Label: TERBUTALINE SULFATE tablet
-
Contains inactivated NDC Code(s)
NDC Code(s): 0115-2611-01, 0115-2611-02, 0115-2611-03, 0115-2622-01, view more0115-2622-02, 0115-2622-03 - Packager: Amneal Pharmaceuticals of New York LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated January 25, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
BOXED WARNING
(What is this?)
WARNING: TOCOLYSIS
Oral terbutaline sulfate has not been approved and should not be used for acute or maintenance tocolysis. In particular, terbutaline sulfate should not be used for maintenance tocolysis in the outpatient or home setting. Serious adverse reactions, including death, have been reported after administration of terbutaline sulfate to pregnant women. In the mother, these adverse reactions include increased heart rate, transient hyperglycemia, hypokalemia, cardiac arrhythmias, pulmonary edema and myocardial ischemia. Increased fetal heart rate and neonatal hypoglycemia may occur as a result of maternal administration (see CONTRAINDICATIONS, Tocolysis).
-
DESCRIPTION
Terbutaline sulfate, USP, the active ingredient of terbutaline sulfate tablets, USP is a beta-adrenergic agonist bronchodilator available as tablets of 2.5 mg (2.05 mg of the free base) and 5 mg (4.1 mg of the free base) for oral administration. Terbutaline sulfate is ±-alpha-[(tert-butylamino)methyl]-3,5-dihydroxybenzyl alcohol sulfate (2:1) (salt).
The empirical formula is (C12H19NO3)2H2SO4 and the structural formula is
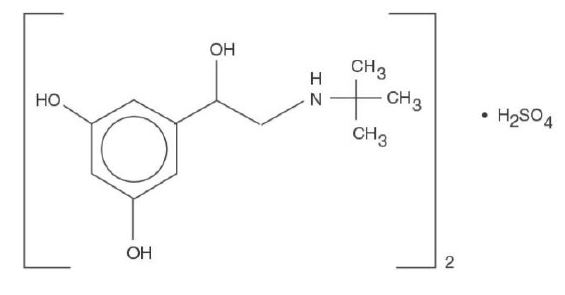
Terbutaline sulfate, USP is a white to gray-white crystalline powder. It is odorless or has a faint odor of acetic acid. It is soluble in water and in 0.1N hydrochloric acid, slightly soluble in methanol, and insoluble in chloroform. Its molecular weight is 548.65.
Inactive Ingredients. Hydroxypropyl methylcellulose, microcrystalline cellulose, anhydrous lactose, magnesium stearate, povidone, and pregelatinized starch.
-
CLINICAL PHARMACOLOGY
In vitro and in vivo pharmacologic studies have demonstrated that terbutaline exerts a preferential effect on beta2-adrenergic receptors. While it is recognized that beta2-adrenergic receptors are the predominant receptors in bronchial smooth muscle, data indicate that there is a population of beta2-receptors in the human heart, existing in a concentration between 10% to 50%. The precise function of these receptors has not been established (see WARNINGS). In controlled clinical studies in patients given terbutaline sulfate orally, proportionally greater changes occurred in pulmonary function parameters than in heart rate or blood pressure. While this suggests a relative preference for the beta2-receptors in man, the usual cardiovascular effects commonly associated with other sympathomimetic agents were also observed with terbutaline sulfate.
The pharmacologic effects of beta-adrenergic agonists, including terbutaline, are at least in part attributable to stimulation through beta-adrenergic receptors of intracellular adenyl cyclase, the enzyme which catalyzes the conversion of adenosine triphosphate (ATP) to cyclic 3', 5'-adenosine monophosphate (cAMP). Increased cAMP levels are associated with relaxation of bronchial smooth muscle and inhibition of release of mediators of immediate hypersensitivity from cells, especially from mast cells.
Controlled clinical studies have shown that terbutaline sulfate relieves bronchospasm in chronic obstructive pulmonary disease by significantly increasing pulmonary function (e.g., an increase of 15% or more in FEV1 and in FEF25% to 75%). After administration of terbutaline sulfate tablets, a measurable change in flow rate usually occurs within 30 minutes, and a clinically significant improvement in pulmonary function occurs within 60 to 120 minutes. The maximum effect usually occurs within 120 to 180 minutes. Terbutaline sulfate also produces a clinically significant decrease in airway and pulmonary resistance, which persists for 4 hours or longer. Significant bronchodilator action (as measured by airway resistance, FEF25% to 75% or PEFR) has also been demonstrated for up to 8 hours in some studies.
