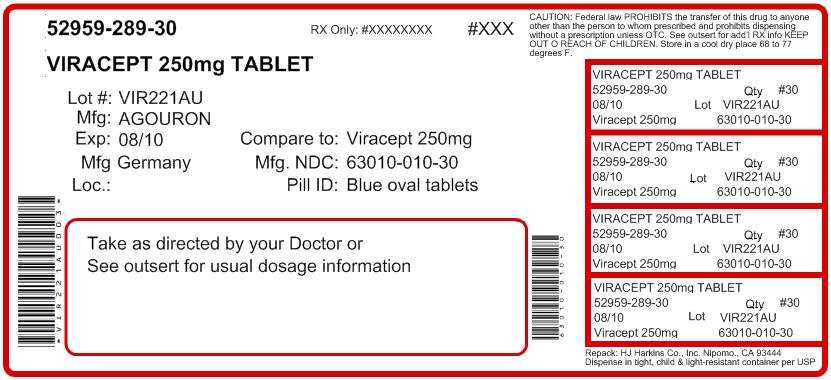
Label: VIRACEPT- nelfinavir mesylate tablet, film coated
-
Contains inactivated NDC Code(s)
NDC Code(s): 52959-289-06, 52959-289-30 - Packager: H.J. Harkins Company, Inc.
- This is a repackaged label.
- Source NDC Code(s): 63010-010
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated April 18, 2012
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
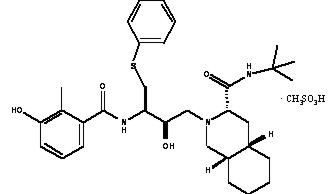
VIRACEPT® (nelfinavir mesylate) is an inhibitor of the human immunodeficiency virus (HIV) protease. VIRACEPT Tablets are available for oral administration as a light blue, capsule-shaped tablet with a clear film coating in 250 mg strength (as nelfinavir free base) and as a white oval tablet with a clear film coating in 625 mg strength (as nelfinavir free base). Each tablet contains the following common inactive ingredients: calcium silicate, crospovidone, magnesium stearate, hypromellose, and triacetin. In addition, the 250 mg tablet contains FD&C blue #2 powder and the 625 mg tablet contains colloidal silicon dioxide. VIRACEPT Oral Powder is available for oral administration in a 50 mg/g strength (as nelfinavir free base) in bottles. The oral powder also contains the following inactive ingredients: microcrystalline cellulose, maltodextrin, dibasic potassium phosphate, crospovidone, hypromellose, aspartame, sucrose palmitate, and natural and artificial flavor. The chemical name for nelfinavir mesylate is [3S-[2(2S*, 3S*), 3α,4aβ,8aβ]]-N-(1,1-dimethylethyl)decahydro-2-[2-hydroxy-3-[(3-hydroxy-2-methylbenzoyl)amino]-4-(phenylthio)butyl]-3-isoquinoline carboxamide mono-methanesulfonate (salt) and the molecular weight is 663.90 (567.79 as the free base). Nelfinavir mesylate has the following structural formula:
Nelfinavir mesylate is a white to off-white amorphous powder, slightly soluble in water at pH ≤4 and freely soluble in methanol, ethanol, 2-propanol and propylene glycol.
-
MICROBIOLOGY
Mechanism of Action
Nelfinavir is an inhibitor of the HIV-1 protease. Inhibition of the viral protease prevents cleavage of the gag and gag-pol polyprotein resulting in the production of immature, non-infectious virus.
Antiviral Activity In Vitro
The antiviral activity of nelfinavir in vitro has been demonstrated in both acute and/or chronic HIV infections in lymphoblastoid cell lines, peripheral blood lymphocytes and monocytes/macrophages. Nelfinavir was found to be active against several laboratory strains and clinical isolates of HIV-1 and the HIV-2 strain ROD. The EC95 (95% effective concentration) of nelfinavir ranged from 7 to 196 nM. Drug combination studies with protease inhibitors showed nelfinavir had antagonistic interactions with indinavir, additive interactions with ritonavir or saquinavir and synergistic interactions with amprenavir and lopinavir. Minimal to no cellular cytotoxicity was observed with any of these protease inhibitors alone or in combination with nelfinavir. In combination with reverse transcriptase inhibitors, nelfinavir demonstrated additive (didanosine or stavudine) to synergistic (abacavir, delavirdine, efavirenz, emtricitabine, lamivudine, nevirapine, tenofovir, zalcitabine or zidovudine) antiviral activity in vitro without enhanced cytotoxicity. Nelfinavir's anti-HIV activity was not antagonized by the anti-HCV drug ribavirin.
Drug Resistance
HIV-1 isolates with reduced susceptibility to nelfinavir have been selected in vitro. HIV isolates from selected patients treated with nelfinavir alone or in combination with reverse transcriptase inhibitors were monitored for phenotypic (n=19) and genotypic (n=195, 157 of which were evaluable) changes in clinical trials over a period of 2 to 82 weeks. One or more viral protease mutations at amino acid positions 30, 35, 36, 46, 71, 77 and 88 were detected in the HIV-1 of >10% of patients with evaluable isolates. The overall incidence of the D30N mutation in the viral protease of evaluable isolates (n=157) from patients receiving nelfinavir monotherapy or nelfinavir in combination with zidovudine and lamivudine or stavudine was 54.8%. The overall incidence of other mutations associated with primary protease inhibitor resistance was 9.6% for the L90M substitution whereas substitutions at 48, 82, or 84 were not observed. Of the 19 clinical isolates for which both phenotypic and genotypic analyses were performed, 9 showed reduced susceptibility (5- to 93-fold) to nelfinavir in vitro. All 9 patient isolates possessed one or more mutations in the viral protease gene. Amino acid position 30 appeared to be the most frequent mutation site.
Cross-resistance
Non-clinical Studies
Patient-derived recombinant HIV isolates containing the D30N mutation (n=4) and demonstrating high-level (>10-fold) NFV-resistance remained susceptible (<2.5-fold resistance) to amprenavir, indinavir, lopinavir, and saquinavir, in vitro. Patient-derived recombinant HIV isolates containing the L90M mutation (n=8) demonstrated moderate to high-level resistance to NFV and had varying levels of susceptibility to amprenavir, indinavir, lopinavir, and saquinavir, in vitro. Most patient-derived recombinant isolates with phenotypic and genotypic evidence of reduced susceptibility (>2.5-fold) to amprenavir, indinavir, lopinavir, and/or saquinavir demonstrated high-level cross-resistance to nelfinavir, in vitro. Mutations associated with resistance to other PIs (e.g. G48V, V82A/F/T, I84V, L90M) appeared to confer high-level cross-resistance to NFV. Following ritonavir therapy 6 of 7 clinical isolates with decreased ritonavir susceptibility (8- to 113-fold) in vitro compared to baseline also exhibited decreased susceptibility to nelfinavir in vitro (5- to 40-fold). Cross-resistance between nelfinavir and reverse transcriptase inhibitors is unlikely because different enzyme targets are involved. Clinical isolates (n=5) with decreased susceptibility to lamivudine, nevirapine or zidovudine remain fully susceptible to nelfinavir in vitro.
Clinical Studies
There have been no controlled or comparative studies evaluating the virologic response to subsequent protease inhibitor-containing regimens in patients who have demonstrated loss of virologic response to a nelfinavir-containing regimen. However, virologic response was evaluated in a single-arm prospective study of 26 patients with extensive prior antiretroviral experience with reverse transcriptase inhibitors (mean 2.9) who had received VIRACEPT for a mean duration of 59.7 weeks and were switched to a ritonavir (400 mg BID)/saquinavir hard-gel (400 mg BID) containing regimen after a prolonged period of VIRACEPT failure (median 48 weeks). Sequence analysis of HIV-1 isolates prior to switch demonstrated a D30N or an L90M substitution in 18 and 6 patients, respectively. Subjects remained on therapy for a mean of 48 weeks (range 40 to 56 weeks) where 17 of 26 (65%) subjects and 13 of 26 (50%) subjects were treatment responders with HIV RNA below the assay limit of detection (<500 HIV RNA copies/mL, Chiron bDNA) at 24 and 48 weeks, respectively.
-
CLINICAL PHARMACOLOGY
Pharmacokinetics
The pharmacokinetic properties of nelfinavir were evaluated in healthy volunteers and HIV-infected patients; no substantial differences were observed between the two groups.
Absorption
Pharmacokinetic parameters of nelfinavir (area under the plasma concentration-time curve during a 24-hour period at steady-state [AUC24], peak plasma concentrations [Cmax], morning and evening trough concentrations [Ctrough]) from a pharmacokinetic study in HIV-positive patients after multiple dosing with 1250 mg (five 250 mg tablets) twice daily (BID) for 28 days (10 patients) and 750 mg (three 250 mg tablets) three times daily (TID) for 28 days (11 patients) are summarized in Table 1.
Table 1 Summary of a Pharmacokinetic Study in HIV-positive Patients with Multiple Dosing of 1250 mg (five 250 mg tablets) BID for 28 days and 750 mg (three 250 mg tablets) TID for 28 days Regimen AUC24
mg.h/LCmax
mg/LCtrough
Morning
mg/LCtrough
Afternoon or Evening
mg/Ldata are mean ± SD 1250 mg BID 52.8 ± 15.7 4.0 ± 0.8 2.2 ± 1.3 0.7 ± 0.4 750 mg TID 43.6 ± 17.8 3.0 ± 1.6 1.4 ± 0.6 1.0 ± 0.5 The difference between morning and afternoon or evening trough concentrations for the TID and BID regimens was also observed in healthy volunteers who were dosed at precisely 8- or 12-hour intervals.
In healthy volunteers receiving a single 1250 mg dose, the 625 mg tablet was not bioequivalent to the 250 mg tablet formulation. Under fasted conditions (n=27), the AUC and Cmax were 34% and 24% higher, respectively, for the 625 mg tablets. In a relative bioavailability assessment under fed conditions (n=28), the AUC was 24% higher for the 625 mg tablet; the Cmax was comparable for both formulations. In HIV-1 infected subjects (N = 21) receiving multiple doses of 1250 mg BID under fed conditions, the 625 mg formulation was bioequivalent to the 250 mg formulation based on similarity in steady state exposure (Cmax and AUC).
Table 2 shows the summary of the steady state pharmacokinetic parameters (mean ± SD) of nelfinavir after multiple dose administration of 1250 mg BID (2 × 625 tablets) to HIV-infected patients (N = 21) for 14 days.
