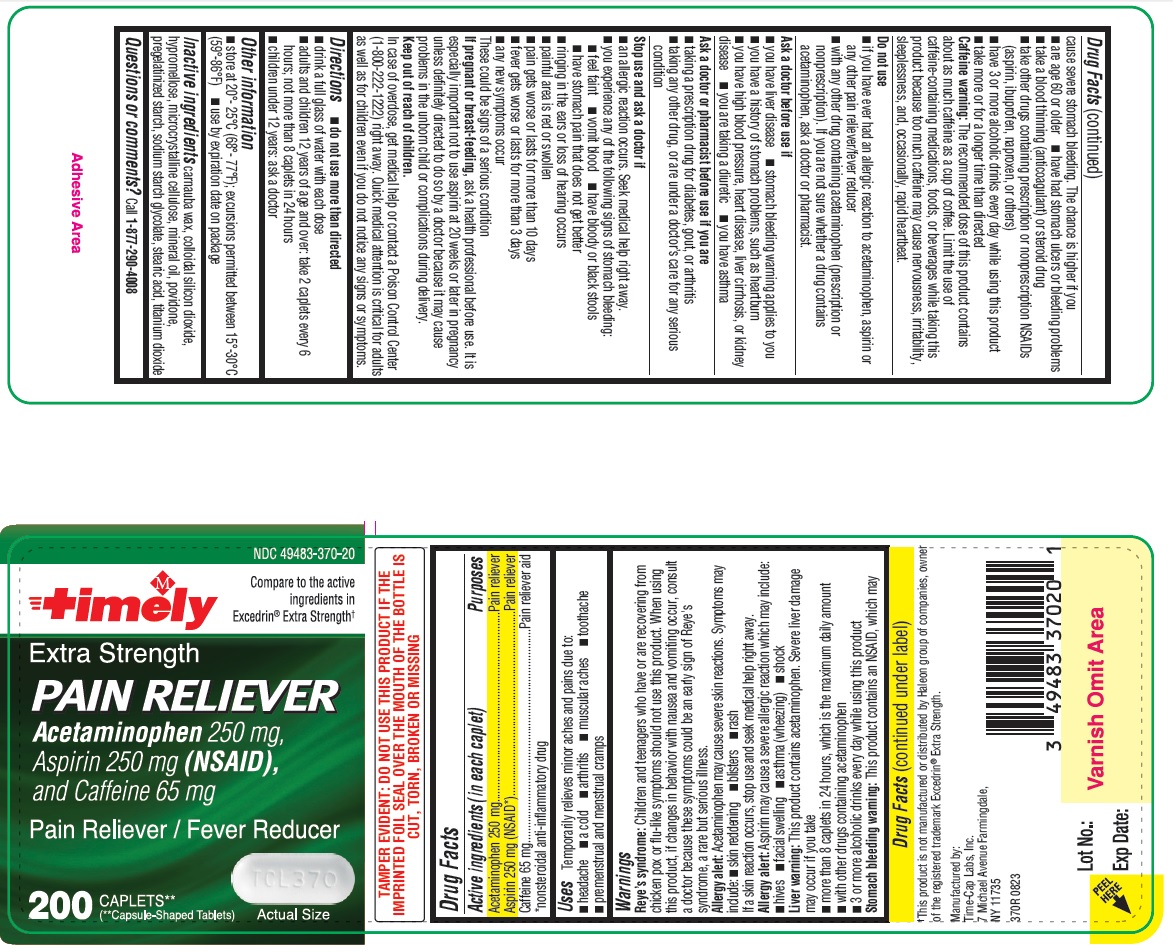
Reye’s syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye’s syndrome, a rare but serious illness.
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
• skin reddening
• blisters
• rash
If a skin reaction occurs, stop use and seek medical help right away.
Allergy alert: Aspirin may cause a severe allergic reaction which may include:
• hives
• facial swelling
• asthma (wheezing)
• shock
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
• more than 8 caplets in 24 hours, which is the maximum daily amount
• with other drugs containing acetaminophen
• 3 or more alcoholic drinks every day while using this product
Stomach bleeding warning: This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you
• are age 60 or older
• have had stomach ulcers or bleeding problems
• take a blood thinning (anticoagulant) or steroid drug
• take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
• have 3 or more alcoholic drinks every day while using this product
• take more or for a longer time than directed
Caffeine warning: The recommended dose of this product contains about as much caffeine as a cup of coffee. Limit the use of caffeine-containing medications, foods, or beverages while taking this product because too much caffeine may cause nervousness, irritability, sleeplessness, and, occasionally, rapid heartbeat.
- DO NOT USE
- ASK DOCTOR
- ASK DOCTOR/PHARMACIST
-
STOP USE
Stop use and ask a doctor if
• an allergic reaction occurs. Seek medical help right away.
• you experience any of the following signs of stomach bleeding:
o feel faint
o vomit blood
o have bloody or black stools
o have stomach pain that does not get better
• ringing in the ears or loss of hearing occurs
• painful area is red or swollen
• pain gets worse or lasts for more than 10 days
• fever gets worse or lasts for more than 3 days
• any new symptoms occur
These could be signs of a serious condition.
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- DIRECTIOINS
- OTHER INFORMATION
- INACTIVE INGREDIENT
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
EXTRA PAIN RELIEF
acetaminophen, aspirin and caffeine tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49483-370 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 250 mg ASPIRIN (UNII: R16CO5Y76E) (ASPIRIN - UNII:R16CO5Y76E) ASPIRIN 250 mg CAFFEINE (UNII: 3G6A5W338E) (CAFFEINE - UNII:3G6A5W338E) CAFFEINE 65 mg Inactive Ingredients Ingredient Name Strength STEARIC ACID (UNII: 4ELV7Z65AP) CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) HYPROMELLOSES (UNII: 3NXW29V3WO) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) MINERAL OIL (UNII: T5L8T28FGP) POVIDONE (UNII: FZ989GH94E) STARCH, CORN (UNII: O8232NY3SJ) SODIUM STARCH GLYCOLATE TYPE A CORN (UNII: AG9B65PV6B) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color white Score no score Shape CAPSULE ((CAPSULE SHAPED TABLET)) Size 18mm Flavor Imprint Code TCL370 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49483-370-01 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 07/16/2021 2 NDC:49483-370-20 200 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 11/01/2013 3 NDC:49483-370-00 20000 in 1 CARTON; Type 0: Not a Combination Product 02/22/2016 4 NDC:49483-370-42 1 in 1 CARTON 07/16/2021 4 24 in 1 BOTTLE; Type 0: Not a Combination Product 5 NDC:49483-370-31 300 in 1 BOTTLE; Type 0: Not a Combination Product 03/15/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M013 11/01/2013 Labeler - TIME CAP LABS (037052099) Registrant - TIME CAP LABS (037052099) Establishment Name Address ID/FEI Business Operations TIME CAP LABS 037052099 manufacture(49483-370)