In studies comparing the effectiveness of terbutaline sulfate with that of ephedrine for up to 3 months, both drugs maintained a significant improvement in pulmonary function throughout this period of treatment.
Preclinical
Studies in laboratory animals (minipigs, rodents, and dogs) have demonstrated the occurrence of cardiac arrhythmias and sudden death (with histologic evidence of myocardial necrosis) when beta-agonists and methylxanthines were administered concurrently. The clinical significance of these findings is unknown.
Pharmacokinetics
Oral administration of 5-mg terbutaline sulfate tablets or 5 mg terbutaline sulfate in solution in 17 healthy, adult, male subjects, resulted in mean (SD) peak plasma terbutaline concentration of 8.3 (3.9) and 8.6 (3.6) ng/mL, which were observed at median (range) times of 2 (1 to 3) and 1.5 (0.5 to 3.0) hours after dosing. The mean (SD) AUC(0 to 48) values were 54.6 (26.8) and 53.1 (23.5) hr∙ng/mL, and corresponded to a bioavailability of 103% for the tablet relative to the solution.
After oral administration of terbutaline, 51 to 62 mcg/kg of body weight, to 3 healthy male subjects, peak serum levels of 3.1 to 6.2 ng/mL were observed 1 to 3 hours later. In the same study, after 3 days only 30% to 50% of the dose was recovered from urine and the remainder from the feces, which may indicate poor absorption.
After an oral dose to asthmatic patients, the elimination half-life of terbutaline was approximately 3.4 hours.
In comparison to oral dosing, subcutaneous administration of 0.5 mg of terbutaline sulfate to 17 healthy, adult, male subjects resulted in a mean (SD) peak plasma terbutaline concentration of 9.6 (3.6) ng/mL, which was observed at a median (range) time of 0.5 (0.08 to 1.0) hours after dosing. The mean (SD) AUC (0 to 48) and total body clearance values were 29.4 (14.2) hr∙ng/mL, and 311 (112) mL/min, respectively. The terminal half-life was determined in 9 of the 17 subjects and had a mean (SD) of 5.7 (2.0) hours.
About 90% of the drug was excreted in the urine at 96 hours after subcutaneous administration, with about 60% of this being unchanged drug. The sulfate conjugate is a major metabolite of terbutaline, and urinary excretion is the primary route of elimination.
There are no reports of any clinical pharmacokinetic studies investigating dose proportionality, effect of food, or special population studies with terbutaline.
- INDICATIONS AND USAGE
-
CONTRAINDICATIONS
1. Tocolysis
Oral terbutaline sulfate has not been approved and should not be used for acute or maintenance tocolysis (see BOXED WARNING: Tocolysis).
-
WARNINGS
Deterioration of Asthma
Asthma may deteriorate acutely over a period of hours or chronically over several days or longer. If the patient needs more doses of terbutaline sulfate than usual, this may be a marker of destabilization of asthma and requires reevaluation of the patient and the treatment regimen, giving special consideration to the possible need for anti-inflammatory treatment, e.g., corticosteroids.
Use of Anti-Inflammatory Agents
The use of beta-adrenergic agonist bronchodilators alone may not be adequate to control asthma in many patients. Early consideration should be given to adding anti-inflammatory agents, e.g., corticosteroids.
Cardiovascular Effects
Terbutaline sulfate, like all other beta-adrenergic agonists, can produce a clinically significant cardiovascular effect in some patients as measured by pulse rate, blood pressure, and/or symptoms. Although such effects are uncommon after administration of terbutaline sulfate at recommended doses, if they occur, the drug may need to be discontinued. In addition, beta-agonists have been reported to produce electrocardiogram (ECG) changes, such as flattening of the T wave, prolongation of the QTc interval, and ST segment depression. The clinical significance of these findings is unknown. Therefore, terbutaline sulfate, like all sympathomimetic amines, should be used with caution in patients with cardiovascular disorders, especially coronary insufficiency, cardiac arrhythmias, and hypertension.