Table 2 Summary of the steady state pharmacokinetic parameters (mean ± SD) of nelfinavir after multiple dose administration of 1250 mg BID (2 × 625 tablets) to HIV-infected patients (N = 21) for 14 days. Regimen AUC12
mg.h/LCmax
mg/LCmin
mg/LAUC12: Steady state AUC Cmax: Maximum plasma concentration at steady state Cmin: Minimum plasma concentration at steady state 1250 mg BID 35.3 (16.4) 4.7 (1.9) 1.5 (1.0) In healthy volunteers receiving a single 750 mg dose under fed conditions, nelfinavir concentrations were similar following administration of the 250 mg tablet and oral powder.
Effect of Food on Oral Absorption
Food increases nelfinavir exposure and decreases nelfinavir pharmacokinetic variability relative to the fasted state. In one study, healthy volunteers received a single dose of 1250 mg of VIRACEPT 250 mg tablets (5 tablets) under fasted or fed conditions (three different meals). In a second study, healthy volunteers received single doses of 1250 mg VIRACEPT (5 × 250 mg tablets) under fasted or fed conditions (two different fat content meals). The results from the two studies are summarized in Table 3 and Table 4, respectively.
Table 3 Increase in AUC, Cmax and Tmax for Nelfinavir in Fed State Relative to Fasted State Following 1250 mg VIRACEPT (5 × 250 mg tablets) Number of Kcal % Fat Number of subjects AUC fold increase Cmax fold increase Increase in Tmax (hr) 125 20 n=21 2.2 2.0 1.00 500 20 n=22 3.1 2.3 2.00 1000 50 n=23 5.2 3.3 2.00 Table 4 Increase in Nelfinavir AUC, Cmax and Tmax in Fed Low Fat (20%) versus High Fat (50%) State Relative to Fasted State Following 1250 mg VIRACEPT (5 × 250 mg tablets) Number of Kcal % Fat Number of subjects AUC fold increase Cmax fold increase Increase in Tmax (hr) 500 20 n=22 3.1 2.5 1.8 500 50 n=22 5.1 3.8 2.1 Nelfinavir exposure can be increased by increasing the calorie or fat content in meals taken with VIRACEPT.
A food effect study has not been conducted with the 625 mg tablet. However, based on a cross-study comparison (n=26 fed vs. n=26 fasted) following single dose administration of nelfinavir 1250 mg, the magnitude of the food effect for the 625 mg nelfinavir tablet appears comparable to that of the 250 mg tablets. VIRACEPT should be taken with a meal.
Distribution
The apparent volume of distribution following oral administration of nelfinavir was 2–7 L/kg. Nelfinavir in serum is extensively protein-bound (>98%).
Metabolism
Unchanged nelfinavir comprised 82–86% of the total plasma radioactivity after a single oral 750 mg dose of 14C-nelfinavir. In vitro, multiple cytochrome P-450 enzymes including CYP3A and CYP2C19 are responsible for metabolism of nelfinavir. One major and several minor oxidative metabolites were found in plasma. The major oxidative metabolite has in vitro antiviral activity comparable to the parent drug.
Elimination
The terminal half-life in plasma was typically 3.5 to 5 hours. The majority (87%) of an oral 750 mg dose containing 14C-nelfinavir was recovered in the feces; fecal radioactivity consisted of numerous oxidative metabolites (78%) and unchanged nelfinavir (22%). Only 1–2% of the dose was recovered in urine, of which unchanged nelfinavir was the major component.
Special Populations
Hepatic Insufficiency
The steady-state pharmacokinetics of nelfinavir (1250 mg BID for 2 weeks) was studied in HIV-seronegative subjects with mild (Child-Pugh Class A; n=6) or moderate (Child-Pugh Class B; n=6) hepatic impairment. When compared with subjects with normal hepatic function, the Cmax and AUC of nelfinavir were not significantly different in subjects with mild hepatic impairment but were increased by 22% and 62%, respectively, in subjects with moderate hepatic impairment. The steady-state pharmacokinetics of nelfinavir has not been studied in HIV-seronegative subjects with severe hepatic impairment.
The steady-state pharmacokinetics of nelfinavir has not been studied in HIV-positive patients with any degree of hepatic impairment.
Renal Insufficiency
The pharmacokinetics of nelfinavir have not been studied in patients with renal insufficiency; however, less than 2% of nelfinavir is excreted in the urine, so the impact of renal impairment on nelfinavir elimination should be minimal.
Gender and Race
No significant pharmacokinetic differences have been detected between males and females. Pharmacokinetic differences due to race have not been evaluated.
Pediatrics
The pharmacokinetics of nelfinavir have been investigated in 5 studies in pediatric patients from birth to 13 years of age either receiving VIRACEPT three times or twice daily. The dosing regimens and associated AUC24 values are summarized in Table 5.
Table 5 Summary of Steady-state AUC24 of Nelfinavir in Pediatric Studies Protocol no. Dosing regimen* N† Age AUC24 (mg.hr/L)
arithmetic mean ± SDCtrough values are not presented in the table because they are not available for all studies AG1343-524 20 (19–28)
mg/kg
TID14 2–13 years 56.1 ± 29.8 PACTG-725 55 (48–60)
mg/kg
BID6 3–11 years 101.8 ± 56.1 PENTA 7 40 (34–43)
mg/kg
TID4 2–9 months 33.8 ± 8.9 PENTA 7 75 (55–83)
mg/kg
BID12 2–9 months 37.2 ± 19.2 PACTG-353 40 (14–56)
mg/kg
BID10 6 weeks 44.1 ± 27.4 1 week 45.8 ± 32.1 Pharmacokinetic data are also available for 86 patients (age 2 to 12 years) who received VIRACEPT 25–35 mg/kg TID in Study AG1343-556. The pharmacokinetic data from Study AG1343-556 were more variable than data from other studies conducted in the pediatric population; the 95% confidence interval for AUC24 was 9 to 121 mg.hr/L.
Overall, use of VIRACEPT in the pediatric population is associated with highly variable drug exposure. The high variability may be due to inconsistent food intake in pediatric patients. (See PRECAUTIONS: Pediatric Use, DOSAGE AND ADMINISTRATION.)
Drug Interactions
(also see CONTRAINDICATIONS, WARNINGS, PRECAUTIONS: Drug Interactions)
CYP3A and CYP2C19 appear to be the predominant enzymes that metabolize nelfinavir in humans. The potential ability of nelfinavir to inhibit the major human cytochrome P450 enzymes (CYP3A, CYP2C19, CYP2D6, CYP2C9, CYP1A2 and CYP2E1) has been investigated in vitro. Only CYP3A was inhibited at concentrations in the therapeutic range. Specific drug interaction studies were performed with nelfinavir and a number of drugs. Table 6 summarizes the effects of nelfinavir on the geometric mean AUC, Cmax and Cmin of coadministered drugs. Table 7 shows the effects of coadministered drugs on the geometric mean AUC, Cmax and Cmin of nelfinavir.
Table 6: Drug Interactions: Changes in Pharmacokinetic Parameters for Coadministered Drug in the Presence of VIRACEPT % Change of Coadministered Drug Pharmacokinetic Parameters* (90% CI) Coadministered Drug Nelfinavir Dose N AUC Cmax Cmin NA: Not relevant for single-dose treatment; ND: Cannot be determined - *
- ↑ Indicates increase ↓ Indicates decrease ↔ Indicates no change (geometric mean exposure increased or decreased < 10%)
- †
- Using the soft-gelatin capsule formulation of saquinavir 1200 mg
- ‡
- Based on non-definitive cross-study comparison, drug plasma concentrations appeared to be unaffected by coadministration
- §
- Rifabutin 150 mg qd changes are relative to Rifabutin 300 mg qd × 8 days without coadministration with nelfinavir
- ¶
- Comparable changes in rifabutin concentrations were observed with VIRACEPT 1250 mg q12h × 7 days
- #
- Changes are reported for total plasma methadone; changes for the individual R-enantiomer and S-enantiomer were similar
- Þ
- Phenytoin exposure measures are reported for total phenytoin exposure. The effect of nelfinavir on unbound phenytoin was similar
HIV-Protease Inhibitors Indinavir 800 mg Single Dose 750 mg q8h × 7 days 6 ↑51%
(↑29–↑77%)↓10%
(↓28–↑13%)NA Ritonavir 500 mg Single Dose 750 mg q8h × 5 doses 10 ↔ ↔ NA Saquinavir 1200 mg Single Dose † 750 mg tid × 4 days 14 ↑392%
