Label: SALONPAS LIDOCAINE PAIN RELIEVING- lidocaine patch
- NDC Code(s): 46581-830-01, 46581-830-02, 46581-830-06, 46581-830-15
- Packager: Hisamitsu Pharmaceutical Co., Inc.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated March 1, 2021
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
-
Warnings
For external use only
Do not use
- more than one patch at a time
- on wounds or damaged skin
- with a heating pad
- if you are allergic to any igredients of this product
When using this product
- use only as directed
- avoid contact with the eyes, mucous membranes or rashes
- do not bandage tightly
-
Directions
Adults and children 12 years of age and over:
- clean and dry affected area
- remove film from patch and apply to the skin (see illustration)
- apply to affected are not more than 3 to 4 times daily
- remove patch from the skin after at most 8-hour application
Children under 12 years of age: consult a doctor
- Other information
- Inactive ingredients
- Questions or comments?
-
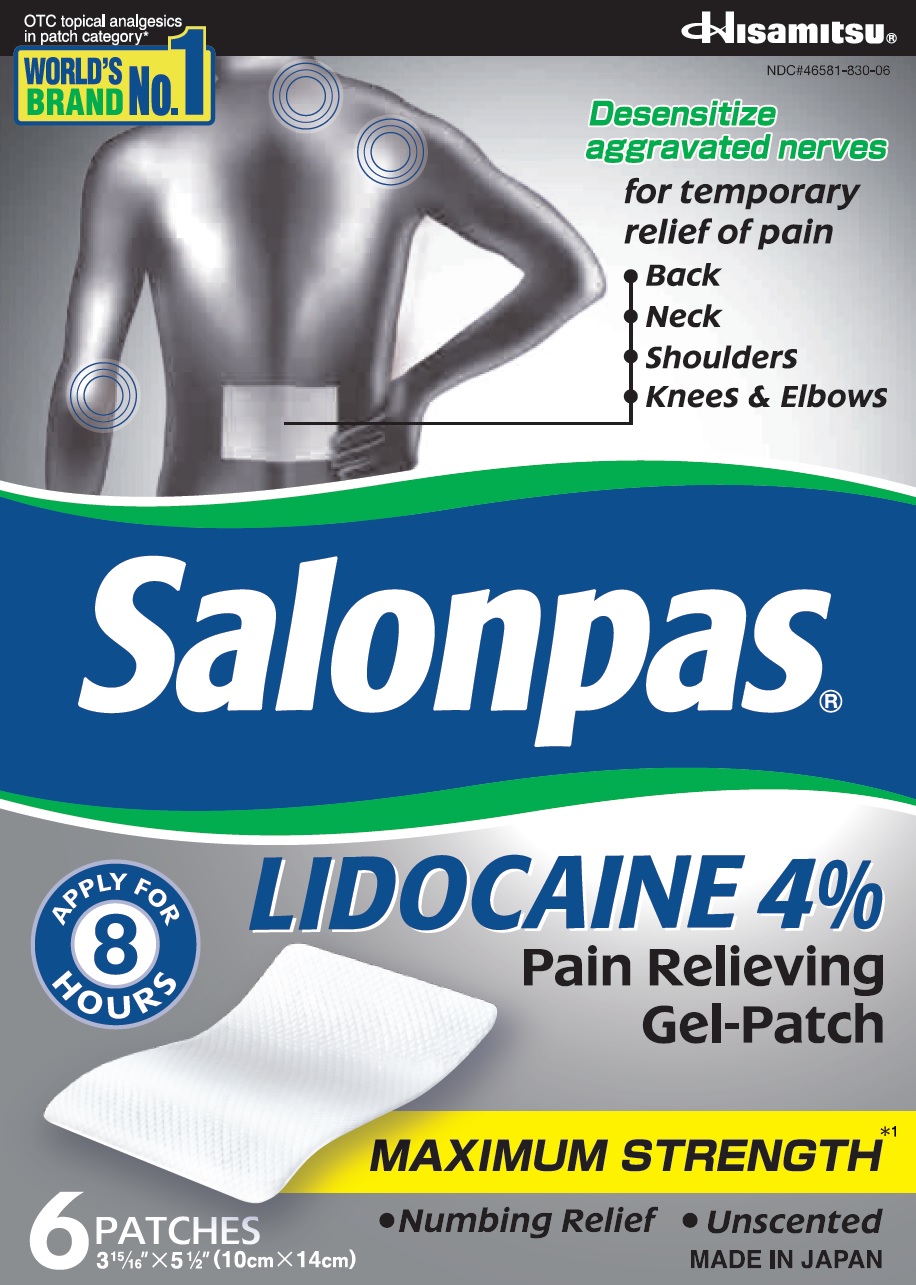
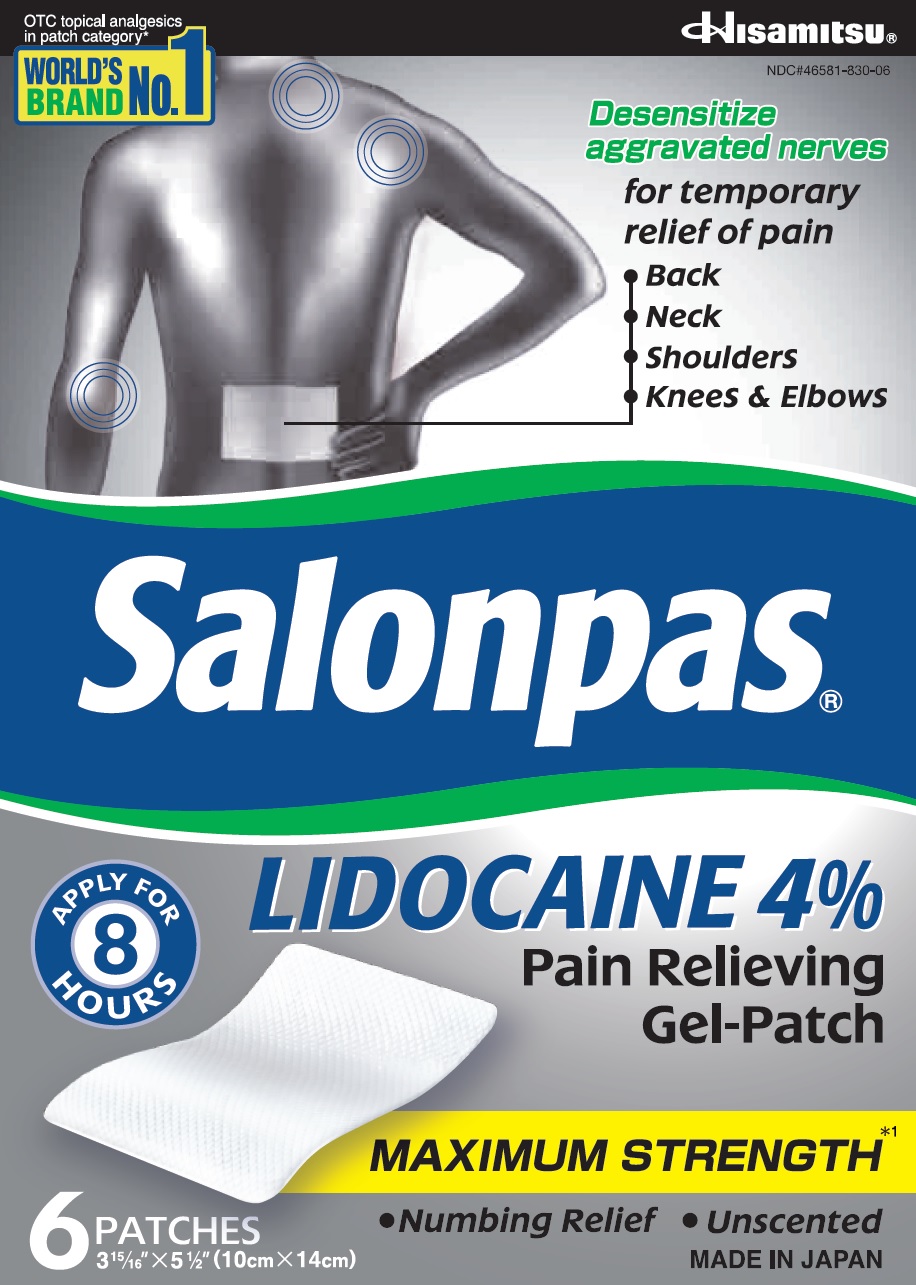
Principal Display Panel
OTC topical analgesics in patch category*
WORLD'S BRAND NO.1Hisamitsu
NDC#46581-830-06Desensitize aggravated nerves
for temporary relief of pain- Back
- Neck
- Shoulders
- Knees and Elbows
Salonpas LIDOCAINE 4% Pain Relieving Gel-Patch
APPLY FOR 8 HOURS
MAXIMUM STRENGTH*1
- Numbing Relief
- Unscented
6 PATCHES
3 15/16" X 5 1/2" (10cm X 14cm)MADE IN JAPAN
-
Principal Display Panel
CUT OPEN HERE
NDC#46581-830-01
HOW TO APPLY
1 Pull apart.