-
PRECAUTIONS
General
Terbutaline, as with all sympathomimetic amines, should be used with caution in patients with cardiovascular disorders, including ischemic heart disease, hypertension, and cardiac arrhythmias; hyperthyroidism; diabetes mellitus; hypersensitivity to sympathomimetic amines; and convulsive disorders. Significant changes in systolic and diastolic blood pressure have been seen and could be expected to occur in some patients after use of any beta-adrenergic bronchodilator.
Immediate hypersensitivity reactions and exacerbation of bronchospasm have been reported after terbutaline administration.
Beta-adrenergic agonist medications may produce significant hypokalemia in some patients, possibly through intracellular shunting, which has the potential to produce adverse cardiovascular effects. The decrease is usually transient, not requiring supplementation.
Large doses of intravenous terbutaline sulfate have been reported to aggravate preexisting diabetes and ketoacidosis.
Information for Patients
The action of terbutaline sulfate should last up to 6 hours or longer. Terbutaline sulfate should not be used more frequently than recommended. Do not increase the dose or frequency of terbutaline sulfate without consulting your physician. If you find that treatment with terbutaline sulfate becomes less effective for symptomatic relief, your symptoms become worse, and/or you need to use the product more frequently than usual, you should seek medical attention immediately. While taking terbutaline sulfate, other inhaled drugs and asthma medications should be taken only as directed by your physician. Common adverse effects include palpitations, chest pain, rapid heart rate, tremor or nervousness. If you are pregnant or nursing, contact your physician about use of terbutaline sulfate. Effective and safe use of terbutaline sulfate includes an understanding of the way that it should be administered.
Drug Interactions
The concomitant use of terbutaline sulfate with other sympathomimetic agents is not recommended, since the combined effect on the cardiovascular system may be deleterious to the patient. However, this does not preclude the use of an aerosol bronchodilator of the adrenergic-stimulant type for the relief of an acute bronchospasm in patients receiving chronic oral therapy with terbutaline sulfate.
Monoamine Oxidase Inhibitors and Tricyclic Antidepressants
Terbutaline sulfate should be administered with extreme caution to patients being treated with monoamine oxidase inhibitors or tricyclic antidepressants, or within 2 weeks of discontinuation of such agents, since the action of terbutaline sulfate on the vascular system may be potentiated.
Beta-Blockers
Beta-adrenergic receptor blocking agents not only block the pulmonary effect of beta-agonists, such as terbutaline sulfate, but may produce severe bronchospasm in asthmatic patients. Therefore, patients with asthma should not normally be treated with beta-blockers. However, under certain circumstances, e.g., as prophylaxis after myocardial infarction, there may be no acceptable alternatives to the use of beta-adrenergic blocking agents in patients with asthma. In this setting, cardioselective beta-blockers could be considered, although they should be administered with caution.
Diuretics
The ECG changes and/or hypokalemia that may result from the administration of non-potassium sparing diuretics (such as loop or thiazide diuretics) can be acutely worsened by beta-agonists, especially when the recommended dose of the beta-agonist is exceeded. Although the clinical significance of these effects is not known, caution is advised in the co-administration of beta-agonists with non-potassium sparing diuretics.
Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 2-year study in Sprague-Dawley rats, terbutaline sulfate caused a significant and dose-related increase in the incidence of benign leiomyomas of the mesovarium at dietary doses of 50 mg/kg, and above (approximately 25 times the maximum recommended daily oral dose for adults on a mg/m2 basis). In a 21-month study in CD-1 mice, terbutaline sulfate showed no evidence of tumorigenicity at dietary doses up to 200 mg/kg (approximately 55 times the maximum recommended daily oral dose for adults on a mg/m2 basis). The mutagenicity potential of terbutaline sulfate has not been determined.
Reproduction studies in rats using terbutaline sulfate demonstrated no impairment of fertility at oral doses up to 50 mg/kg (approximately 25 times the maximum recommended daily oral dose for adults on a mg/m2 basis).
Pregnancy
Teratogenic Effects
Pregnancy Category C
There are no adequate and well-controlled studies of terbutaline sulfate in pregnant women. Published animal studies show that rat offspring exhibit alterations in behavior and brain development, including decreased cellular proliferation and differentiation when dams were treated subcutaneously with terbutaline during the late stage of pregnancy and lactation period. Terbutaline exposures in rat dams were approximately 6.5 times the common human dose in adults of 15 mg/day, on a mg/m2 basis.
