Label: OPRECARE 3SET (07,12,21)- sodium monofluorophosphate, silicon dioxide kit
-
Contains inactivated NDC Code(s)
NDC Code(s): 71764-100-01, 71764-100-02, 71764-101-01, 71764-101-02, view more71764-102-01, 71764-102-02, 71764-104-01 - Packager: O'PRECARE
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated October 12, 2017
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
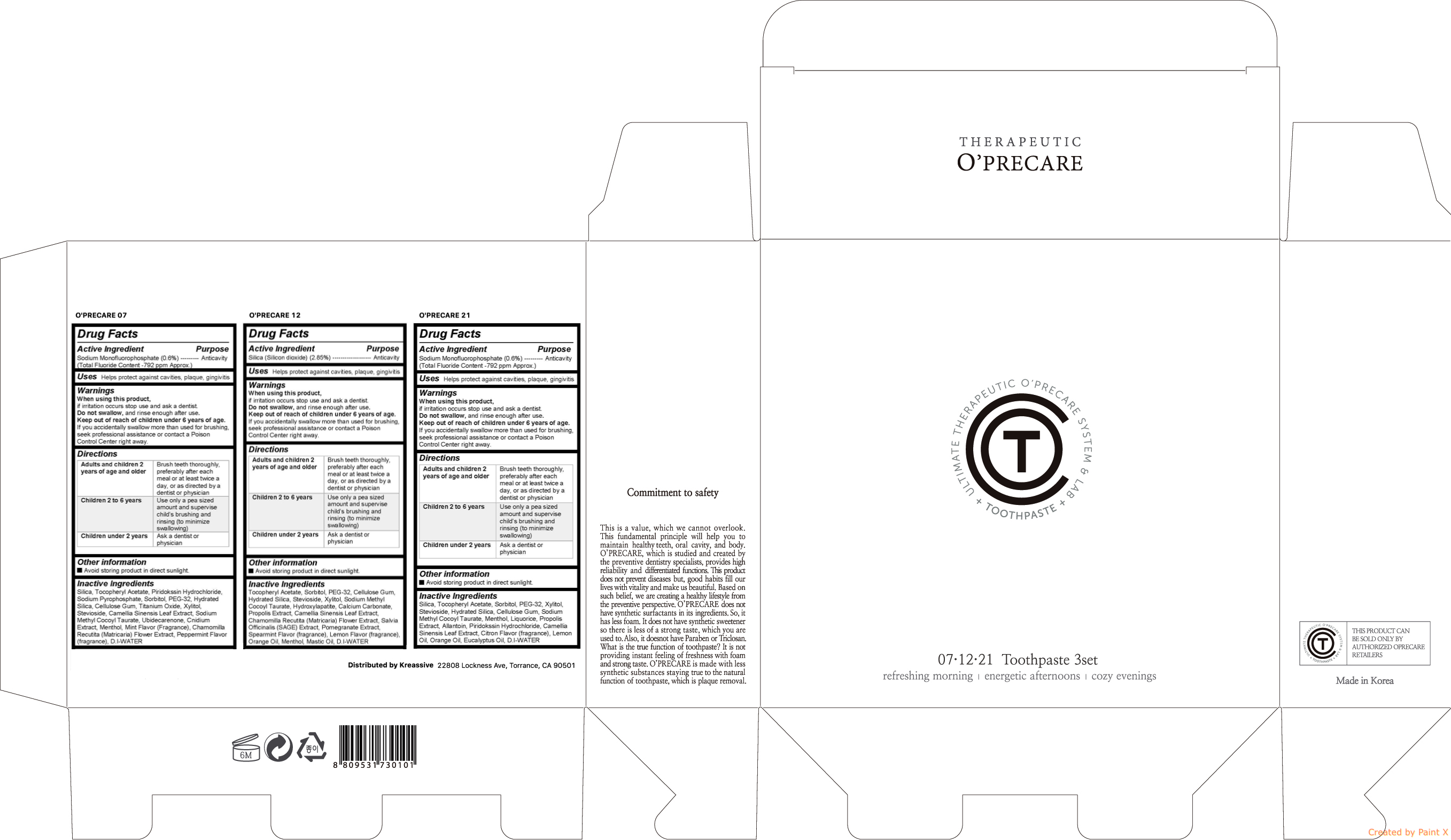
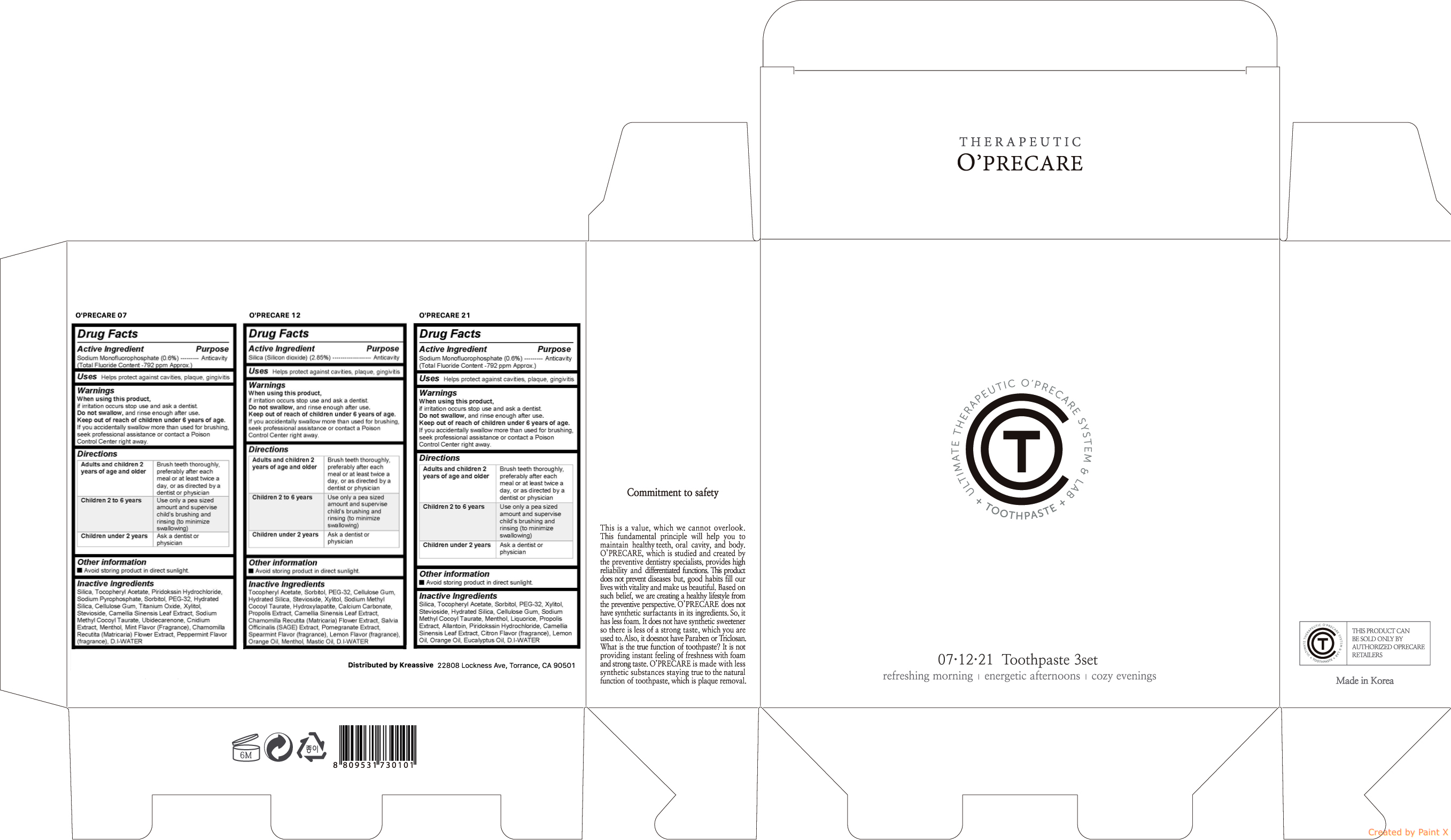
- Active ingredient
- Purpose
-
Warnings
When using this product, if irritation occurs stop use and ask a dentist. Do not swallow, and rinse enough after use. Keep out of reach of children under 6 years of age. If you accidentally swallow more than used for brushing, seek professional assistance or contact a Poison Control Center right away.