2 Peel off one side of the film.
3 Place on affected area.
4 Peel off the remaining film.
5 Press patch thoroughly.Salonpas LIDOCAINE 4% Pain Relieving Gel-Patch
APPLY FOR 8 HOURS
MAXIMUM STRENGTH*1
- Numbing Relief
- Unscented
1 PATCH
3 15/16" X 5 1/2" (10cm X 14cm)Child-resistant packaging.
Hisamitsu
MADE IN JAPAN

-
INGREDIENTS AND APPEARANCE
SALONPAS LIDOCAINE PAIN RELIEVING
lidocaine patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:46581-830 Route of Administration TOPICAL, PERCUTANEOUS, TRANSDERMAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE (UNII: 98PI200987) (LIDOCAINE - UNII:98PI200987) LIDOCAINE 560 mg Inactive Ingredients Ingredient Name Strength ALUMINUM SILICATE (UNII: T1FAD4SS2M) DIHYDROXYALUMINUM AMINOACETATE (UNII: DO250MG0W6) EDETATE DISODIUM (UNII: 7FLD91C86K) GELATIN (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) METHYLPARABEN (UNII: A2I8C7HI9T) OLEIC ACID (UNII: 2UMI9U37CP) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PROPYLPARABEN (UNII: Z8IX2SC1OH) TARTARIC ACID (UNII: W4888I119H) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:46581-830-06 6 in 1 BOX 08/01/2016 1 1 in 1 POUCH; Type 0: Not a Combination Product 2 NDC:46581-830-02 2 in 1 BOX 10/01/2017 2 1 in 1 POUCH; Type 0: Not a Combination Product 3 NDC:46581-830-15 15 in 1 BOX 08/01/2017 3 1 in 1 POUCH; Type 0: Not a Combination Product 4 NDC:46581-830-01 1 in 1 POUCH; Type 0: Not a Combination Product 08/01/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 08/01/2016 Labeler - Hisamitsu Pharmaceutical Co., Inc. (690539713)