Label: HYDROCHLOROTHIAZIDE capsule, gelatin coated
- NDC Code(s): 67544-739-60
- Packager: Aphena Pharma Solutions - Tennessee, LLC
- This is a repackaged label.
- Source NDC Code(s): 0591-0347
- Category: HUMAN PRESCRIPTION DRUG LABEL
Drug Label Information
Updated December 21, 2017
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
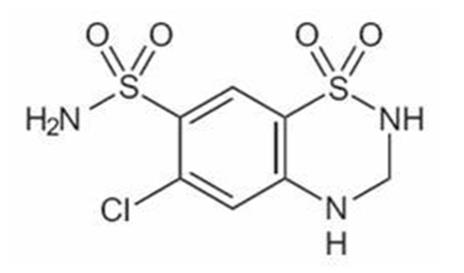
Hydrochlorothiazide Capsules, USP, 12.5 mg is the 3,4-dihydro derivative of chlorothiazide. Its chemical name is 6-chloro-3,4-dihydro-2H-1,2,4-benzothiadiazine-7-sulfonamide 1,1-dioxide. Its empirical formula is C7H8ClN3O4S2; its molecular weight is 297.74; and its structural formula is:
It is a white, or practically white, crystalline powder which is slightly soluble in water, but freely soluble in sodium hydroxide solution.
Hydrochlorothiazide is supplied as 12.5 mg capsules for oral use.
Inactive ingredients: colloidal silicon dioxide, corn starch, lactose monohydrate, magnesium stearate. Gelatin capsules contain D&C Red No. 28, D&C Yellow No. 10, FD&C Blue No. 1, gelatin, titanium dioxide. The capsules are printed with edible ink containing black iron oxide, D&C Yellow No. 10, FD&C Blue No. 1, FD&C Blue No. 2, FD&C Red No. 40.
-
CLINICAL PHARMACOLOGY
Hydrochlorothiazide blocks the reabsorption of sodium and chloride ions, and it thereby increases the quantity of sodium traversing the distal tubule and the volume of water excreted. A portion of the additional sodium presented to the distal tubule is exchanged there for potassium and hydrogen ions. With continued use of hydrochlorothiazide and depletion of sodium, compensatory mechanisms tend to increase this exchange and may produce excessive loss of potassium, hydrogen and chloride ions. Hydrochlorothiazide also decreases the excretion of calcium and uric acid, may increase the excretion of iodide and may reduce glomerular filtration rate. Metabolic toxicities associated with excessive electrolyte changes caused by hydrochlorothiazide have been shown to be dose-related.
Pharmacokinetics and Metabolism:
Hydrochlorothiazide is well absorbed (65% to 75%) following oral administration. Absorption of hydrochlorothiazide is reduced in patients with congestive heart failure.
Peak plasma concentrations are observed within 1 to 5 hours of dosing, and range from 70 to 490 ng/mL following oral doses of 12.5 to 100 mg. Plasma concentrations are linearly related to the administered dose. Concentrations of hydrochlorothiazide are 1.6 to 1.8 times higher in whole blood than in plasma. Binding to serum proteins has been reported to be approximately 40% to 68%. The plasma elimination half-life has been reported to be 6 to 15 hours. Hydrochlorothiazide is eliminated primarily by renal pathways. Following oral doses of 12.5 to 100 mg, 55% to 77% of the administered dose appears in urine and greater than 95% of the absorbed dose is excreted in urine as unchanged drug. In patients with renal disease, plasma concentrations of hydrochlorothiazide are increased and the elimination half-life is prolonged.
When hydrochlorothiazide is administered with food, its bioavailability is reduced by 10%, the maximum plasma concentration is reduced by 20%, and the time to maximum concentration increases from 1.6 to 2.9 hours.
Pharmacodynamics:
Acute antihypertensive effects of thiazides are thought to result from a reduction in blood volume and cardiac output, secondary to a natriuretic effect, although a direct vasodilatory mechanism has also been proposed. With chronic administration, plasma volume returns toward normal, but peripheral vascular resistance is decreased. The exact mechanism of the antihypertensive effect of hydrochlorothiazide is not known.
Thiazides do not affect normal blood pressure. Onset of action occurs within 2 hours of dosing, peak effect is observed at about 4 hours, and activity persists for up to 24 hours.
Clinical Studies:
In an 87 patient 4-week double-blind, placebo controlled, parallel group trial, patients who received hydrochlorothiazide had reductions in seated systolic and diastolic blood pressure that were significantly greater than those seen in patients who received placebo. In published placebo-controlled trials comparing 12.5 mg of hydrochlorothiazide to 25 mg, the 12.5 mg dose preserved most of the placebo-corrected blood pressure reduction seen with 25 mg.