Oral terbutaline sulfate has not been approved and should not be used for acute or maintenance tocolysis. In particular, terbutaline sulfate should not be used for tocolysis in the outpatient or home setting. Serious adverse reactions, including death, have been reported after administration of terbutaline sulfate to pregnant women. In the mother, these adverse reactions include increased heart rate, transient hyperglycemia, hypokalemia, cardiac arrhythmias, pulmonary edema and myocardial ischemia. Increased fetal heart rate and neonatal hypoglycemia may occur as a result of maternal administration (see BOXED WARNING: Tocolysis and CONTRAINDICATIONS, Tocolysis).
In animal embryofetal development studies, no teratogenic effects were observed in offspring when pregnant rats and rabbits received terbutaline sulfate at oral doses up to 50 mg/kg/day, approximately 32 and 65 times, respectively, the maximum recommended daily oral dose for adults, on a mg/m2 basis.
Terbutaline sulfate should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Use in Labor and Delivery
Because of the potential for beta-agonist interference with uterine contractility, use of terbutaline sulfate for relief of bronchospasm during labor should be restricted to those patients in whom the benefits clearly outweigh the risk.
Terbutaline crosses the placenta. After single-dose IV (intra-venous) administration of terbutaline to 22 women in late pregnancy who were delivered by elective Cesarean section due to clinical reasons, umbilical blood levels of terbutaline were found to range from 11% to 48% of the maternal blood levels.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Therefore, terbutaline sulfate should be used during nursing only if the potential benefit justifies the possible risk to the newborn.
Pediatric Use
Terbutaline sulfate is not recommended for patients under the age of 12 years because of insufficient clinical data to establish safety and effectiveness (see DOSAGE AND ADMINISTRATION).
-
ADVERSE REACTIONS
Adverse reactions observed with terbutaline sulfate are similar to those commonly seen with other sympathomimetic amines. All of these reactions are generally transient in nature and usually do not require treatment. The frequency of these side effects appears to diminish with continued therapy.
The following table lists the adverse reactions seen in 199 patients treated with terbutaline sulfate tablets during six double-blind crossover studies and four double-blind parallel studies (short- and long-term) performed in the United States.
Percent Incidence of Adverse Reactions (Total Daily Dosage Range 5 mg to 15 mg) Terbutaline N=199
Reaction
%
Nervous System
Nervousness
35.0
Tremor
15.0
Somnolence
5.5
Dizziness
3.5
Anxiety
1.0
Insomnia
1.5
Cardiovascular
Palpitations
5.0
Tachycardia
3.5
Extrasystoles ventricular
1.5
Vasodilations
1.0
Digestive
Nausea
3.0
Dry Mouth
1.5
Body as a Whole
Headache
7.5
Asthenia
2.0
Skin and Appendages
Sweating
1.0
The following adverse effects each occurred in fewer than 1% of patients: hallucinations, rash, paresthesia, hypertonia, (muscle cramps), vomiting.
There have been rare reports of elevation in liver enzymes and of hypersensitivity vasculitis.
To report SUSPECTED ADVERSE REACTIONS, contact Amneal Pharmaceuticals at 1-877-835-5472 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
-
DOSAGE AND ADMINISTRATION
Adults
The usual oral dose of terbutaline sulfate for adults is 5 mg administered at approximately six-hour intervals, three times daily, during the hours the patient is usually awake. If side effects are particularly disturbing, the dose may be reduced to 2.5 mg three times daily, and still provide a clinically significant improvement in pulmonary function. The total dose within 24 hours should not exceed 15 mg.
Children
Terbutaline sulfate is not recommended for use in children below the age of 12 years. A dosage of 2.5 mg three times daily is recommended for children 12 to 15 years of age. The total dose within 24 hours should not exceed 7.5 mg.
If a previously effective dosage regimen fails to provide the usual relief, medical advice should be sought immediately as this is often a sign of seriously worsening asthma that would require reassessment of therapy.
-
OVERDOSAGE
The median subcutaneous lethal dose of terbutaline sulfate in mature rats is approximately 165 mg/kg (approximately 90 times the maximum recommended daily oral dose for adults on a mg/m2 basis). The median subcutaneous lethal dose of terbutaline sulfate in young rats is approximately 2000 mg/kg (approximately 1100 times the maximum recommended daily oral dose for adults on a mg/m2 basis).