(↑291–↑521%)↑179%
(↑117–↑259%)NA Amprenavir 800 mg tid × 14 days 750 mg tid × 14 days 6 ↔ ↓14%
(↓38–↑20%)↑189%
(↑52–↑448%)Nucleoside Reverse Transcriptase Inhibitors Lamivudine 150 mg Single Dose 750 mg q8h × 7–10 days 11 ↑10%
(↑2–↑18%)↑31%
(↑9–↑56%)NA Stavudine 30–40 mg bid × 56 days 750 mg tid × 56 days 8 See footnote ‡ Zidovudine 200 mg Single Dose 750 mg q8h × 7–10 days 11 ↓35%
(↓29–↓40%)↓31%
(↓13–↓46%)NA Non-Nucleoside Reverse Transcriptase Inhibitors Efavirenz 600 mg qd × 7 days 750 mg q8h × 7 days 10 ↓12%
(↓31–↑12%)↓12%
(↓29–↑8%)↓22%
(↓54–↑32%)Nevirapine 200 mg qd × 14 days ‡
Followed by 200 mg bid × 14 days750 mg tid × 36 days 23 See footnote ‡ Delavirdine 400 mg q8h × 14 days 750 mg q8h × 7 days 7 ↓31%
(↓57–↑10%)↓27%
(↓49–↑4%)↓33%
(↓70–↑49%)Anti-infective Agents Rifabutin 150 mg qd × 8 days § 750 mg q8h × 7–8 days ¶ 12 ↑83%
(↑72–↑96%)↑19%
(↑11–↑28%)↑177%
(↑144–↑215%)Rifabutin 300 mg qd × 8 days 750 mg q8h × 7–8 days 10 ↑207%
(↑161–↑263%)↑146%
(↑118–↑178%)↑305%
(↑245–↑375%)Azithromycin 1200 mg Single Dose 750 mg tid × 11 days 12 ↑112%
(↑80–↑150%)↑136%
(↑77–↑215%)NA HMG-CoA Reductase Inhibitors Atorvastatin 10 mg qd × 28 days 1250 mg bid × 14 days 15 ↑74%
(↑41–↑116%)↑122%
(↑68–↑193%)↑39%
(↓21–↑145%)Simvastatin 20 mg qd × 28 days 1250 mg bid × 14 days 16 ↑505%
(↑393–↑643%)↑517%
(↑367–↑715%)ND Other Agents Ethinyl estradiol 35 µg qd × 15 days 750 mg q8h × 7 days 12 ↓47%
(↓42–↓52%)↓28%
(↓16–↓37%)↓62%
(↓57–↓67%)Norethindrone 0.4 mg qd × 15 days 750 mg q8h × 7 days 12 ↓18%
(↓13–↓23%)↔ ↓46%
(↓38–↓53%)Methadone 80 mg + / - 21 mg qd # > 1 month 1250 mg bid × 8 days 13 ↓47%
(↓42–↓51%)↓46%
(↓42–↓49%)↓53%
(↓49–↓57%)Phenytoin 300 mg qd × 14 days Þ 1250 mg bid × 7 days 12 ↓29%
(↓17–↓39%)↓21%
(↓12–↓29%)↓39%
(↓27–↓49%)Table 7: Drug Interactions: Changes in Pharmacokinetic Parameters for Nelfinavir in the Presence of the Coadministered Drug % Change of Nelfinavir Pharmacokinetic Parameters* (90% CI) Coadministered Drug Nelfinavir Dose N AUC Cmax Cmin NA: Not relevant for single-dose treatment - *
- ↑ Indicates increase ↓ Indicates decrease ↔ Indicates no change (geometric mean exposure increased or decreased < 10%)
- †
- Using the soft-gelatin capsule formulation of saquinavir 1200 mg
- ‡
- Based on non-definitive cross-study comparison, nelfinavir plasma concentrations appeared to be unaffected by coadministration
HIV-Protease Inhibitors Indinavir 800 mg q8h × 7 days 750 mg Single Dose 6 ↑83%
(↑42–↑137%)↑31%
(↑16–↑48%)NA Ritonavir 500 mg q12h × 3 doses 750 mg Single Dose 10 ↑152%
(↑96–↑224%)↑44%
(↑28–↑63%)NA Saquinavir 1200 mg tid × 4 days † 750 mg Single Dose 14 ↑18%
(↑7–↑30%)↔ NA Amprenavir 800 mg tid × 14 days 750 mg tid × 14 days 6 See footnote ‡ Nucleoside Reverse Transcriptase Inhibitors Didanosine 200 mg Single Dose 750 mg Single Dose 9 ↔ ↔ NA Zidovudine 200 mg + Lamivudine 150 mg Single Dose 750 mg q8h × 7–10 days 11 ↔ ↔ ↔ Non-Nucleoside Reverse Transcriptase Inhibitors Efavirenz 600 mg qd × 7 days 750 mg q8h × 7 days 7 ↑20%
(↑8–↑34%)↑21%
(↑10–↑33%)↔ Nevirapine 200 mg qd × 14 days
Followed by 200 mg bid × 14 days750 mg tid × 36 days 23 ↔ ↔ ↓32%
(↓50–↑5%)Delavirdine 400 mg q8h × 7 days 750 mg q8h × 14 days 12 ↑107%
(↑83–↑135%)↑88%
(↑66–↑113%)↑136%
(↑103–↑175%)Anti-infective Agents Ketoconazole 400 mg qd × 7 days 500 mg q8h × 5–6 days 12 ↑35%
(↑24–↑46%)↑25%
(↑11–↑40%)↑14%
(↓23–↑69%)Rifabutin 150 mg qd × 8 days 750 mg q8h × 7–8 days 11 ↓23%
(↓14–↓31%)↓18%
(↓8–↓27%)↓25%
(↓8–↓39%)1250 mg q12h × 7–8 days 11 ↔ ↔ ↓15%
(↓43–↑27%)Rifabutin 300 mg qd × 8 days 750 mg q8h × 7–8 days 10 ↓32%
(↓15–↓46%)↓24%
(↓10–↓36%)↓53%
(↓15–↓73%)Rifampin 600 mg qd × 7 days 750 mg q8h × 5–6 days 12 ↓83%
(↓79–↓86%)↓76%
(↓69–↓82%)↓92%
(↓86–↓95%)Azithromycin 1200 mg Single Dose 750 mg tid × 9 days 12 ↓15%
(↓7–↓22%)↓10%
(↓19–↑1%)↓29%
(↓19–↓38%)HMG-CoA Reductase Inhibitors Atorvastatin 10 mg qd × 28 days 1250 mg bid × 14 days 15 See footnote ‡ Simvastatin 20 mg qd × 28 days 1250 mg bid × 14 days 16 See footnote ‡ Other Agents Methadone 80 mg + / - 21 mg qd > 1 month 1250 mg bid × 8 days 13 See footnote ‡ Phenytoin 300 mg qd × 7 days 1250 mg bid × 14 days 15 ↔ ↔ ↓18%
(↓45–↑23%)Omeprazole 40 mg qd × 4 days administered 30 minutes before nelfinavir 1250 mg bid × 4 days 19 ↓36%
(↓20–↓49%)↓37%
(↓23–↓49%)↓39%
(↓15–↓57%)For information regarding clinical recommendations see CONTRAINDICATIONS, WARNINGS, PRECAUTIONS: Drug Interactions.
-
INDICATIONS AND USAGE
VIRACEPT in combination with other antiretroviral agents is indicated for the treatment of HIV infection.
Description of Studies
In the clinical studies described below, efficacy was evaluated by the percent of patients with plasma HIV RNA < 400 copies/mL (Studies 511 and 542) or < 500 copies/mL (Study ACTG 364), using the Roche RT-PCR (Amplicor) HIV-1 Monitor or < 50 copies/mL, using the Roche HIV-1 Ultrasensitive assay (Study Avanti 3). In the analysis presented in each figure, patients who terminated the study early for any reason, switched therapy due to inadequate efficacy or who had a missing HIV-RNA measurement that was either preceded or followed by a measurement above the limit of assay quantification were considered to have HIV-RNA above 400 copies/mL, above 500 copies/mL, or above 50 copies/mL at subsequent time points, depending on the assay that was used.
a. Studies in Antiretroviral Treatment Naive Patients
Study 511: VIRACEPT + zidovudine + lamivudine versus zidovudine + lamivudine
Study 511 was a double-blind, randomized, placebo-controlled trial comparing treatment with zidovudine (ZDV; 200 mg TID) and lamivudine (3TC; 150 mg BID) plus 2 doses of VIRACEPT (750 mg and 500 mg TID) to zidovudine (200 mg TID) and lamivudine (150 mg BID) alone in 297 antiretroviral naive HIV-1 infected patients (median age 35 years [range 21 to 63], 89% male and 78% Caucasian). Mean baseline CD4 cell count was 288 cells/mm3 and mean baseline plasma HIV RNA was 5.21 log10 copies/mL (160,394 copies/mL). The percent of patients with plasma HIV RNA < 400 copies/mL and mean changes in CD4 cell count are summarized in Figures 1 and 2, respectively.
Figure 1
Study 511: Percentage of Patients With HIV RNA Below 400 Copies/mL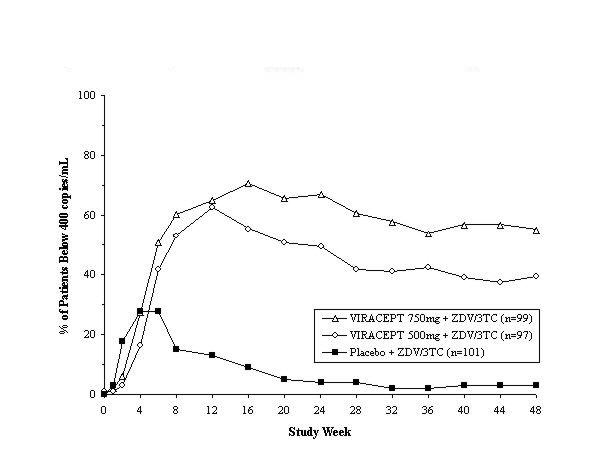
Figure 2
Study 511: Mean Change From Baseline in CD4 Cell Counts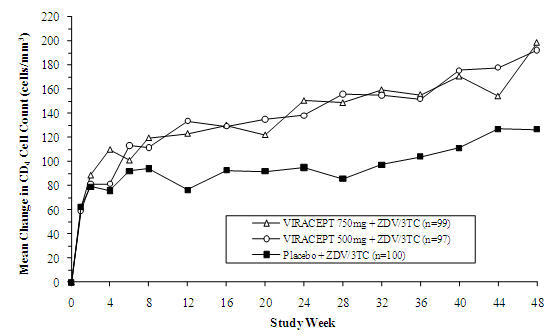
Study 542: VIRACEPT BID + stavudine + lamivudine compared to VIRACEPT TID + stavudine + lamivudine
Study 542 is an ongoing, randomized, open-label trial comparing the HIV RNA suppression achieved by VIRACEPT 1250 mg BID versus VIRACEPT 750 mg TID in patients also receiving stavudine (d4T; 30–40 mg BID) and lamivudine (3TC; 150 mg BID). Patients had a median age of 36 years (range 18 to 83), were 84% male, and were 91% Caucasian. Patients had received less than 6 months of therapy with nucleoside transcriptase inhibitors and were naïve to protease inhibitors. Mean baseline CD4 cell count was 296 cells/mm3 and mean baseline plasma HIV RNA was 5.0 log10 copies/mL (100,706 copies/mL).
Results showed that there was no significant difference in mean CD4 cell count among treatment groups; the mean increases from baseline for the BID and TID arms were 150 cells/mm3 at 24 weeks and approximately 200 cells/mm3 at 48 weeks.
The percent of patients with HIV RNA < 400 copies/mL is summarized in Figure 3. The outcomes of patients through 48 weeks of treatment are summarized in Table 8.
Figure 3
Study 542: Percentage of Patients With HIV RNA Below 400 Copies/mL
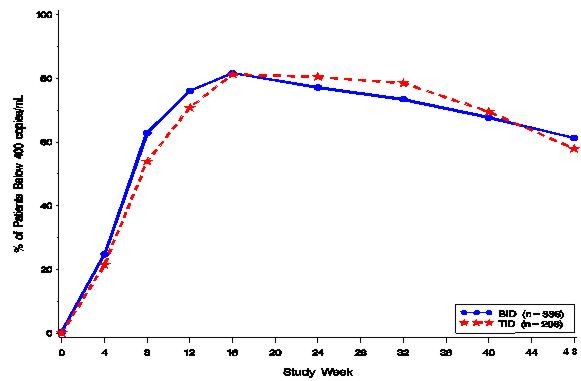
Table 8 Outcomes of Randomized Treatment Through 48 Weeks Outcome VIRACEPT 1250 mg BID Regimen VIRACEPT 750 mg TID Regimen - *
- Twelve patients in the BID arm and fourteen patients in the TID arm had not yet reached 48 weeks of therapy.