-
INDICATIONS AND USAGE
Hydrochlorothiazide is indicated in the management of hypertension either as the sole therapeutic agent, or in combination with other antihypertensives. Unlike potassium sparing combination diuretic products, hydrochlorothiazide may be used in those patients in whom the development of hyperkalemia cannot be risked, including patients taking ACE inhibitors.
Usage in Pregnancy: The routine use of diuretics in an otherwise healthy woman is inappropriate and exposes mother and fetus to unnecessary hazard. Diuretics do not prevent development of toxemia of pregnancy, and there is no satisfactory evidence that they are useful in the treatment of developed toxemia.
Edema during pregnancy may arise from pathological causes or from the physiologic and mechanical consequences of pregnancy. Diuretics are indicated in pregnancy when edema is due to pathologic causes, just as they are in the absence of pregnancy. Dependent edema in pregnancy resulting from restriction of venous return by the expanded uterus is properly treated through elevation of the lower extremities and use of support hose; use of diuretics to lower intravascular volume in this case is illogical and unnecessary. There is hypervolemia during normal pregnancy which is harmful to neither the fetus nor the mother (in the absence of cardiovascular disease), but which is associated with edema, including generalized edema in the majority of pregnant women. If this edema produces discomfort, increased recumbency will often provide relief. In rare instances this edema may cause extreme discomfort which is not relieved by rest. In these cases a short course of diuretics may provide relief and may be appropriate.
- CONTRAINDICATIONS
-
WARNINGS
Acute Myopia and Secondary Angle-Closure Glaucoma: Hydrochlorothiazide, a sulfonamide, can cause an idiosyncratic reaction, resulting in acute transient myopia and acute angle-closure glaucoma. Symptoms include acute onset of decreased visual acuity or ocular pain and typically occur within hours to weeks of drug initiation. Untreated acute angle-closure glaucoma can lead to permanent vision loss. The primary treatment is to discontinue hydrochlorothiazide as rapidly as possible. Prompt medical or surgical treatments may need to be considered if the intraocular pressure remains uncontrolled. Risk factors for developing acute angle-closure glaucoma may include a history of sulfonamide or penicillin allergy.
Diabetes and Hypoglycemia: Latent diabetes mellitus may become manifest and diabetic patients given thiazides may require adjustment of their insulin dose.
Renal Disease: Cumulative effects of the thiazides may develop in patients with impaired renal function. In such patients, thiazides may precipitate azotemia.
-
PRECAUTIONS
Electrolyte and Fluid Balance Status: In published studies, clinically significant hypokalemia has been consistently less common in patients who received 12.5 mg of hydrochlorothiazide than in patients who received higher doses. Nevertheless, periodic determination of serum electrolytes should be performed in patients who may be at risk for the development of hypokalemia. Patients should be observed for signs of fluid or electrolyte disturbances, i.e., hyponatremia, hypochloremic alkalosis, and hypokalemia and hypomagnesemia.
Warning signs or symptoms of fluid and electrolyte imbalance include dryness of mouth, thirst, weakness, lethargy, drowsiness, restlessness, muscle pains or cramps, muscular fatigue, hypotension, oliguria, tachycardia, and gastrointestinal disturbances such as nausea and vomiting.
Hypokalemia may develop, especially with brisk diuresis when severe cirrhosis is present, during concomitant use of corticosteroid or adrenocorticotropic hormone (ACTH) or after prolonged therapy. Interference with adequate oral electrolyte intake will also contribute to hypokalemia. Hypokalemia and hypomagnesemia can provoke ventricular arrhythmias or sensitize or exaggerate the response of the heart to the toxic effects of digitalis. Hypokalemia may be avoided or treated by potassium supplementation or increased intake of potassium rich foods.
Dilutional hyponatremia is life-threatening and may occur in edematous patients in hot weather; appropriate therapy is water restriction rather than salt administration, except in rare instances when the hyponatremia is life-threatening. In actual salt depletion, appropriate replacement is the therapy of choice.
Hyperuricemia: Hyperuricemia or acute gout may be precipitated in certain patients receiving thiazide diuretics.
Impaired Hepatic Function: Thiazides should be used with caution in patients with impaired hepatic function. They can precipitate hepatic coma in patients with severe liver disease.
Parathyroid Disease: Calcium excretion is decreased by thiazides, and pathologic changes in the parathyroid glands, with hypercalcemia and hypophosphatemia, have been observed in a few patients on prolonged thiazide therapy.
Drug Interactions: When given concurrently the following drugs may interact with thiazide diuretics:
Alcohol, barbiturates, or narcotics - potentiation of orthostatic hypotension may occur.