- Uses
-
Directions
Directions Adults and children 2 years of age and older Brush teeth thoroughly, preferably after each meal or at least twice a day, or as directed by a dentist or physician Children 2 to 6 years Use only a pea sized amount and supervise childs brushing and rinsing (to minimize swallowing) Children under 2 years Ask a dentist or physician - Keep out of reach of children
-
Inactive ingredients
Inactive ingredients O'PRECARE 07 Silica, Tocopheryl Acetate, Piridokssin Hydrochloride, Sodium Pyrophosphate, Sorbitol, PEG-32, Hydrated Silica, Cellulose Gum, Titanium Oxide, Xylitol, Stevioside, Camellia Sinensis Leaf Extract, Sodium Methyl Cocoyl Taurate, Ubidecarenone, Cnidium Extract, Menthol, Mint Flavor (Fragrance), Chamomilla Recutita (Matricaria) Flower Extract, Peppermint Flavor (fragrance), D.I-WATER O'PRECARE 12 Tocopheryl Acetate, Sorbitol, PEG-32, Cellulose Gum, Hydrated Silica, Stevioside, Xylitol, Sodium Methyl Cocoyl Taurate, Hydroxylapatite, Calcium Carbonate, Propolis Extract, Camellia Sinensis Leaf Extract, Chamomilla Recutita (Matricaria) Flower Extract, Salvia Officinalis (SAGE) Extract, Pomegranate Extract, Spearmint Flavor (fragrance), Lemon Flavor (fragrance), Orange Oil, Menthol, Mastic Oil, D.I-WATER O'PRECARE 21 Silica, Tocopheryl Acetate, Piridokssin Hydrochloride, Sodium Pyrophosphate, Sorbitol, PEG-32, Hydrated Silica, Cellulose Gum, Titanium Oxide, Xylitol, Stevioside, Camellia Sinensis Leaf Extract, Sodium Methyl Cocoyl Taurate, Ubidecarenone, Cnidium Extract, Menthol, Mint Flavor (Fragrance), Chamomilla Recutita (Matricaria) Flower Extract, Peppermint Flavor (fragrance), D.I-WATER - O'PRECARE TOOTHPASTE 3SET (07,12,21)
-
INGREDIENTS AND APPEARANCE
OPRECARE 3SET (07,12,21)
sodium monofluorophosphate, silicon dioxide kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71764-104 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71764-104-01 1 in 1 PACKAGE; Type 1: Convenience Kit of Co-Package 10/12/2017 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BOTTLE, PUMP 60 g Part 2 1 BOTTLE, PUMP 60 g Part 3 1 BOTTLE, PUMP 60 g Part 1 of 3 OPRECARE 07
sodium monofluorophosphate pasteProduct Information Item Code (Source) NDC:71764-100 Route of Administration DENTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM MONOFLUOROPHOSPHATE (UNII: C810JCZ56Q) (FLUORIDE ION - UNII:Q80VPU408O) FLUORIDE ION 0.36 g in 60 g Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED FORM (UNII: K679OBS311) XYLITOL (UNII: VCQ006KQ1E) MENTHOL (UNII: L7T10EIP3A) SODIUM METHYL COCOYL TAURATE (UNII: JVL98CG53G) GREEN TEA LEAF (UNII: W2ZU1RY8B0) UBIDECARENONE (UNII: EJ27X76M46) SODIUM PYROPHOSPHATE (UNII: O352864B8Z) SORBITOL (UNII: 506T60A25R) HYDRATED SILICA (UNII: Y6O7T4G8P9) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) STEVIOSIDE (UNII: 0YON5MXJ9P) POLYETHYLENE GLYCOL 1500 (UNII: 1212Z7S33A) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) CHAMOMILE (UNII: FGL3685T2X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71764-100-02 1 in 1 PACKAGE 1 NDC:71764-100-01 60 g in 1 BOTTLE, PUMP; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part355 10/12/2017 Part 2 of 3 OPRECARE 12