- †
- These rates only reflect dose-limiting toxicities that were counted as the initial reason for treatment failure in the analysis (see ADVERSE REACTIONS for a description of the safety profile of these regimens).
- ‡
- Consent withdrawn, lost to follow-up, intercurrent illness, noncompliance or missing data; all assumed as failures.
Number of patients evaluable* 323 192 HIV RNA < 400
copies/mL198 (61%) 111 (58%) HIV RNA ≥ 400
copies/mL46 (14%) 22 (11%) Discontinued due to VIRACEPT toxicity† 9 (3%) 2 (1%) Discontinued due to other antiretroviral agents' toxicity† 3 (1%) 3 (2%) Others‡ 67 (21%) 54 (28%) Study Avanti 3: VIRACEPT TID + zidovudine + lamivudine compared to zidovudine + lamivudine
Study Avanti 3 was a placebo-controlled, randomized, double-blind study designed to evaluate the safety and efficacy of VIRACEPT (750 mg TID) in combination with zidovudine (ZDV; 300 mg BID) and lamivudine (3TC; 150 mg BID) (n=53) versus placebo in combination with ZDV and 3TC (n=52) administered to antiretroviral-naive patients with HIV infection and a CD4 cell count between 150 and 500 cells/µL. Patients had a mean age of 35 (range 22–59), were 89% male, and 88% Caucasian. Mean baseline CD4 cell count was 304 cells/mm3 and mean baseline plasma HIV RNA was 4.8 log10 copies/mL (57,887 copies/mL). The percent of patients with plasma HIV RNA < 50 copies/mL at 52 weeks was 54% for the VIRACEPT + ZDV + 3TC treatment group and 13% for the ZDV + 3TC treatment group.
b. Studies in Antiretroviral Treatment Experienced Patients
Study ACTG 364: VIRACEPT TID + 2NRTIs compared to efavirenz + 2NRTIs compared to VIRACEPT + efavirenz + 2NRTIs
Study ACTG 364 was a randomized, double-blind study that evaluated the combination of VIRACEPT 750 mg TID and/or efavirenz 600 mg QD with 2 NRTIs (either didanosine [ddI] + d4T, ddI + 3TC, or d4T + 3TC) in patients with prolonged prior nucleoside exposure who had completed 2 previous ACTG studies. Patients had a mean age of 41 years (range 18 to 75), were 88% male, and were 74% Caucasian. Mean baseline CD4 cell count was 389 cells/mm3 and mean baseline plasma HIV RNA was 3.9 log10 copies/mL (7,954 copies/mL).
The percent of patients with plasma HIV RNA < 500 copies/mL at 48 weeks was 42%, 62%, and 72% for the VIRACEPT (n=66), EFV (n=65), and VIRACEPT + EFV (n=64) treatment groups, respectively. The 4-drug combination of VIRACEPT + EFV + 2 NRTIs was more effective in suppressing plasma HIV RNA in these patients than either 3-drug regimen.
-
CONTRAINDICATIONS
VIRACEPT is contraindicated in patients with clinically significant hypersensitivity to any of its components.
Coadministration of VIRACEPT is contraindicated with drugs that are highly dependent on CYP3A for clearance and for which elevated plasma concentrations are associated with serious and/or life-threatening events. These drugs are listed in Table 9.
Table 9 Drugs That Are Contraindicated With VIRACEPT Drug Class Drugs Within Class That Are Contraindicated With VIRACEPT Alpha 1-adrenoreceptor antagonist Alfuzosin Antiarrhythmics Amiodarone, Quinidine Ergot Derivatives Dihydroergotamine, Ergonovine, Ergotamine, Methylergonovine Neuroleptic Pimozide PDE5 inhibitors Sildenafil (REVATIO®) [for treatment of pulmonary arterial hypertension] Sedative/Hypnotics Midazolam, Triazolam -
WARNINGS
ALERT: Find out about medicines that should not be taken with VIRACEPT. This statement is included on the product's bottle label.
Drug Interactions
(also see PRECAUTIONS)
Nelfinavir is an inhibitor of the CYP3A enzyme. Coadministration of VIRACEPT and drugs primarily metabolized by CYP3A may result in increased plasma concentrations of the other drug that could increase or prolong its therapeutic and adverse effects. Caution should be exercised when inhibitors of CYP3A, including VIRACEPT, are coadministered with drugs that are metabolized by CYP3A and that prolong the QT interval. (See ADVERSE REACTIONS: Post-Marketing Experience.) Nelfinavir is metabolized by CYP3A and CYP2C19. Coadministration of VIRACEPT and drugs that induce CYP3A or CYP2C19 may decrease nelfinavir plasma concentrations and reduce its therapeutic effect. Coadministration of VIRACEPT and drugs that inhibit CYP3A or CYP2C19 may increase nelfinavir plasma concentrations. (Also see PRECAUTIONS: Table 10: Drugs That Should Not Be Coadministered With VIRACEPT, Table 11: Established and Other Potentially Significant Drug Interactions.)
Concomitant use of VIRACEPT with lovastatin or simvastatin is not recommended. Caution should be exercised if HIV protease inhibitors, including VIRACEPT, are used concurrently with other HMG-CoA reductase inhibitors that are also metabolized by the CYP3A pathway (e.g., atorvastatin). (Also see Tables 6 and 7: Drug Interactions). The risk of myopathy including rhabdomyolysis may be increased when protease inhibitors, including VIRACEPT, are used in combination with these drugs.
Particular caution should be used when prescribing sildenafil, or other PDE5 inhibitors, in patients receiving protease inhibitors, including VIRACEPT. Coadministration of these drugs is expected to substantially increase PDE5 inhibitor concentrations and may result in an increase in PDE5 inhibitor-associated adverse events, including hypotension, visual changes, and priapism. (See PRECAUTIONS, Drug Interactions and Information For Patients, and the complete prescribing information for sildenafil and other PDE5 inhibitors.)
Concomitant use of St. John's wort (hypericum perforatum) or St. John's wort-containing products and VIRACEPT is not recommended. Coadministration of St. John's wort with protease inhibitors, including VIRACEPT, is expected to substantially decrease protease inhibitor concentrations and may result in sub-optimal levels of VIRACEPT and lead to loss of virologic response and possible resistance to VIRACEPT or to the class of protease inhibitors.
Patients with Phenylketonuria
Patients with Phenylketonuria: VIRACEPT Oral Powder contains 11.2 mg phenylalanine per gram of powder.
Diabetes mellitus/Hyperglycemia
New onset diabetes mellitus, exacerbation of pre-existing diabetes mellitus and hyperglycemia have been reported during post-marketing surveillance in HIV-infected patients receiving protease inhibitor therapy. Some patients required either initiation or dose adjustments of insulin or oral hypoglycemic agents for treatment of these events. In some cases diabetic ketoacidosis has occurred. In those patients who discontinued protease inhibitor therapy, hyperglycemia persisted in some cases. Because these events have been reported voluntarily during clinical practice, estimates of frequency cannot be made and a causal relationship between protease inhibitor therapy and these events has not been established.
-
PRECAUTIONS
General
Nelfinavir is principally metabolized by the liver. See DOSAGE AND ADMINISTRATION when administering this drug to patients with hepatic impairment.
Resistance/Cross Resistance
HIV cross-resistance between protease inhibitors has been observed. (See MICROBIOLOGY.)
Hemophilia
There have been reports of increased bleeding, including spontaneous skin hematomas and hemarthrosis, in patients with hemophilia type A and B treated with protease inhibitors. In some patients, additional factor VIII was given. In more than half of the reported cases, treatment with protease inhibitors was continued or reintroduced. A causal relationship has not been established.
Fat Redistribution
Redistribution/accumulation of body fat including central obesity, dorsocervical fat enlargement ("buffalo hump"), peripheral wasting, facial wasting, breast enlargement, and "cushingoid appearance" have been observed in patients receiving antiretroviral therapy. The mechanism and long-term consequences of these events are currently unknown. A causal relationship has not been established.
Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including VIRACEPT. During the initial phase of combination antiretroviral treatment, patients whose immune system responds may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jirovecii pneumonia (PCP), or tuberculosis), which may necessitate further evaluation and treatment.
Information For Patients
A statement to patients and healthcare providers is included on the product's bottle label: ALERT: Find out about medicines that should NOT be taken with VIRACEPT. A Patient Package Insert (PPI) for VIRACEPT is available for patient information.
For optimal absorption, patients should be advised to take VIRACEPT with food (see CLINICAL PHARMACOLOGY: Pharmacokinetics and DOSAGE AND ADMINISTRATION).
Patients should be informed that VIRACEPT is not a cure for HIV infection and that they may continue to acquire illnesses associated with advanced HIV infection, including opportunistic infections.
Patients should be told that there is currently no data demonstrating that VIRACEPT therapy can reduce the risk of transmitting HIV to others through sexual contact or blood contamination.
Patients should be told that sustained decreases in plasma HIV RNA have been associated with a reduced risk of progression to AIDS and death. Patients should be advised to take VIRACEPT and other concomitant antiretroviral therapy every day as prescribed. Patients should not alter the dose or discontinue therapy without consulting with their doctor. If a dose of VIRACEPT is missed, patients should take the dose as soon as possible and then return to their normal schedule. However, if a dose is skipped, the patient should not double the next dose.
Patients should be informed that VIRACEPT Tablets are film-coated and that this film-coating is intended to make the tablets easier to swallow.
The most frequent adverse event associated with VIRACEPT is diarrhea, which can usually be controlled with non-prescription drugs, such as loperamide, which slow gastrointestinal motility.
Patients should be informed that redistribution or accumulation of body fat may occur in patients receiving antiretroviral therapy and that the cause and long-term health effects of these conditions are not known at this time.
VIRACEPT may interact with some drugs; therefore, patients should be advised to report to their doctor the use of any other prescription, non-prescription medication or herbal products, particularly St. John's wort.
Patients receiving oral contraceptives should be instructed that alternate or additional contraceptive measures should be used during therapy with VIRACEPT.
Patients receiving sildenafil, or other PDE5 inhibitors, and nelfinavir should be advised that they may be at an increased risk of PDE5 inhibitor-associated adverse events including hypotension, visual changes, and prolonged penile erection, and should promptly report any symptoms to their doctor.