Antidiabetic drugs - (oral agents and insulin) dosage adjustment of the antidiabetic drug may be required.
Other antihypertensive drugs - additive effect or potentiation.
Cholestyramine and colestipol resins - Cholestyramine and colestipol resins bind the hydrochlorothiazide and reduce its absorption from the gastrointestinal tract by up to 85 and 43 percent, respectively.
Corticosteroid, ACTH – intensified electrolyte depletion, particularly hypokalemia.
Pressor amines (e.g., norepinephrine) - possible decreased response to pressor amines but not sufficient to preclude their use.
Skeletal muscle relaxants, nondepolarizing (e.g., tubocurarine) - possible increased responsiveness to the muscle relaxant.
Lithium - generally should not be given with diuretics. Diuretic agents reduce the renal clearance of lithium and greatly increase the risk of lithium toxicity. Refer to the package insert for lithium preparations before use of such preparations with hydrochlorothiazide.
Non-steroidal anti-inflammatory drugs - In some patients, the administration of a non-steroidal anti-inflammatory agent can reduce the diuretic, natriuretic, and antihypertensive effects of loop, potassium-sparing and thiazide diuretics. When hydrochlorothiazide and non-steroidal anti-inflammatory agents are used concomitantly, the patients should be observed closely to determine if the desired effect of the diuretic is obtained.
Drug/Laboratory Test Interactions - Thiazides should be discontinued before carrying out tests for parathyroid function (see PRECAUTIONS, General).
Carcinogenesis, Mutagenesis, Impairment of Fertility: Two-year feeding studies in mice and rats conducted under the auspices of the National Toxicology Program (NTP) uncovered no evidence of a carcinogenic potential of hydrochlorothiazide in female mice (at doses of up to approximately 600 mg/kg/day) or in male and female rats (at doses of approximately 100 mg/kg/day). The NTP, however, found equivocal evidence for hepatocarcinogenicity in male mice. Hydrochlorothiazide was not genotoxic in vitro in the Ames mutagenicity assay of Salmonella typhimurium strains TA 98, TA 100, TA 1535, TA 1537, and TA 1538 and in the Chinese Hamster Ovary (CHO) test for chromosomal aberrations, or in vivo in assays using mouse germinal cell chromosomes, Chinese hamster bone marrow chromosomes, and the Drosophila sex-linked recessive lethal trait gene. Positive test results were obtained only in the in vitro CHO Sister Chromatid Exchange (clastogenicity) and in the Mouse Lymphoma Cell (mutagenicity) assays, using concentrations of hydrochlorothiazide from 43 to 1300 mcg/mL, and in the Aspergillus nidulans non-disjunction assay at an unspecified concentration.
Hydrochlorothiazide had no adverse effects on the fertility of mice and rats of either sex in studies wherein these species were exposed, via their diet, to doses of up to 100 and 4 mg/kg, respectively, prior to conception and throughout gestation.
Pregnancy:
Teratogenic Effects:
Pregnancy Category B: Studies in which hydrochlorothiazide was orally administered to pregnant mice and rats during their respective periods of major organogenesis at doses up to 3000 and 1000 mg hydrochlorothiazide/kg, respectively, provided no evidence of harm to the fetus.
There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Nonteratogenic Effects:
Thiazides cross the placental barrier and appear in cord blood. There is a risk of fetal or neonatal jaundice, thrombocytopenia, and possibly other adverse reactions that have occurred in adults.
Nursing Mothers:
Thiazides are excreted in breast milk. Because of the potential for serious adverse reactions in nursing infants, a decision should be made whether to discontinue nursing or to discontinue hydrochlorothiazide, taking into account the importance of the drug to the mother.
Elderly Use:
A greater blood pressure reduction and an increase in side effects may be observed in the elderly (i.e., > 65 years) with hydrochlorothiazide. Starting treatment with the lowest available dose of hydrochlorothiazide (12.5 mg) is therefore recommended. If further titration is required, 12.5 mg increments should be utilized.
-
ADVERSE REACTIONS
The adverse reactions associated with hydrochlorothiazide have been shown to be dose related. In controlled clinical trials, the adverse events reported with doses of 12.5 mg hydrochlorothiazide once daily were comparable to placebo. The following adverse reactions have been reported for doses of hydrochlorothiazide 25 mg and greater and, within each category, are listed in the order of decreasing severity.
Body as a whole: Weakness.
Cardiovascular: Hypotension including orthostatic hypotension (may be aggravated by alcohol, barbiturates, narcotics or antihypertensive drugs).
Digestive: Pancreatitis, jaundice (intrahepatic cholestatic jaundice), diarrhea, vomiting, sialadenitis, cramping, constipation, gastric irritation, nausea, anorexia.
