Label: STERILE WATER injection
- NDC Code(s): 13985-808-25, 13985-808-50, 13985-808-60
- Packager: MWI (VetOne)
- Category: PRESCRIPTION ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated June 10, 2014
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
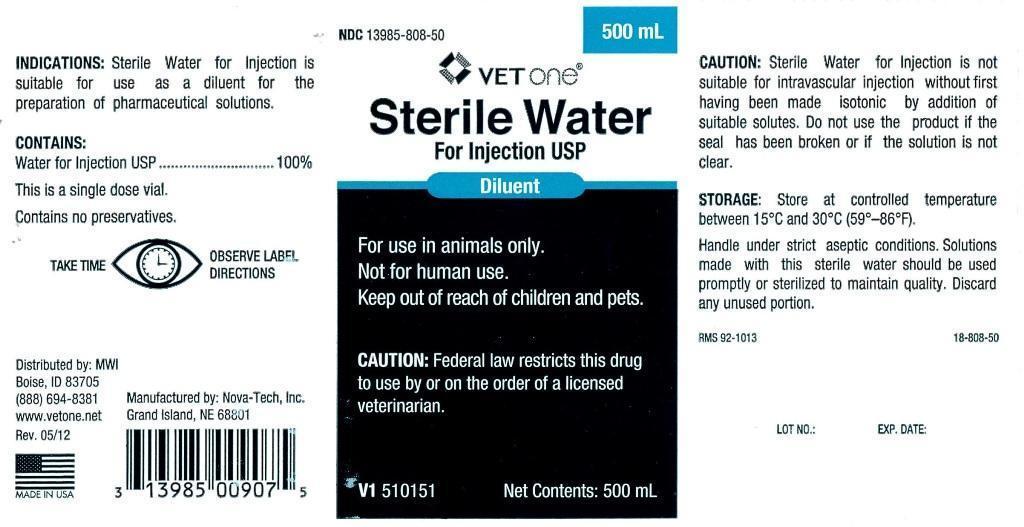
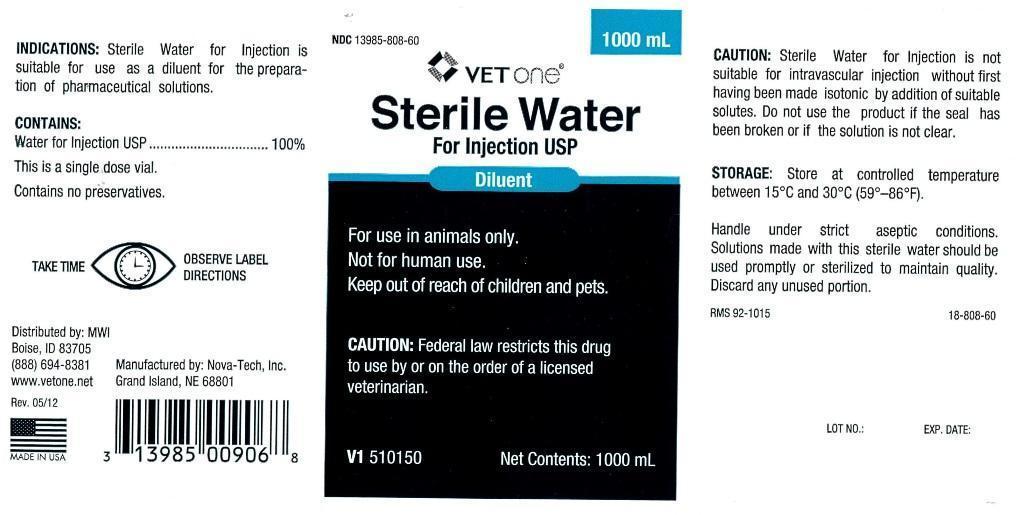
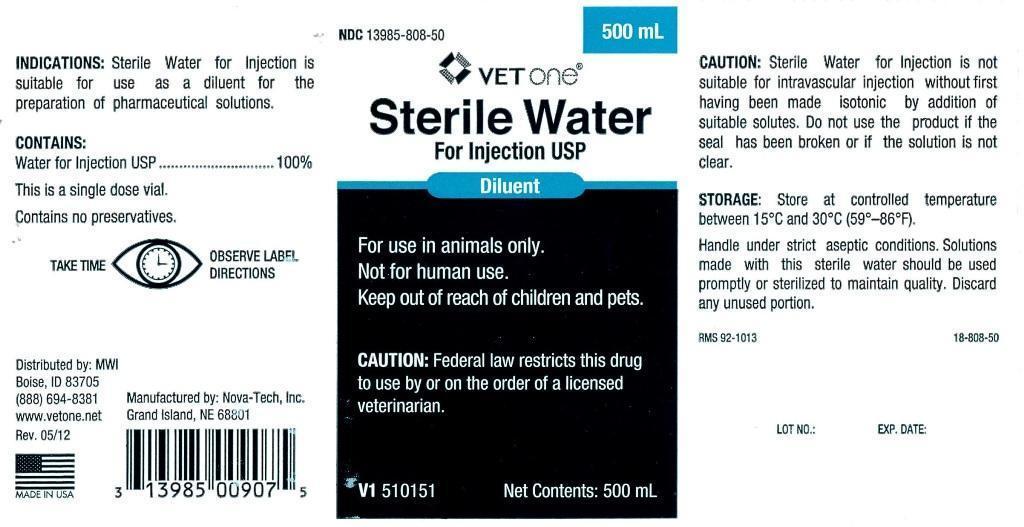
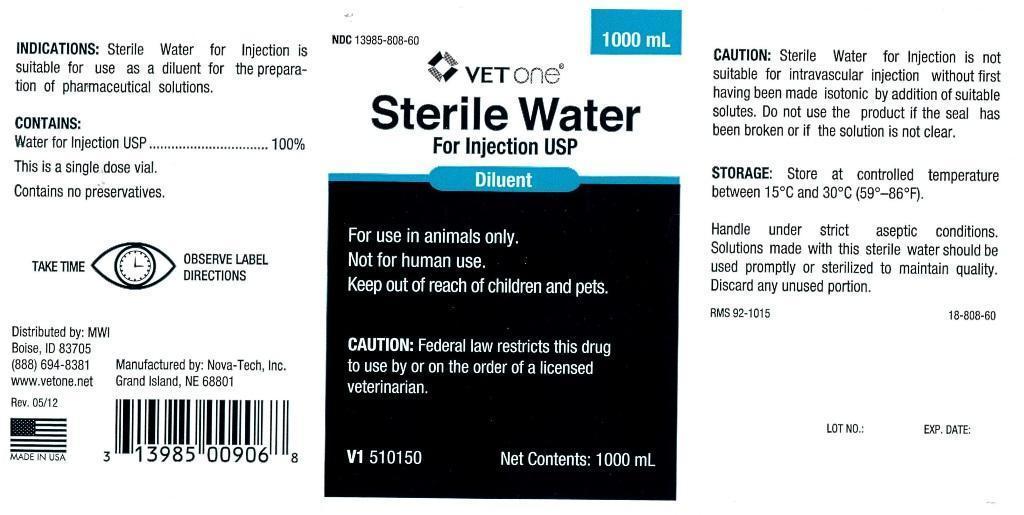
- INDICATIONS:
- CONTAINS:
- WARNINGS AND PRECAUTIONS
-
INFORMATION FOR OWNERS/CAREGIVERS
Distributed by: MWI
Boise, ID 83705
(888) 694-8381
www.vetone.netRev. 05/12
Manufactured by: Nova-Tech, Inc.
Grand Island, NE 68801V1 501046
Net Contents: 250 mL
RMS 92-1011 18-808-25
LOT NO.: EXP. DATE:
- VETERINARY INDICATIONS
- KEEP OUT OF REACH OF CHILDREN
- CAUTION:
- CAUTION:
- STORAGE:
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
STERILE WATER
sterile water injectionProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:13985-808 Route of Administration PARENTERAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength WATER (UNII: 059QF0KO0R) (WATER - UNII:059QF0KO0R) WATER 250 mL in 250 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13985-808-25 250 mL in 1 BOTTLE, PLASTIC 2 NDC:13985-808-50 500 mL in 1 BOTTLE, PLASTIC 3 NDC:13985-808-60 1000 mL in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 07/30/2012 Labeler - MWI (VetOne) (019926120) Registrant - MWI (VetOne) (019926120) Establishment Name Address ID/FEI Business Operations Nova-Tech, Inc. 196078976 manufacture, api manufacture