Drug Interactions
(Also see CONTRAINDICATIONS, WARNINGS, CLINICAL PHARMACOLOGY: Drug Interactions)
Nelfinavir is an inhibitor of CYP3A. Coadministration of VIRACEPT and drugs primarily metabolized by CYP3A (e.g., dihydropyridine calcium channel blockers, HMG-CoA reductase inhibitors, immunosuppressants and PDE5 inhibitors) may result in increased plasma concentrations of the other drug that could increase or prolong both its therapeutic and adverse effects. (See Tables 10 and 11). Nelfinavir is metabolized by CYP3A and CYP2C19. Coadministration of VIRACEPT and drugs that induce CYP3A or CYP2C19, such as rifampin, may decrease nelfinavir plasma concentrations and reduce its therapeutic effect. Coadministration of VIRACEPT and drugs that inhibit CYP3A or CYP2C19 may increase nelfinavir plasma concentrations.
Drug interaction studies reveal no clinically significant drug interactions between nelfinavir and didanosine, lamivudine, stavudine, zidovudine, efavirenz, nevirapine, or ketoconazole and no dose adjustments are needed. In the case of didanosine, it is recommended that didanosine be administered on an empty stomach; therefore, nelfinavir should be administered with food one hour after or more than 2 hours before didanosine.
Based on known metabolic profiles, clinically significant drug interactions are not expected between VIRACEPT and dapsone, trimethoprim/sulfamethoxazole, or itraconazole.
Table 10 Drugs That Should Not Be Coadministered With VIRACEPT Drug Class: Drug Name Clinical Comment Alpha 1-adrenoreceptor antagonist:
alfuzosinPotentially increased alfuzosin concentrations can result in hypotension. Antiarrhythmics:
amiodarone, quinidineCONTRAINDICATED due to potential for serious and/or life threatening reactions such as cardiac arrhythmias. Antimycobacterial:
rifampinMay lead to loss of virologic response and possible resistance to VIRACEPT or other coadministered antiretroviral agents. Ergot Derivatives:
dihydroergotamine, ergonovine, ergotamine, methylergonovineCONTRAINDICATED due to potential for serious and/or life threatening reactions such as acute ergot toxicity characterized by peripheral vasospasm and ischemia of the extremities and other tissues. Herbal Products:
St. John's wort (hypericum perforatum)May lead to loss of virologic response and possible resistance to VIRACEPT or other coadministered antiretroviral agents. HMG-CoA Reductase Inhibitors:
lovastatin, simvastatinPotential for serious reactions such as risk of myopathy including rhabdomyolysis. Neuroleptic:
pimozideCONTRAINDICATED due to potential for serious and/or life threatening reactions such as cardiac arrhythmias. PDE5 inhibitor:
sildenafil (REVATIO) [for treatment of pulmonary arterial hypertension]A safe and effective dose has not been established when used with VIRACEPT. There is increased potential for sildenafil-associated adverse events (which include visual disturbances, hypotension, prolonged erection, and syncope). Proton Pump Inhibitors Omeprazole decreases the plasma concentrations of nelfinavir. Concomitant use of proton pump inhibitors and VIRACEPT may lead to a loss of virologic response and development of resistance. Sedative/Hypnotics:
midazolam, triazolamCONTRAINDICATED due to potential for serious and/or life threatening reactions such as prolonged or increased sedation or respiratory depression. Table 11 Established and Other Potentially Significant Drug Interactions: Alteration in Dose or Regimen May Be Recommended Based on Drug Interaction Studies (see CLINICAL PHARMACOLOGY, for Magnitude of Interaction, Tables 6 and 7) Concomitant Drug Class: Drug Name Effect on Concentration Clinical Comment HIV-Antiviral Agents Non-nucleoside Reverse Transcriptase Inhibitors: Appropriate doses for these combinations, with respect to safety and efficacy, have not been established. delavirdine ↑ nelfinavir
↓ delavirdinenevirapine ↓ nelfinavir (Cmin) Nucleoside Reverse Transcriptase Inhibitor:
didanosineIt is recommended that didanosine be administered on an empty stomach; therefore, didanosine should be given one hour before or two hours after VIRACEPT (given with food).
Protease Inhibitors: Appropriate doses for these combinations, with respect to safety and efficacy, have not been established. indinavir ↑ nelfinavir
↑ indinavirritonavir ↑ nelfinavir saquinavir ↑ saquinavir Other Agents Anti-coagulant:
warfarinwarfarin Coadministration of warfarin and VIRACEPT may affect concentrations of warfarin. It is recommended that the INR (international normalized ratio) be monitored carefully during treatment with VIRACEPT, especially when commencing therapy. Anti-convulsants: May decrease nelfinavir plasma concentrations. VIRACEPT may not be effective due to decreased nelfinavir plasma concentrations in patients taking these agents concomitantly. carbamazepine
phenobarbital↓ nelfinavir Anti-convulsant: Phenytoin plasma/serum concentrations should be monitored; phenytoin dose may require adjustment to compensate for altered phenytoin concentration. phenytoin ↓ phenytoin Anti-depressant: trazodone ↑ trazodone Concomitant use of trazodone and VIRACEPT may increase plasma concentrations of trazodone. Adverse events of nausea, dizziness, hypotension and syncope have been observed following coadministration of trazodone and ritonavir. If trazodone is used with a CYP3A4 inhibitor such as VIRACEPT, the combination should be used with caution and a lower dose of trazodone should be considered. Anti-gout
colchicine↑ colchicine Treatment of gout flares–
coadministration of colchicine in patients on VIRACEPT:
0.6 mg (1 tablet) × 1 dose, followed by 0.3 mg (half tablet) 1 hour later. Dose to be repeated no earlier than 3 days.
Prophylaxis of gout-flares–
coadministration of colchicine in patients on VIRACEPT:
If the original colchicine regimen was 0.6 mg twice a day, the regimen should be adjusted to 0.3 mg once a day.
If the original colchicine regimen was 0.6 mg once a day, the regimen should be adjusted to 0.3 mg once every other day.
Treatment of familial Mediterranean fever (FMF)–
coadministration of colchicine in patients on VIRACEPT:Maximum daily dose of 0.6 mg (may be given as 0.3 mg twice a day).
Patients with renal or hepatic impairment should not be given colchicine with VIRACEPT.Anti-Mycobacterial: It is recommended that the dose of rifabutin be reduced to one-half the usual dose when administered with VIRACEPT; 1250 mg BID is the preferred dose of VIRACEPT when coadministered with rifabutin. rifabutin ↑ rifabutin
↓ nelfinavir
(750 mg TID)
↔ nelfinavir
(1250 mg BID)Endothelin receptor antagonists:
bosentan↑ bosentan Coadministration of bosentan in patients on VIRACEPT or coadministration of VIRACEPT in patients on bosentan:
Start at or adjust bosentan to 62.5 mg once daily or every other day based upon individual tolerability.
HMG-CoA Reductase Inhibitor: Use lowest possible dose of atorvastatin or rosuvastatin with careful monitoring, or consider other HMG-CoA reductase inhibitors such as pravastatin or fluvastatin in combination with VIRACEPT. atorvastatin ↑ atorvastatin rosuvastatin ↑ rosuvastatin Immuno-suppressants: Plasma concentrations may be increased by VIRACEPT. cyclosporine
tacrolimus
sirolimus↑ immuno-suppressants Inhaled beta agonist:
salmeterol↑ salmeterol Concurrent administration of salmeterol with VIRACEPT is not recommended. The combination may result in increased risk of cardiovascular adverse events associated with salmeterol, including QT prolongation, palpitations and sinus tachycardia. Inhaled/nasal steroid: Fluticasone ↑ fluticasone Concomitant use of fluticasone propionate and VIRACEPT may increase plasma concentrations of fluticasone propionate. Use with caution. Consider alternatives to fluticasone propionate, particularly for long-term use. Macrolide Antibiotic:
azithromycin
↑ azithromycinDose adjustment of azithromycin is not recommended, but close monitoring for known side effects such as liver enzyme abnormalities and hearing impairment is warranted. Narcotic Analgesic: Dosage of methadone may need to be increased when coadministered with VIRACEPT. methadone ↓ methadone Oral Contraceptive: Alternative or additional contraceptive measures should be used when oral contraceptives and VIRACEPT are coadministered. ethinyl estradiol ↓ ethinyl estradiol PDE5 Inhibitors:
sildenafil
vardenafil
tadalafil
↑ PDE5 InhibitorsConcomitant use of PDE5 inhibitors and VIRACEPT should be undertaken with caution.
May result in an increase in PDE5 inhibitor-associated adverse events, including hypotension, syncope, visual disturbances, and priapism.
Use of PDE-5 inhibitors for pulmonary arterial hypertension (PAH):
- Use of sildenafil (REVATIO) is contraindicated when used for the treatment of pulmonary arterial hypertension (PAH) [see Contraindications].
- The following dose adjustments are recommended for use of tadalafil (ADCIRCA™) with VIRACEPT:
Coadministration of ADCIRCA in patients on VIRACEPT or coadministration of VIRACEPT in patients on ADCIRCA:
Start at or adjust ADCIRCA to 20 mg once daily. Increase to 40 mg once daily based upon individual tolerability.
Use of PDE5 inhibitors for erectile dysfunction:
Sildenafil at a single dose not exceeding 25 mg in 48 hours, vardenafil at a single dose not exceeding 2.5 mg in 24 hours, or tadalafil at a single dose not exceeding 10 mg dose in 72 hours, is recommended. Use with increased monitoring for adverse events.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies in mice and rats were conducted with nelfinavir at oral doses up to 1000 mg/kg/day. No evidence of a tumorigenic effect was noted in mice at systemic exposures (Cmax) up to 9-fold those measured in humans at the recommended therapeutic dose (750 mg TID or 1250 mg BID). In rats, thyroid follicular cell adenomas and carcinomas were increased in males at 300 mg/kg/day and higher and in females at 1000 mg/kg/day. Systemic exposures (Cmax) at 300 and 1000 mg/kg/day were 1- to 3-fold, respectively, those measured in humans at the recommended therapeutic dose. Repeated administration of nelfinavir to rats produced effects consistent with hepatic microsomal enzyme induction and increased thyroid hormone deposition; these effects predispose rats, but not humans, to thyroid follicular cell neoplasms. Nelfinavir showed no evidence of mutagenic or clastogenic activity in a battery of in vitro and in vivo genetic toxicology assays. These studies included bacterial mutation assays in S. typhimurium and E. coli, a mouse lymphoma tyrosine kinase assay, a chromosomal aberration assay in human lymphocytes, and an in vivo mouse bone marrow micronucleus assay.