silicon dioxide gelProduct Information Item Code (Source) NDC:71764-101 Route of Administration DENTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) (SILICON DIOXIDE - UNII:ETJ7Z6XBU4) SILICON DIOXIDE 1.71 g in 60 g Inactive Ingredients Ingredient Name Strength .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) SORBITOL (UNII: 506T60A25R) POLYETHYLENE GLYCOL 1500 (UNII: 1212Z7S33A) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED FORM (UNII: K679OBS311) HYDRATED SILICA (UNII: Y6O7T4G8P9) STEVIOSIDE (UNII: 0YON5MXJ9P) Xylitol (UNII: VCQ006KQ1E) SODIUM METHYL COCOYL TAURATE (UNII: JVL98CG53G) TRIBASIC CALCIUM PHOSPHATE (UNII: 91D9GV0Z28) Calcium Carbonate (UNII: H0G9379FGK) PROPOLIS WAX (UNII: 6Y8XYV2NOF) GREEN TEA LEAF (UNII: W2ZU1RY8B0) CHAMOMILE (UNII: FGL3685T2X) SALVIA OFFICINALIS ROOT (UNII: 236QY0A1BL) POMEGRANATE (UNII: 56687D1Z4D) ORANGE OIL (UNII: AKN3KSD11B) Menthol (UNII: L7T10EIP3A) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71764-101-02 1 in 1 PACKAGE 1 NDC:71764-101-01 60 g in 1 BOTTLE, PUMP; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 10/12/2017 Part 3 of 3 OPRECARE 21
sodium monofluorophosphate gelProduct Information Item Code (Source) NDC:71764-102 Route of Administration DENTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM MONOFLUOROPHOSPHATE (UNII: C810JCZ56Q) (FLUORIDE ION - UNII:Q80VPU408O) FLUORIDE ION 0.36 g in 60 g Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) SORBITOL (UNII: 506T60A25R) POLYETHYLENE GLYCOL 1500 (UNII: 1212Z7S33A) Xylitol (UNII: VCQ006KQ1E) Stevioside (UNII: 0YON5MXJ9P) Hydrated Silica (UNII: Y6O7T4G8P9) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED FORM (UNII: K679OBS311) SODIUM METHYL COCOYL TAURATE (UNII: JVL98CG53G) Menthol (UNII: L7T10EIP3A) LICORICE (UNII: 61ZBX54883) PROPOLIS WAX (UNII: 6Y8XYV2NOF) Allantoin (UNII: 344S277G0Z) PIRIDOCAINE HYDROCHLORIDE (UNII: VG6P406YHV) GREEN TEA LEAF (UNII: W2ZU1RY8B0) Lemon Oil (UNII: I9GRO824LL) Orange Oil (UNII: AKN3KSD11B) Eucalyptus Oil (UNII: 2R04ONI662) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71764-102-02 1 in 1 PACKAGE 1 NDC:71764-102-01 60 g in 1 BOTTLE, PUMP; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part355 10/12/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 10/12/2017 Labeler - O'PRECARE (694604592) Registrant - O'PRECARE (694604592) Establishment Name Address ID/FEI Business Operations O'PRECARE 694604592 relabel(71764-104) Establishment Name Address ID/FEI Business Operations Kolmar Korea Co., Ltd. 689512611 manufacture(71764-104)