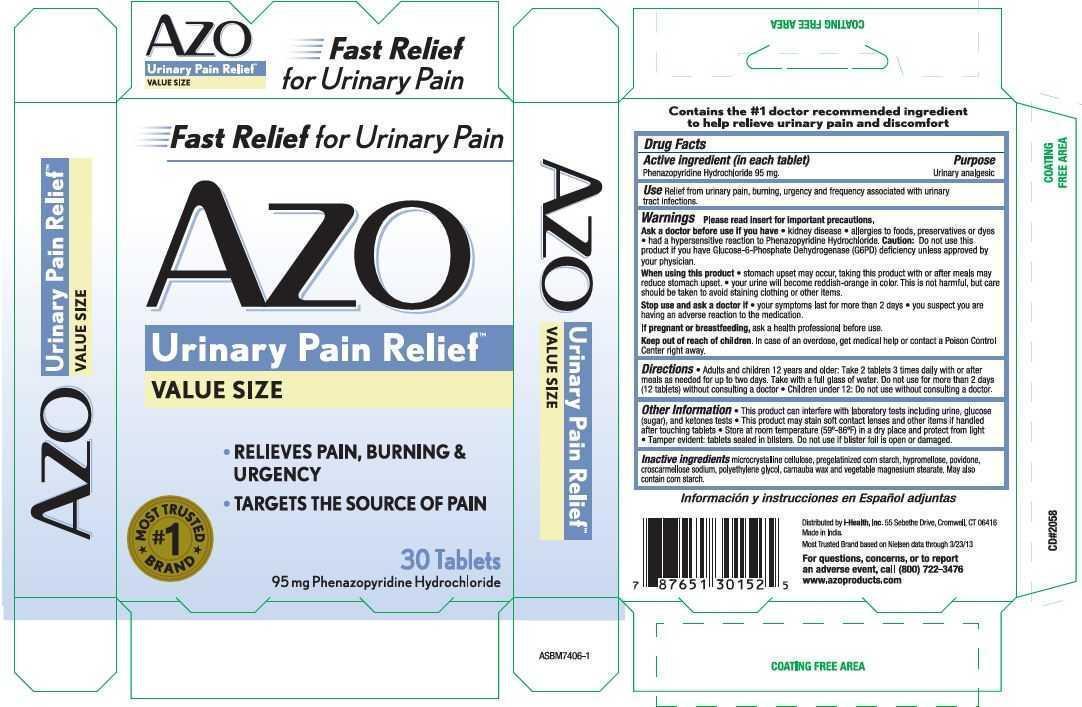
Label: AZO- urinary pain relief tablet
- NDC Code(s): 49973-301-30, 49973-301-31, 49973-301-32
- Packager: i-Health, Inc.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated December 12, 2020
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings Please read insert for important precautions.
Ask a doctor before use if you have
- kidney disease
- allergies to foods, preservatives or dyes
- had a hypersensitive reaction to Phenazopyridine Hydrochloride.
Caution: Do not use this product if you have Glucose-6-Phosphate Dehydrogenase (G6PD) deficiency unless approved by your physician.
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
-
GENERAL PRECAUTIONS
Other Information
- This product can interfere with laboratory tests including urine, glucose (sugar), and ketones tests
- This product may stain soft contact lenses and other items if handled after touching tablets
- Store at room temperature (59-86 F) in a dry place and protect from light
- Tamper evident: tablets sealed in blisters. Do not use if blister foil or seal is open or damaged.
- INACTIVE INGREDIENT
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
AZO
urinary pain relief tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49973-301 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Phenazopyridine Hydrochloride (UNII: 0EWG668W17) (Phenazopyridine - UNII:K2J09EMJ52) Phenazopyridine Hydrochloride 95 mg Inactive Ingredients Ingredient Name Strength CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) STARCH, CORN (UNII: O8232NY3SJ) HYPROMELLOSES (UNII: 3NXW29V3WO) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) CARNAUBA WAX (UNII: R12CBM0EIZ) MAGNESIUM STEARATE (UNII: 70097M6I30) Product Characteristics Color brown (Maroon) Score no score Shape ROUND (Tablet) Size 7mm Flavor Imprint Code W Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49973-301-31 15 in 1 BOX 05/24/2012 1 2 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC:49973-301-30 15 in 1 BOX 05/24/2012 12/01/2020 2 2 in 1 BLISTER PACK; Type 0: Not a Combination Product 3 NDC:49973-301-32 1 in 1 BOX 11/11/2017 11/30/2020 3 18 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 05/24/2012 Labeler - i-Health, Inc. (061427694)