Nelfinavir produced no effects on either male or female mating and fertility or embryo survival in rats at systemic exposures comparable to the human therapeutic exposure.
Pregnancy - Pregnancy Category B
There were no effects on fetal development or maternal toxicity when nelfinavir was administered to pregnant rats at systemic exposures (AUC) comparable to human exposure. Administration of nelfinavir to pregnant rabbits resulted in no fetal development effects up to a dose at which a slight decrease in maternal body weight was observed; however, even at the highest dose evaluated, systemic exposure in rabbits was significantly lower than human exposure. Additional studies in rats indicated that exposure to nelfinavir in females from mid-pregnancy through lactation had no effect on the survival, growth, and development of the offspring to weaning. Subsequent reproductive performance of these offspring was also not affected by maternal exposure to nelfinavir. However, there are no adequate and well-controlled studies in pregnant women taking VIRACEPT. Because animal reproduction studies are not always predictive of human response, VIRACEPT should be used during pregnancy only if clearly needed.
Nursing Mothers
The Centers for Disease Control and Prevention recommends that HIV-infected mothers not breast-feed their infants to avoid risking postnatal transmission of HIV. Studies in lactating rats have demonstrated that nelfinavir is excreted in milk. Because of both the potential for HIV transmission and the potential for serious adverse reactions in nursing infants, mothers should be instructed not to breast-feed if they are receiving VIRACEPT.
Pediatric Use
The safety and effectiveness of VIRACEPT have been established in patients from 2 to 13 years of age. The use of VIRACEPT in these age groups is supported by evidence from adequate and well-controlled studies of VIRACEPT in adults and pharmacokinetic studies and studies supporting activity in pediatric patients. In patients less than 2 years of age, VIRACEPT was found to be safe at the doses studied, but a reliably effective dose could not be established (see CLINICAL PHARMACOLOGY: Special Populations, ADVERSE REACTIONS: Pediatric Population, and DOSAGE AND ADMINISTRATION: Pediatric Patients).
The following issues should be considered when initiating VIRACEPT in pediatric patients:
- In pediatric patients ≥ 2 years of age receiving VIRACEPT as part of triple combination antiretroviral therapy in randomized studies, the proportion of patients achieving a HIV RNA level <400 copies/mL through 48 weeks ranged from 26% to 42%.
- Response rates in children <2 years of age appeared to be poorer than those in patients ≥ 2 years of age in some studies.
- Highly variable drug exposure remains a significant problem in the use of VIRACEPT in pediatric patients. Unpredictable drug exposure may be exacerbated in pediatric patients because of increased clearance compared to adults and difficulties with compliance and adequate food intake with dosing. Pharmacokinetic results from the pediatric studies are reported in Table 5 (see CLINICAL PHARMACOLOGY, Special Populations).
Study 556 was a randomized, double-blind, placebo-controlled trial with VIRACEPT or placebo coadministered with ZDV and ddI in 141 HIV-positive children who had received minimal antiretroviral therapy. The mean age of the children was 3.9 years. Ninety four (67%) children were between 2–12 years, and 47 (33%) were < 2 years of age. The mean baseline HIV RNA value was 5.0 log for all patients and the mean CD4 cell count was 886 cells/mm3 for all patients. The efficacy of VIRACEPT measured by HIV RNA <400 at 48 weeks in children ≥ 2 years of age was 26% compared to 2% of placebo patients (p=0.0008). In the children < 2 years of age, only 1 of 27 and 2 of 20 maintained an undetectable HIV RNA level at 48 weeks for placebo and VIRACEPT patients, respectively.
PACTG 377 was an open-label study that randomized 181 HIV treatment-experienced pediatric patients to receive: d4T+NVP+RTV, d4T+3TC+NFV, or d4T+3TC+NVP+NFV with NFV given on a TID schedule. The median age was 5.9 years and 46% were male. At baseline the median HIV RNA was 4.4 log and median CD4 cell count was 690 cells/mm3. Substudy PACTG 725 evaluated d4T+3TC+NFV with NFV given on a BID schedule. The proportion of patients with detectable viral load at baseline achieving HIV RNA <400 copies/mL at 48 weeks was: 41% for d4T+NVP+RTV, 42% for d4T+3TC+NFV, 30% for d4T+NVP+NFV, and 52% for d4T+3TC+NVP+NFV. No significant clinical differences were identified between patients receiving VIRACEPT in BID or TID schedules.
VIRACEPT has been evaluated in 2 studies of young infants. The PENTA 7 study was an open-label study to evaluate the toxicity, tolerability, pharmacokinetics, and activity of NFV+d4T+ddI in 20 HIV-infected infants less than 12 weeks of age. PACTG 353 evaluated the pharmacokinetics and safety of VIRACEPT in infants born to HIV-infected women receiving NFV as part of combination therapy during pregnancy.
-
ADVERSE REACTIONS
The safety of VIRACEPT was studied in over 5000 patients who received drug either alone or in combination with nucleoside analogues. The majority of adverse events were of mild intensity. The most frequently reported adverse event among patients receiving VIRACEPT was diarrhea, which was generally of mild to moderate intensity.
Drug-related clinical adverse experiences of moderate or severe intensity in ≥ 2% of patients treated with VIRACEPT coadministered with d4T and 3TC (Study 542) for up to 48 weeks or with ZDV plus 3TC (Study 511) for up to 24 weeks are presented in Table 12.
Table 12 Percentage of Patients with Treatment-Emergent* Adverse Events of Moderate or Severe Intensity Reported in ≥ 2% of Patients Study 511
24 weeksStudy 542
48 weeks
Adverse EventsPlacebo
+ ZDV/3TC
(n=101)500 mg TID VIRACEPT + ZDV/3TC
(n=97)750 mg TID VIRACEPT + ZDV/3TC
(n=100)1250 mg BID VIRACEPT + d4T/3TC
(n=344)750 mg TID VIRACEPT + d4T/3TC
(n=210)- *
- Includes those adverse events at least possibly related to study drug or of unknown relationship and excludes concurrent HIV conditions
Digestive System Diarrhea 3% 14% 20% 20% 15% Nausea 4% 3% 7% 3% 3% Flatulence 0 5% 2% 1% 1% Skin/Appendages Rash 1% 1% 3% 2% 1% Adverse events occurring in less than 2% of patients receiving VIRACEPT in all phase II/III clinical trials and considered at least possibly related or of unknown relationship to treatment and of at least moderate severity are listed below.
Body as a Whole: abdominal pain, accidental injury, allergic reaction, asthenia, back pain, fever, headache, malaise, pain, and redistribution/accumulation of body fat (see PRECAUTIONS, Fat Redistribution).
Digestive System: anorexia, dyspepsia, epigastric pain, gastrointestinal bleeding, hepatitis, mouth ulceration, pancreatitis, and vomiting.
Hemic/Lymphatic System: anemia, leukopenia, and thrombocytopenia.
Metabolic/Nutritional System: increases in alkaline phosphatase, amylase, creatine phosphokinase, lactic dehydrogenase, SGOT, SGPT, and gamma glutamyl transpeptidase; hyperlipemia, hyperuricemia, hyperglycemia, hypoglycemia, dehydration, and liver function tests abnormal.
Musculoskeletal System: arthralgia, arthritis, cramps, myalgia, myasthenia, and myopathy.
Nervous System: anxiety, depression, dizziness, emotional lability, hyperkinesia, insomnia, migraine, paresthesia, seizures, sleep disorder, somnolence, and suicide ideation.
Respiratory System: dyspnea, pharyngitis, rhinitis, and sinusitis.
Skin/Appendages: dermatitis, folliculitis, fungal dermatitis, maculopapular rash, pruritus, sweating, and urticaria.
Special Senses: acute iritis and eye disorder.
Urogenital System: kidney calculus, sexual dysfunction, and urine abnormality.
Post-Marketing Experience
The following additional adverse experiences have been reported from postmarketing surveillance as at least possibly related or of unknown relationship to VIRACEPT:
Body as a Whole: hypersensitivity reactions (including bronchospasm, moderate to severe rash, fever, and edema).
Cardiovascular System: QTc prolongation, torsades de pointes.
Digestive System: jaundice.
Metabolic/Nutritional System: bilirubinemia, metabolic acidosis.
Laboratory Abnormalities
The percentage of patients with marked laboratory abnormalities in Studies 542 and 511 are presented in Table 13. Marked laboratory abnormalities are defined as a Grade 3 or 4 abnormality in a patient with a normal baseline value, or a Grade 4 abnormality in a patient with a Grade 1 abnormality at baseline.
Table 13 Percentage of Patients by Treatment Group With Marked Laboratory Abnormalities * in > 2% of Patients Study 511 Study 542 Placebo
+ ZDV/3TC
(n=101)500 mg TID VIRACEPT + ZDV/3TC
(n=97)750 mg TID VIRACEPT + ZDV/3TC
(n=100)1250 mg BID VIRACEPT + d4T/3TC
(n=344)750 mg TID VIRACEPT + d4T/3TC
(n=210)- *
- Marked laboratory abnormalities are defined as a shift from Grade 0 at baseline to at least Grade 3 or from Grade 1 to Grade 4
Hematology Hemoglobin 6% 3% 2% 0 0 Neutrophils 4% 3% 5% 2% 1% Lymphocytes 1% 6% 1% 1% 0 Chemistry ALT (SGPT) 6% 1% 1% 2% 1% AST (SGOT) 4% 1% 0 2% 1% Creatine Kinase 7% 2% 2% NA NA Pediatric Population
VIRACEPT has been studied in approximately 400 pediatric patients in clinical trials from birth to 13 years of age. The adverse event profile seen during pediatric clinical trials was similar to that for adults.
The most commonly reported drug-related, treatment-emergent adverse events reported in the pediatric studies included: diarrhea, leukopenia/neutropenia, rash, anorexia, and abdominal pain. Diarrhea, regardless of assigned relationship to study drug, was reported in 39% to 47% of pediatric patients receiving VIRACEPT in 2 of the larger treatment trials. Leukopenia/neutropenia was the laboratory abnormality most commonly reported as a significant event across the pediatric studies.
-
OVERDOSAGE
Human experience of acute overdose with VIRACEPT is limited. There is no specific antidote for overdose with VIRACEPT. If indicated, elimination of unabsorbed drug should be achieved by emesis or gastric lavage. Administration of activated charcoal may also be used to aid removal of unabsorbed drug. Since nelfinavir is highly protein bound, dialysis is unlikely to significantly remove drug from blood.
