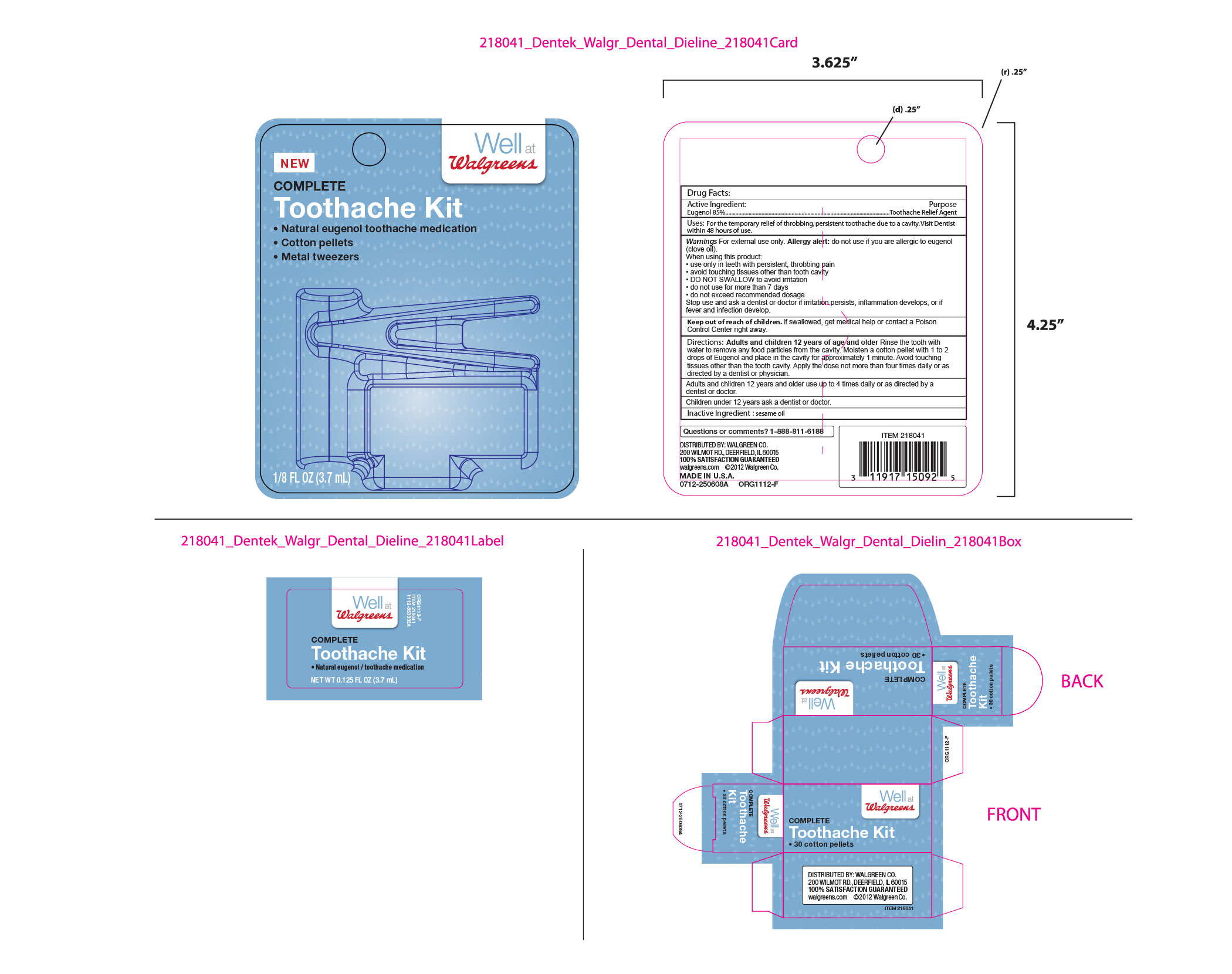
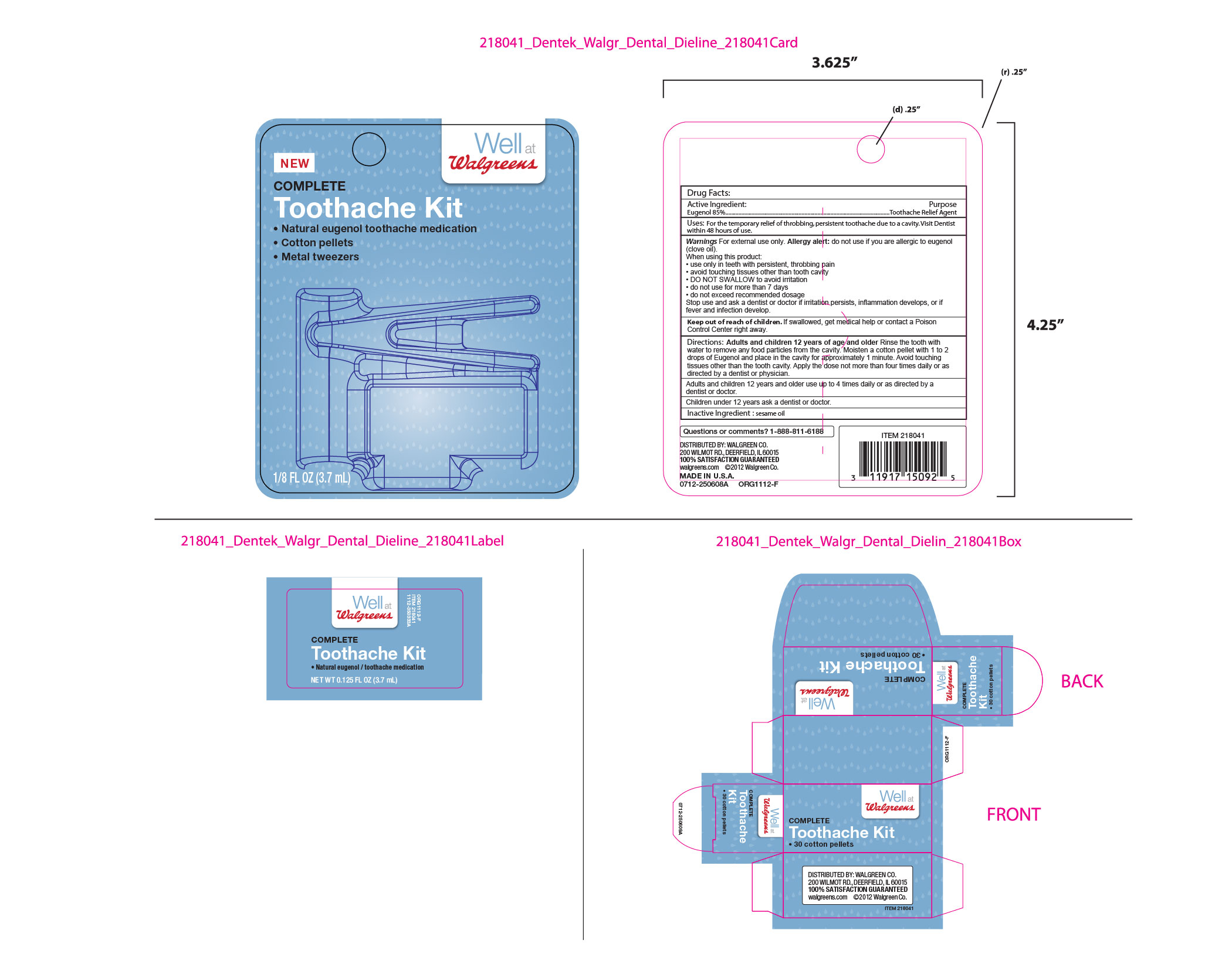
Label: WALGREENS NATURAL EUGENOL/TOOTHACHE MEDICATION WITH COTTON PELLETS AND TWEEZERS- eugenol liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 60630-608-01, 60630-608-02 - Packager: DenTek Oral Care, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 2, 2015
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Outer package card labeling
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings For external use only. Allergy alert: do not use if you are allergic to eugenol (clove oil).
When using this product:
. use only in teeth with pesistent, throbbing pain
. avoid touching tissues other than tooth cavity
. DO NOT SWALLOW to avoid irritation
. do not use for more than 7 days
. do not exceed recommended dosage
Stop use and ask a dentist or doctor if irritation persists, inflammation develops, or if fever and infection develop.
- KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
Directions:Adults and children 12 years of age and older Rinse the tooth with water to remove any food particles from the cavity.
Moisten a cotton pellet with 1 to 2 drops of Eugenol and place in the cavity for approximately 1 minute. Avoid touching tissues other than the tooth cavity. Apply the dose not more than four times daily or as directed by a dentist or physician.
Children under 12 years ask a dentist or doctor.
- INACTIVE INGREDIENT
- QUESTIONS
-
INGREDIENTS AND APPEARANCE
WALGREENS NATURAL EUGENOL/TOOTHACHE MEDICATION WITH COTTON PELLETS AND TWEEZERS
eugenol liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:60630-608 Route of Administration DENTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength EUGENOL (UNII: 3T8H1794QW) (EUGENOL - UNII:3T8H1794QW) EUGENOL 850 mg in 1 mL Inactive Ingredients Ingredient Name Strength SESAME OIL (UNII: QX10HYY4QV) 150 mg in 1 mL Product Characteristics Color yellow (pale yellow liquid, with odor of cloves) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:60630-608-02 1 in 1 PACKAGE 07/01/2015 1 NDC:60630-608-01 3.7 mL in 1 VIAL, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part356 07/01/2015 Labeler - DenTek Oral Care, Inc. (153818646) Registrant - DenTek Oral Care, Inc. (153818646)