The expected symptoms with overdosage are those of excessive beta-adrenergic stimulation and/or occurrence or exaggeration of any of the symptoms listed under ADVERSE REACTIONS, e.g., seizures, angina, hypertension or hypotension, tachycardia with rates up to 200 beats per minute, arrhythmias, nervousness, headache, tremor, dry mouth, palpitation, nausea, dizziness, fatigue, malaise, and insomnia. Hypokalemia may also occur.
There is no specific antidote. Treatment consists of discontinuation of terbutaline sulfate together with appropriate symptomatic therapy. The judicious use of a cardioselective beta-receptor blocker may be considered, bearing in mind that such medication can produce bronchospasm. There is insufficient evidence to determine if dialysis is beneficial for overdosage of terbutaline sulfate.
In the alert patient who has taken excessive oral medication, the stomach should be emptied by induced emesis followed by lavage. In the unconscious patient, the airway should be secured with a cuffed endotracheal tube before lavage, and emesis should not be induced. Instillation of activated charcoal slurry may help reduce absorption of terbutaline. Adequate respiratory exchange should be maintained, and cardiac and respiratory support provided as needed. The patient should be monitored until signs and symptoms of overdosage have subsided.
-
HOW SUPPLIED
Terbutaline sulfate tablets, USP are packaged in bottles of 100 and 500 tablets. Descriptions of the 2.5 mg and 5 mg tablets follow:
Tablets 2.5 mg–Each off-white, oval, convex tablet debossed with "G" on one side and "2611" on the other side
Bottles of 100: NDC 0115-2611-01
Bottles of 500: NDC 0115-2611-02
Tablets 5 mg–Each off-white, round, convex tablet debossed with "G" on one side and "2622" on the other side
Bottles of 100: NDC 0115-2622-01
Bottles of 500: NDC 0115-2622-02
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 2.5 mg Tablet Bottle Label
- PRINCIPAL DISPLAY PANEL - 5 mg Tablet Bottle Label
-
INGREDIENTS AND APPEARANCE
TERBUTALINE SULFATE
terbutaline sulfate tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0115-2611 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TERBUTALINE SULFATE (UNII: 576PU70Y8E) (TERBUTALINE - UNII:N8ONU3L3PG) TERBUTALINE SULFATE 2.5 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) HYPROMELLOSE 2910 (6 MPA.S) (UNII: 0WZ8WG20P6) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) STARCH, CORN (UNII: O8232NY3SJ) POVIDONE (UNII: FZ989GH94E) MAGNESIUM STEARATE (UNII: 70097M6I30) Product Characteristics Color WHITE (off-white) Score no score Shape OVAL (convex) Size 11mm Flavor Imprint Code G;2611 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0115-2611-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 06/26/2001 2 NDC:0115-2611-02 500 in 1 BOTTLE; Type 0: Not a Combination Product 06/26/2001 3 NDC:0115-2611-03 1000 in 1 BOTTLE; Type 0: Not a Combination Product 06/26/2001 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA075877 06/26/2001 TERBUTALINE SULFATE
terbutaline sulfate tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0115-2622 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TERBUTALINE SULFATE (UNII: 576PU70Y8E) (TERBUTALINE - UNII:N8ONU3L3PG) TERBUTALINE SULFATE 5 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) HYPROMELLOSE 2910 (6 MPA.S) (UNII: 0WZ8WG20P6) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) STARCH, CORN (UNII: O8232NY3SJ) POVIDONE (UNII: FZ989GH94E) MAGNESIUM STEARATE (UNII: 70097M6I30) Product Characteristics Color WHITE (off-white) Score no score Shape ROUND (convex) Size 8mm Flavor Imprint Code G;2622 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0115-2622-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 06/26/2001 2 NDC:0115-2622-02 500 in 1 BOTTLE; Type 0: Not a Combination Product 06/26/2001 3 NDC:0115-2622-03 1000 in 1 BOTTLE; Type 0: Not a Combination Product 06/26/2001 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA075877 06/26/2001 Labeler - Amneal Pharmaceuticals of New York LLC (123797875) Establishment Name Address ID/FEI Business Operations Amneal Pharmaceuticals Private Limited Oral Solid Dosage Unit 650762060 ANALYSIS(0115-2611, 0115-2622) , LABEL(0115-2611, 0115-2622) , MANUFACTURE(0115-2611, 0115-2622) , PACK(0115-2611, 0115-2622)