-
DOSAGE AND ADMINISTRATION
Adults
The recommended dose is 1250 mg (five 250 mg tablets or two 625 mg tablets) twice daily or 750 mg (three 250 mg tablets) three times daily. VIRACEPT should be taken with a meal. Patients unable to swallow the 250 or 625 mg tablets may dissolve the tablets in a small amount of water. Once dissolved, patients should mix the cloudy liquid well, and consume it immediately. The glass should be rinsed with water and the rinse swallowed to ensure the entire dose is consumed.
Pediatric Patients (2–13 years)
In children 2 years of age and older, the recommended oral dose of VIRACEPT Oral Powder or 250 mg tablets is 45 to 55 mg/kg twice daily or 25 to 35 mg/kg three times daily. All doses should be taken with a meal. Doses higher than the adult maximum dose of 2500 mg per day have not been studied in children. For children unable to take tablets, VIRACEPT Oral Powder may be administered. The oral powder may be mixed with a small amount of water, milk, formula, soy formula, soy milk, or dietary supplements; once mixed, the entire contents must be consumed in order to obtain the full dose. If the mixture is not consumed immediately, it must be stored under refrigeration, but storage must not exceed 6 hours. Acidic food or juice (e.g., orange juice, apple juice, or apple sauce) are not recommended to be used in combination with VIRACEPT, because the combination may result in a bitter taste. VIRACEPT Oral Powder should not be reconstituted with water in its original container.
The healthcare provider should assess appropriate formulation and dosage for each patient. Crushed 250 mg tablets can be used in lieu of powder. Tables 14 and 15 provide dosing guidelines for VIRACEPT tablets and powder based on age and body weight.
Table 14 Dosing Table for Children ≥ 2 years of age (tablets) Body weight Twice daily (BID)
45 – 55 mg/kg
≥2 yearsThree times daily (TID)
25 – 35 mg/kg
≥2 yearsKg. Lbs. # of tablets
(250 mg)# of tablets
(250 mg)10 – 12 22 –26.4 2 1 13 – 18 28.6 – 39.6 3 2 19 – 20 41.8 – 44 4 2 ≥21 ≥46.2 4 – 5* 3† Table 15 Dosing Table for Children ≥ 2 years of age (powder) Body weight Twice daily (BID)
45 – 55 mg/kgThree times daily (TID)
25 – 35 mg/kgKg. Lbs. Scoops of powder
(50 mg/1 g)Teaspoons* of powder Scoops of powder
(50 mg/1 g)Teaspoons* of powder 9.0 to < 10.5 20 to < 23 10 2½ 6 1½ 10.5 to < 12 23 to < 26.5 11 2¾ 7 1¾ 12 to < 14 26.5 to < 31 13 3¼ 8 2 14 to < 16 31 to < 35 15 3¾ 9 2¼ 16 to < 18 35 to < 39.5 Not recommended† Not recommended† 10 2½ 18 to < 23 39.5 to < 50.5 Not recommended† Not recommended† 12 3 ≥23 ≥50.5 Not recommended† Not recommended† 15 3¾ Hepatic Impairment
Viracept can be used in patients with mild hepatic impairment without any dose adjustment. VIRACEPT should not be used in patients with either moderate or severe hepatic impairment (see CLINICAL PHARMACOLOGY: Special Populations).
-
HOW SUPPLIED
VIRACEPT (nelfinavir mesylate) 250 mg: Light blue, capsule-shaped tablets with a clear film coating engraved with "VIRACEPT" on one side and "250 mg" on the other.
Bottles of 300, 250 mg tablets………………………………...NDC 63010-010-30
VIRACEPT (nelfinavir mesylate) 625 mg: White oval tablet with a clear film coating engraved with "V" on one side and "625" on the other.
Bottles of 120, 625 mg tablets………………………………...NDC 63010-027-70
VIRACEPT (nelfinavir mesylate) Oral Powder is available as a 50 mg/g off-white powder containing 50 mg (as nelfinavir free base) in each level scoopful (1 gram).
Multiple use bottles of 144 grams of powder with scoop …….NDC 63010-011-90
- SPL UNCLASSIFIED SECTION
-
VIRACEPT® (nelfinavir mesylate) TABLETS and ORAL POWDER
ALERT: Find out about medicines that should NOT be taken with VIRACEPT. Please also read the section "MEDICINES YOU SHOULD NOT TAKE WITH VIRACEPT".
Patient Information
VIRACEPT® (VI-ra-cept)
Generic Name: nelfinavir (nel-FIN-na-veer) mesylate
Please read this information carefully before taking VIRACEPT. Also, please read this leaflet each time you renew the prescription, in case anything has changed. This is a summary and not a replacement for a careful discussion with your healthcare provider. You and your healthcare provider should discuss VIRACEPT when you start taking this medication and at regular checkups. You should remain under a healthcare provider's care when taking VIRACEPT and should not change or stop treatment without first talking with your healthcare provider.
What is VIRACEPT and how does it work?
VIRACEPT is a type of medicine called an HIV (human immunodeficiency virus) protease (PRO-tee-ase) inhibitor. VIRACEPT is always used in combination with other antiretroviral drugs in the treatment of people with HIV infection. VIRACEPT is for adults and for children 2 years of age and older.
Infection with HIV leads to the destruction of CD4 (T) cells, which are important to the immune system. After a large number of CD4 (T) cells have been destroyed, the infected person develops acquired immune deficiency syndrome (AIDS).
VIRACEPT works by blocking HIV protease (a protein-cutting enzyme), which is required for HIV to multiply. VIRACEPT has been shown to significantly reduce the amount of HIV in the blood. Although VIRACEPT is not a cure for HIV or AIDS, VIRACEPT can help reduce your risk for death and illness associated with HIV. Patients who took VIRACEPT also had significant increases in the number of CD4 (T) cells.
Does VIRACEPT cure HIV or AIDS?
VIRACEPT is not a cure for HIV infection or AIDS. People taking VIRACEPT may still develop opportunistic infections or other conditions associated with HIV infection. Some of these conditions are pneumonia, herpes virus infections, Mycobacterium avium complex (MAC) infections, and Kaposi's sarcoma.
Does VIRACEPT reduce the risk of passing HIV to others?
VIRACEPT does not reduce the risk of transmitting HIV to others through sexual contact or blood contamination. Continue to practice safe sex and do not use or share dirty needles.
How should I take VIRACEPT?
- You should stay under a healthcare provider's care when taking VIRACEPT. Do not change your treatment or stop treatment without first talking with your healthcare provider.
- You must take VIRACEPT every day exactly as your healthcare provider prescribed it. Follow the directions from your healthcare provider, exactly as written on the label.
- Dosing in adults (including children 14 years of age and older): The recommended adult dose of VIRACEPT is 1250 mg (five 250 mg tablets or two 625 mg tablets) taken two times a day or 750 mg (three 250 mg tablets) taken three times a day. Each dose should always be taken with a meal to help achieve higher VIRACEPT levels. VIRACEPT Tablets are film-coated to help make the tablets easier to swallow.
- Dosing in children 2 years of age and older: The VIRACEPT dose in children depends on their weight. The recommended oral dose of VIRACEPT oral powder or 250 mg tablets is 45 to 55 mg/kg twice daily or 25 to 35 mg/kg three times daily. All doses should be taken with a meal. Children who cannot swallow tablets may take VIRACEPT Oral Powder or crushed tablets.
- If you or your child is unable to swallow the tablets, dissolve the tablets in a small amount of water. Once dissolved, mix the cloudy liquid well, and consume immediately. The glass should be rinsed with water and the rinse swallowed to ensure the entire dose is consumed.
- Do not change your dose or stop taking VIRACEPT without first consulting with your healthcare provider.
- When your VIRACEPT supply starts to run low, get in touch with your healthcare provider or pharmacy. This is very important because the amount of virus in your blood may increase if the medicine is stopped for even a short time. The virus may develop resistance to antiretroviral medications, including VIRACEPT, but there are other antiretroviral treatment options. Talk to your healthcare provider about how to optimize your long-term treatment.
- Be sure to set up a schedule and follow it carefully.
- Only take medicine that has been prescribed specifically for you. Do not give VIRACEPT to others or take medicine prescribed for someone else.
How should VIRACEPT Oral Powder be prepared?
- The oral powder may be mixed with a small amount of water, milk, formula, soy formula, soy milk, dietary supplements, or dairy foods such as pudding or ice cream. Once mixed, the entire amount must be taken to obtain the full dose. If the mixture is not consumed immediately, it must be stored under refrigeration, but storage must not exceed 6 hours. Do not heat the mixed dose once it has been prepared.
- Do not mix the powder with any acidic food or juice, such as orange or grapefruit juice, apple juice, or apple sauce, because this may create a bitter taste.
- Do not add water to bottles of oral powder.
- VIRACEPT powder is supplied with a scoop for measuring. For help in determining the exact dose of powder for your child, please ask your doctor, nurse, pharmacist, or other healthcare provider.
What should I do if I miss a dose?
If you forget to take a dose of VIRACEPT, take it as soon as possible. However, if you skip the dose entirely, do not double the next dose. If you forget a lot of doses, talk to your healthcare provider about how you should continue taking your medicine.
What happens if I take too much VIRACEPT?
If you suspect that you took more than the prescribed dose of this medicine, contact your local poison control center or emergency room immediately.
Who should not take VIRACEPT?
Together with your healthcare provider, you need to decide whether VIRACEPT is appropriate for you.
- Do not take VIRACEPT if you are taking certain medicines. These could cause serious side effects that could cause death. Before you take VIRACEPT, you must tell your healthcare provider about all medicines you are taking or are planning to take. These include prescription and non-prescription medicines and herbal supplements.
For more information about medicines you should not take with VIRACEPT, please read the section titled "MEDICINES YOU SHOULD NOT TAKE WITH VIRACEPT."
- Do not take VIRACEPT if you have an allergy to VIRACEPT. Also tell your healthcare provider if you have any known allergies to other medicines, foods, preservatives, or dyes.
- Tell your healthcare provider if you are pregnant or plan to become pregnant. The effects of VIRACEPT on pregnant women or their unborn babies are not known.
- If you are breast-feeding, it is very important that you speak with your healthcare provider about the best way to feed your baby. If your baby does not already have HIV, there is a chance that it can be transmitted through breast-feeding. The Centers for Disease Control and Prevention recommends that women with HIV do not breast-feed.