Hematologic: Aplastic anemia, agranulocytosis, leukopenia, hemolytic anemia, thrombocytopenia.
Hypersensitivity: Anaphylactic reactions, necrotizing angiitis (vasculitis and cutaneous vasculitis), respiratory distress including pneumonitis and pulmonary edema, photosensitivity, fever, urticaria, rash, purpura.
Metabolic: Electrolyte imbalance (see PRECAUTIONS), hyperglycemia, glycosuria, hyperuricemia.
Musculoskeletal: Muscle spasm.
Nervous System/Psychiatric: Vertigo, paresthesia, dizziness, headache, restlessness.
Renal: Renal failure, renal dysfunction, interstitial nephritis (see WARNINGS).
Skin: Erythema multiforme including Stevens-Johnson syndrome, exfoliative dermatitis including toxic epidermal necrolysis, alopecia.
Special Senses: Transient blurred vision, xanthopsia.
Urogenital: Impotence.
Whenever adverse reactions are moderate or severe, thiazide dosage should be reduced or therapy withdrawn.
-
OVERDOSAGE
The most common signs and symptoms observed are those caused by electrolyte depletion (hypokalemia, hypochloremia, hyponatremia) and dehydration resulting from excessive diuresis. If digitalis has also been administered, hypokalemia may accentuate cardiac arrhythmias.
In the event of overdosage, symptomatic and supportive measures should be employed. Emesis should be induced or gastric lavage performed. Correct dehydration, electrolyte imbalance, hepatic coma and hypotension by established procedures. If required, give oxygen or artificial respiration for respiratory impairment. The degree to which hydrochlorothiazide is removed by hemodialysis has not been established.
The oral LD50 of hydrochlorothiazide is greater than 10 g/kg in the mouse and rat.
- DOSAGE AND ADMINISTRATION
-
HOW SUPPLIED
Hydrochlorothiazide Capsules, USP are #4 Teal Opaque/White Opaque two piece hard gelatin capsules imprinted with WATSON 347 and 12.5 mg in black ink. They are supplied in bottles of 100 and 500. Bottles of 100 are supplied with child resistant closures.
Dispense in a tight, light-resistant container as defined in the USP.
Keep out of reach of children.
Store at 20°- 25°C (68°-77°F). [See USP controlled room temperature.] Protect from light, moisture, freezing, -20°C (-4°F). Keep container tightly closed.
Rx only
For all medical inquiries contact:
WATSON
Medical Communications
Parsippany, NJ 07054 USA
800-272-5525Manufactured By:
Watson Pharma Private Limited
Verna, Salcette Goa 403 722 INDIADistributed By:
Watson Pharma, Inc.
Parsippany, NJ 07054 USARevised: October 2011
174788-3 -
Repackaging Information
Please reference the How Supplied section listed above for a description of individual tablets. This drug product has been received by Aphena Pharma - TN in a manufacturer or distributor packaged configuration and repackaged in full compliance with all applicable cGMP regulations. The package configurations available from Aphena are listed below:
Count 12.5 mg 90 67544-739-60 Store between 20°-25°C (68°-77°F). See USP Controlled Room Temperature. Dispense in a tight light-resistant container as defined by USP. Keep this and all drugs out of the reach of children.
Repackaged by:

Cookeville, TN 38506
20171221DKJ - PRINCIPAL DISPLAY PANEL - 12.5mg
-
INGREDIENTS AND APPEARANCE
HYDROCHLOROTHIAZIDE
hydrochlorothiazide capsule, gelatin coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:67544-739(NDC:0591-0347) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYDROCHLOROTHIAZIDE (UNII: 0J48LPH2TH) (HYDROCHLOROTHIAZIDE - UNII:0J48LPH2TH) HYDROCHLOROTHIAZIDE 12.5 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STARCH, CORN (UNII: O8232NY3SJ) D&C RED NO. 28 (UNII: 767IP0Y5NH) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) GELATIN (UNII: 2G86QN327L) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERROSOFERRIC OXIDE (UNII: XM0M87F357) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) FD&C RED NO. 40 (UNII: WZB9127XOA) Product Characteristics Color WHITE (WHITE OPAQUE) , TURQUOISE (TEAL OPAQUE) Score no score Shape CAPSULE Size 14mm Flavor Imprint Code WATSON;347;and;12;5;mg Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:67544-739-60 90 in 1 PACKAGE; Type 0: Not a Combination Product 10/21/2014 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA AUTHORIZED GENERIC NDA020504 12/27/1996 Labeler - Aphena Pharma Solutions - Tennessee, LLC (128385585) Establishment Name Address ID/FEI Business Operations Aphena Pharma Solutions - Tennessee, LLC 128385585 REPACK(67544-739)


