Label: KERANIQUE FOR WOMEN HAIR REGROWTH TREATMENT- minoxidil liquid
- NDC Code(s): 65121-562-02
- Packager: Pure Source, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated February 10, 2017
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- KERANIQUE For Women Hair Regrowth Treatment
- Active ingredient
- Use
-
Warnings
For external use only
Flammable: Keep away from fire or flame
Do not use if
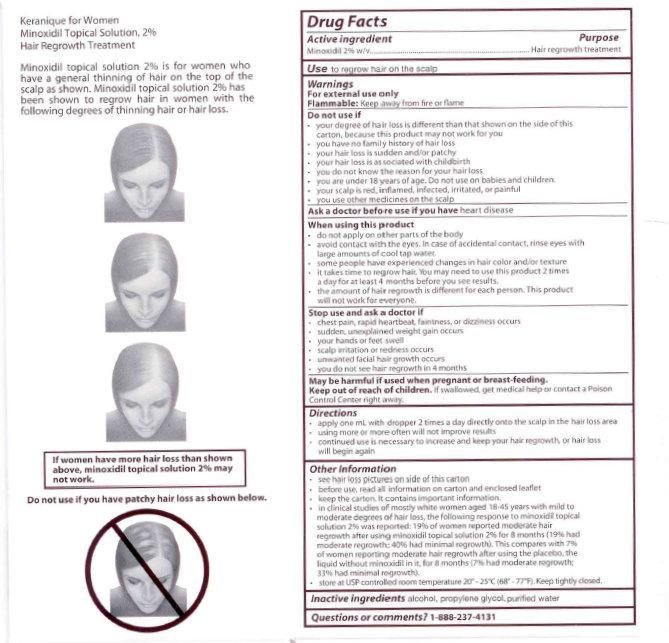
- your degree of hair loss is different than that shown on the side of this carton, because this product may not work for you
- you have no family history of hair loss
- your hair loss is sudden and/or patchy
- your hair loss is associated with childbirth
- you do not know the reason for your hair loss
- you are under 18 years of age. Do not use on babies and children.
- your scalp is red, inflamed, infected, irritated, or painful
- you use other medicines on the scalp
When using this product
- do not apply on other parts of the body
- avoid contact with the eyes. In case of accidental contact, rinse eyes with large amounts of cool tap water.
- some people have experienced changes in hair color and/or texture
- It takes time to regrow hair. You may need to use this product 2 times a day for at least 4 months before you see results.
- the amount of hair regrowth is different for each person. This product will not work for everyone.
- Directions
-
Other Information
- see hair loss pictures on side of this carton
- before use, read all information on carton and enclosed leaflet
- keep the carton. It contains important information.
- In clinical studies of mostly white women aged 18-45 years with mild to moderate degrees of hair loss, the following response to minoxidil topical solutions 2% was reported: 19% of women reported moderate hair regrowth after using minoxidil topical solution 2% for 8 months (19% had moderate regrowth: 40% had minimal regrowth). This compares with 7% of women reporting moderate hair regrowth after using the placebo, the liquid without minoxidil in it, for 8 months (7% had moderate regrowth: 33 % had minimal regrowth).
- store at USP controlled room temperature 20
o - 25
oC. (68
o - 77
oF). Keep tightly closed.
- Inactive ingredients
- Questions or comments?
- KERANIQUE For Women Hair Regrowth Treatment 2-60ml (65121-562-02)
-
INGREDIENTS AND APPEARANCE
KERANIQUE FOR WOMEN HAIR REGROWTH TREATMENT
minoxidil liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:65121-562 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MINOXIDIL (UNII: 5965120SH1) (MINOXIDIL - UNII:5965120SH1) MINOXIDIL 20 mg in 1 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:65121-562-02 1 in 1 CARTON 02/10/2017 1 60 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA078176 02/06/2014 Labeler - Pure Source, LLC (080354456) Establishment Name Address ID/FEI Business Operations Pure Source, LLC 080354456 manufacture(65121-562) , repack(65121-562)