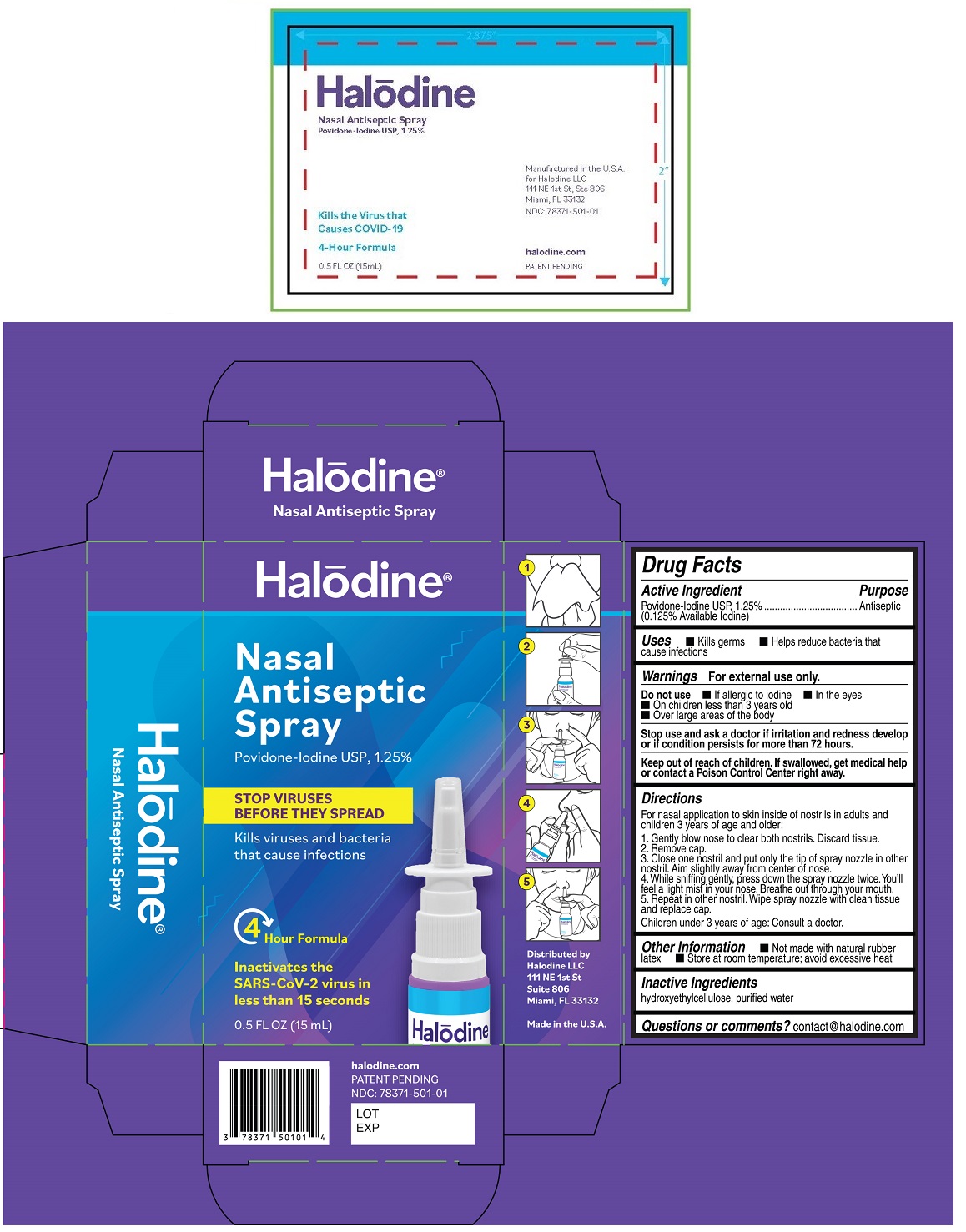
Label: HALODINE NASAL ANTISEPTIC- povidone-iodine spray
-
Contains inactivated NDC Code(s)
NDC Code(s): 78371-501-01 - Packager: Halodine Llc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 4, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredient
- Purpose
- Uses
- Warnings
-
Directions
For nasal application to skin inside of nostrils in adults and children 3 years of age and older:
1. Gently blow nose to clear both nostrils. Discard tissue.
2. Remove cap.
3. Close one nostril and put only the tip of spray nozzle in other nostril. Aim slightly away from center of nose.
4. While sniffing gently, press down the spray nozzle twice. You'll feel a light mist in your nose. Breathe out through your mouth.
5. Repeat in other nostril. Wipe spray nozzle with clean tissue and replace cap.
Children under 3 years of age: Consult a doctor.
- Other Information
- Inactive Ingredients
- Questions or comments?
- SPL UNCLASSIFIED SECTION
- Packaging
-
INGREDIENTS AND APPEARANCE
HALODINE NASAL ANTISEPTIC
povidone-iodine sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:78371-501 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POVIDONE-IODINE (UNII: 85H0HZU99M) (IODINE - UNII:9679TC07X4) IODINE 1.25 g in 100 mL Inactive Ingredients Ingredient Name Strength HYDROXYETHYL CELLULOSE, UNSPECIFIED (UNII: T4V6TWG28D) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:78371-501-01 1 in 1 BOX 02/01/2021 1 15 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 02/01/2021 Labeler - Halodine Llc (117526113)