- Talk with your healthcare provider if you have liver or kidney disease. VIRACEPT has not been extensively studied in people with liver or kidney disease. VIRACEPT can be used in patients with mild liver disease without any dose adjustment. VIRACEPT should not be used in patients with either moderate or severe liver disease.
- Certain medical problems may affect the use of VIRACEPT. Be sure to tell your healthcare provider of any other medical problems you may have.
Can VIRACEPT be taken with other medications?
VIRACEPT may interact with other drugs, including those you take without a prescription. You must tell your healthcare provider about all medicines you are taking or planning to take before you take VIRACEPT. It is a good idea to keep a complete list of all the medicines that you take, including non-prescription medicines, herbal remedies and supplements and street drugs. Update this list when medicines are added or stopped. Give copies of this list to all of your healthcare providers every time you visit or fill a prescription.
MEDICINES YOU SHOULD NOT TAKE WITH VIRACEPT:
Do not take the following drugs because they can cause serious problems or death if taken with VIRACEPT:
- Cordarone® (amiodarone) (for irregular heartbeat)
- Orap® (pimozide) (for seizures)
- Quinidine (for irregular heartbeat), also known as Quinaglute®, Cardioquin®, Quinidex®, and others
- D.H.E. 45® Injection, Ergomar, Migranal®, Wigraine® and Cafergot® (for migraine headaches) and Methergine® (for bleeding after childbirth)
- Halcion® (triazolam) (for sleep problem)
- Versed® (midazolam) (sedative hypnotic)
- Revatio® (sildenafil) (for treatment of pulmonary arterial hypertension)
- Alfuzosin (for treatment of benign prostate enlargement)
Do not take the following medicines when you take VIRACEPT. They may reduce the levels of VIRACEPT in the blood and make it less effective. Talk with your healthcare provider if you are currently taking these medicines because other medicines may have to be given to take their place:
- Prilosec® (omeprazole) (for stomach acid reduction)
- Rifampin (also known as Rimactane®, Rifadin®, Rifater®, or Rifamate®) (for tuberculosis)
- Phenobarbital (for seizures)
- Tegretol® (carbamazepine) (for seizures)
Do not take VIRACEPT with St. John's wort (hypericum perforatum), an herbal product sold as a dietary supplement, or products containing St. John's wort. Talk with your healthcare provider if you are taking or planning to take St. John's wort. Taking St. John's wort may decrease VIRACEPT levels and lead to increased viral load and possible resistance to VIRACEPT.
Do not take VIRACEPT with cholesterol-lowering medicines Mevacor® (lovastatin) or Zocor® (simvastatin) because of possible serious reactions.
Do not take VIRACEPT with Serevent® (salmeterol) because of possible serious reactions.
Talk to your healthcare provider before you start taking any new prescription or non-prescription medicines or herbal supplements with VIRACEPT.
Medicines that require dose adjustments:
It is possible that your healthcare provider may need to increase or decrease the dose of other medicines when you are also taking VIRACEPT.
There is also an increased risk of drug interactions between VIRACEPT and Lipitor® (atorvastatin), Crestor® (rosuvastatin), Pravachol® (pravastatin) and Lescol® (fluvastatin); talk to your healthcare provider before you take any of these cholesterol-reducing medicines with VIRACEPT.
Before you take PDE5 Inhibitors, such as Viagra® (sildenafil), Levitra® (vardenafil) or Cialis® (tadalafil) with VIRACEPT, talk to your healthcare provider about possible drug interactions and side effects. If you take these types of drugs and VIRACEPT together, you may be at increased risk of side effects of these drugs such as low blood pressure, visual changes, and penile erection lasting more than 4 hours. If an erection lasts longer than 4 hours, you should seek immediate medical assistance to avoid permanent damage to your penis. Your healthcare provider can explain these symptoms to you.
Before you take Adcirca™ (tadalafil) with VIRACEPT, talk to your healthcare provider since VIRACEPT may increase the amount of tadalafil in your blood.
Before you take Tracleer® (bosentan) with VIRACEPT, talk to your healthcare provider since VIRACEPT may increase the amount of bosentan in your blood.
Before you take colchicine with VIRACEPT, talk to your healthcare provider since VIRACEPT may increase the amount of colchicine in your blood.
Before you take warfarin (Coumadin®) with VIRACEPT, talk to your healthcare provider since VIRACEPT may affect the amount of warfarin in your blood.
-
If you are taking both didanosine (Videx®) and VIRACEPT:
You should take VIRACEPT with food one hour after or more than two hours before you take Videx buffered tablets. - If you are taking oral contraceptives ("the pill") to prevent pregnancy, you should use an additional or different type of contraception since VIRACEPT may reduce the effectiveness of oral contraceptives.
- Non-nucleoside reverse transcriptase inhibitors (NNRTIs): RESCRIPTOR® (delavirdine) may increase the amount of VIRACEPT in your blood and VIRACEPT may lower the amount of RESCRIPTOR in your blood.
- Protease Inhibitors (PIs): VIRACEPT may increase the amount of Crixivan® (indinavir), Norvir® (ritonavir), and Invirase® or Fortovase® (saquinavir) in your blood. As a result, your healthcare provider may choose to lower the dose of VIRACEPT or one of these other medicines or monitor certain lab tests if VIRACEPT is taken in combination with one or more of these other medicines.
- If you are taking Mycobutin® (rifabutin), your healthcare provider may lower the dose of Mycobutin.
- If you are taking Dilantin® (phenytoin), your healthcare provider will need to monitor the levels of phenytoin in your blood and may need to adjust the dose of phenytoin.
-
Other Special considerations
VIRACEPT Oral Powder contains aspartame, a low-calorie sweetener, and therefore should not be taken by children with phenylketonuria (PKU).
What are the possible side effects of VIRACEPT?
- This list of side effects is not complete. If you have questions about side effects, ask your healthcare provider, nurse, or pharmacist. You should report any new or continuing symptoms to your healthcare provider right away. Your healthcare provider may be able to help you manage these side effects. Most of the side effects experienced with VIRACEPT have been mild to moderate.
- Diarrhea is the most common side effect in people taking VIRACEPT, and most adult patients had at least mild diarrhea at some point during treatment. In clinical studies, about 15–20% of patients receiving VIRACEPT 750 mg (three 250 mg tablets) three times daily or 1250 mg (five 250 mg tablets or two 625 mg tablets) two times daily had four or more loose stools a day. In most cases, VIRACEPT-associated diarrhea can be controlled using antidiarrheal medicines, such as Imodium A-D® (loperamide).
- Other side effects that occurred in 3–7% of patients receiving VIRACEPT include nausea, gas, and rash.
- The side effects observed in children and adults receiving VIRACEPT are similar. Diarrhea was also the most common side effect seen in children. Some children experienced low white blood cells (leukopenia/neutropenia), which resolved without treatment interruption in most cases.
- Diabetes and high blood sugar (hyperglycemia) occur in patients taking protease inhibitors such as VIRACEPT. Some patients had diabetes before starting protease inhibitors, others did not. Some patients needed changes in their diabetes medicine. Others needed new diabetes medicine after starting their VIRACEPT medicine.
- Changes in body fat have been seen in some patients taking antiretroviral therapy. These changes may include increased amount of fat in the upper back and neck ("buffalo hump"), breast and around the trunk. Loss of fat from the legs, arms and face may also happen. The cause and long-term health effects of these conditions are not known at this time.
- Some patients with hemophilia have increased bleeding with protease inhibitors.
- There were other side effects, some of them serious, noted in clinical studies that occurred in less than 2% of patients receiving VIRACEPT. However, these side effects may have been due to other drugs that patients were taking or to the illness itself. Except for diarrhea, there were not many differences in side effects in patients who took VIRACEPT along with other drugs compared with those who took only the other drugs.
- Before you start using any medicine, talk with your healthcare provider about what to expect and discuss ways to reduce the side effects you may have.
How should VIRACEPT be stored?
- Keep VIRACEPT and all other medicines out of the reach of children.
- Keep bottle closed and store at room temperature (between 59°F and 86°F) away from sources of moisture such as a sink or other damp place. Heat and moisture may reduce the effectiveness of VIRACEPT.
- Do not keep medicine that is out of date or that you no longer need. Be sure that if you throw any medicine away, it is out of the reach of children.
- Store in original container.
General advice about prescription medicines
Discuss all questions about your health with your healthcare provider. If you have questions about VIRACEPT or any other medication you are taking, ask your doctor, nurse, pharmacist, or other healthcare provider. You can also call 800-438-1985 toll free.
VIRACEPT and Agouron are registered trademarks of Agouron Pharmaceuticals, Inc.
Trademarks are the property of their respective owners.
AGOURON PHARMACEUTICALS, INC.
La Jolla, CA 92037, USALAB-0346-8.0
February 2011
-
PRINCIPAL DISPLAY PANEL - 250 mg Bottle Label
NDC 63010-010-30
300 TabletsVIRACEPT®
(nelfinavir mesylate)
TabletsEach tablet contains nelfinavir mesylate
equivalent to 250 mg of nelfinavir (free base).ALERT: Find out about
medicines that should NOT
be taken with VIRACEPT.Note to Pharmacist: Do not
cover ALERT box with
pharmacy label.Rx only
250 mg
Agouron®
Pharmaceuticals, Inc.
A Pfizer Company300 Tablets
Repacked by:
H.J. Harkins Company, Inc.
Grover Beach, CA 93433
-
INGREDIENTS AND APPEARANCE
VIRACEPT
nelfinavir mesylate tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:52959-289(NDC:63010-010) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength nelfinavir mesylate (UNII: 98D603VP8V) (nelfinavir - UNII:HO3OGH5D7I) nelfinavir 250 mg Inactive Ingredients Ingredient Name Strength calcium silicate (UNII: S4255P4G5M) crospovidone (UNII: 68401960MK) magnesium stearate (UNII: 70097M6I30) hypromelloses (UNII: 3NXW29V3WO) triacetin (UNII: XHX3C3X673) FD&C blue No. 2 (UNII: L06K8R7DQK) Product Characteristics Color BLUE (light blue) Score no score Shape OVAL (capsule-shaped) Size 19mm Flavor Imprint Code VIRACEPT;250;mg Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:52959-289-06 6 in 1 BOTTLE 2 NDC:52959-289-30 30 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA020779 03/14/1997 Labeler - H.J. Harkins Company, Inc. (147681894) Registrant - Pfizer Inc (113480771) Establishment Name Address ID/FEI Business Operations Pfizer Manufacturing Deutschland GmbH 341970073 MANUFACTURE