Label: ESOMEPRAZOLE MAGNESIUM capsule, delayed release
- NDC Code(s): 16571-880-41, 16571-880-42
- Packager: Rising Pharma Holdings, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated May 11, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Active ingredient (in each capsule)
Purpose
Esomeprazole 20 mg
Acid reducer
(Each delayed-release capsule corresponds to 22.250 mg esomeprazole magnesium trihydrate)
- treats frequent heartburn (occurs 2 or more days a week)
- not intended for immediate relief of heartburn; this drug may take 1 to 4 days for full effect
Allergy alert
Do not use if you are allergic to esomeprazole
Do not use if you have
- trouble or pain swallowing food, vomiting with blood, or bloody or black stools
- heartburn with lightheadedness, sweating or dizziness
- chest pain or shoulder pain with shortness of breath; sweating; pain spreading to arms, neck or shoulders; or lightheadedness
- frequent chest pain
These may be signs of a serious condition. See your doctor.
Ask a doctor before use if you have
- had heartburn over 3 months. This may be a sign of a more serious condition.
- frequent wheezing, particularly with heartburn
- unexplained weight loss
- nausea or vomiting
- stomach pain
Ask a doctor or pharmacist before use if you are taking a prescription drug. Acid reducers may interact with certain prescription drugs.
Stop use and ask a doctor if
- your heartburn continues or worsens
- you need to take this product for more than 14 days
- you need to take more than 1 course of treatment every 4 months
- you get diarrhea
- you develop a rash or joint pain
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
- adults 18 years of age and older
- this product is to be used once a day (every 24 hours), every day for 14 days
- may take 1 to 4 days for full effect
- swallow 1 capsule with a glass of water before eating in the morning
- take every day for 14 days
- do not take more than 1 capsule a day
- swallow whole. Do not crush or chew capsules.
- do not use for more than 14 days unless directed by your doctor
Repeated 14-Day Courses (if needed)
- you may repeat a 14-day course every 4 months
- do not take for more than 14 days or more often than every 4 months unless directed by a doctor
- children under 18 years of age: ask a doctor before use. Heartburn in children may sometimes be caused by a serious condition.
- read the directions and warnings before use
- keep the carton. It contains important information.
- Store at 20° to 25°C (68° to 77°F).
Esomeprazole Magnesium Delayed-Release Capsules USP, 20 mg:
FD&C Blue 2, Gelatin, Hydroxypropyl Cellulose, Hypromellose, Magnesium Stearate, Methacrylic Acid and Ethyl Acrylate Copolymer Dispersion, Mono-and Di-Glycerides, Pharmaceutical Ink, Polysorbate 80, Sugar Spheres, Talc, Triethyl Citrate.
Questions or comments?
Call toll-free weekdays 9 AM to 5 PM EST at 1-844-474-7464.
Contact Graviti Pharmaceuticals Inc., or www.gravitipharma.com; or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Made in India
See new warning information
- Treats Frequent Heartburn
Esomeprazole magnesium delayed-release capsules 20 mg/acid reducer
May take 1 to 4 days for full effect
14 CAPSULES
One 14-day course of treatment Capsules
Tips for Managing Heartburn
- Avoid foods or drinks that are more likely to cause heartburn, such as rich, spicy, fatty and fried foods, chocolate, caffeine, alcohol and even some acidic fruits and vegetables.
- Eat slowly and do not eat big meals.
- Do not eat late at night or just before bedtime.
- Do not lie flat or bend over soon after eating.
- Raise the head of your bed.
- Wear loose-fitting clothing
- around your stomach.
- If you are overweight, lose weight.
- If you smoke, quit smoking
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- WARNINGS
- DOSAGE & ADMINISTRATION
- INACTIVE INGREDIENT
-
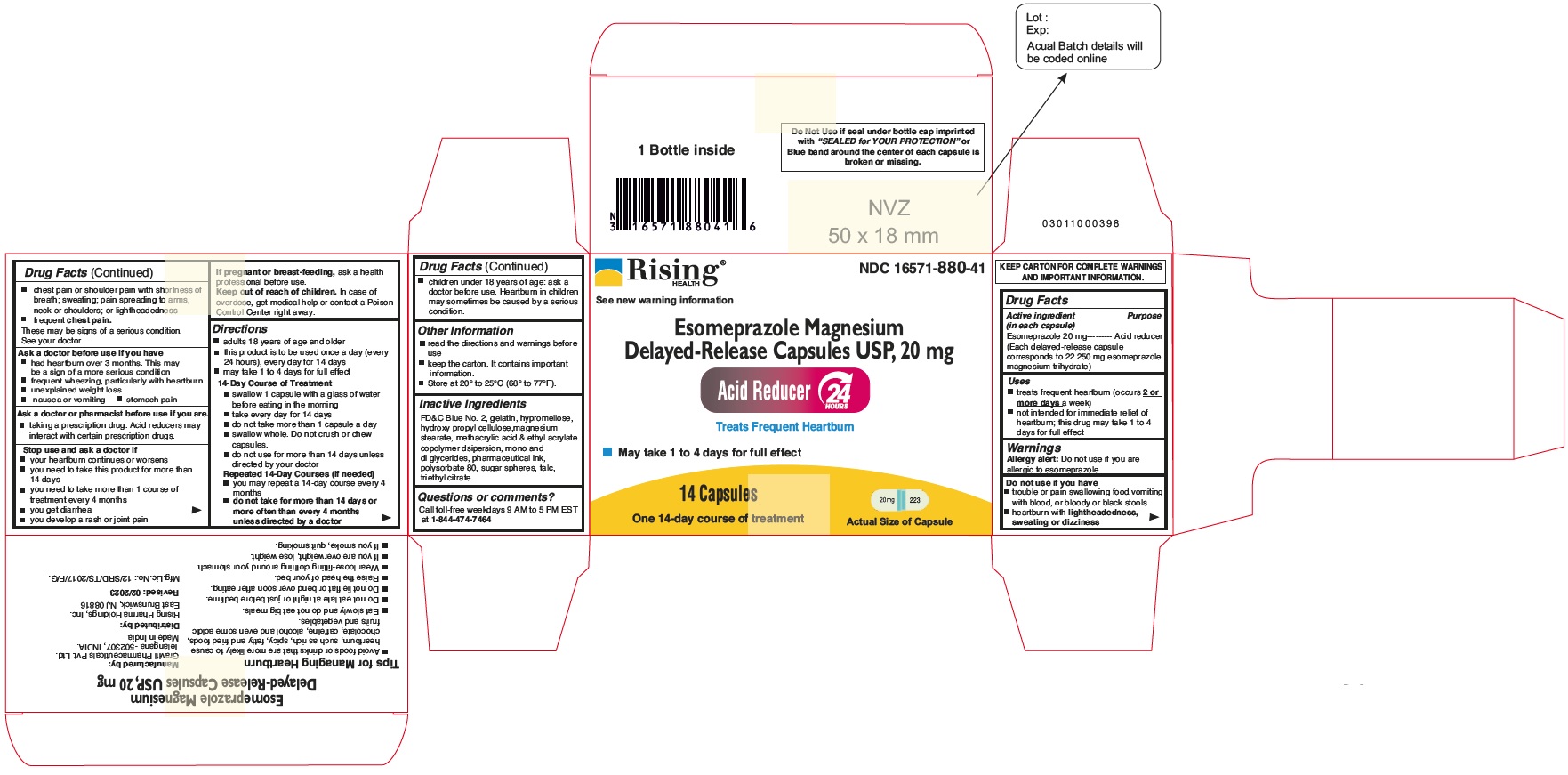
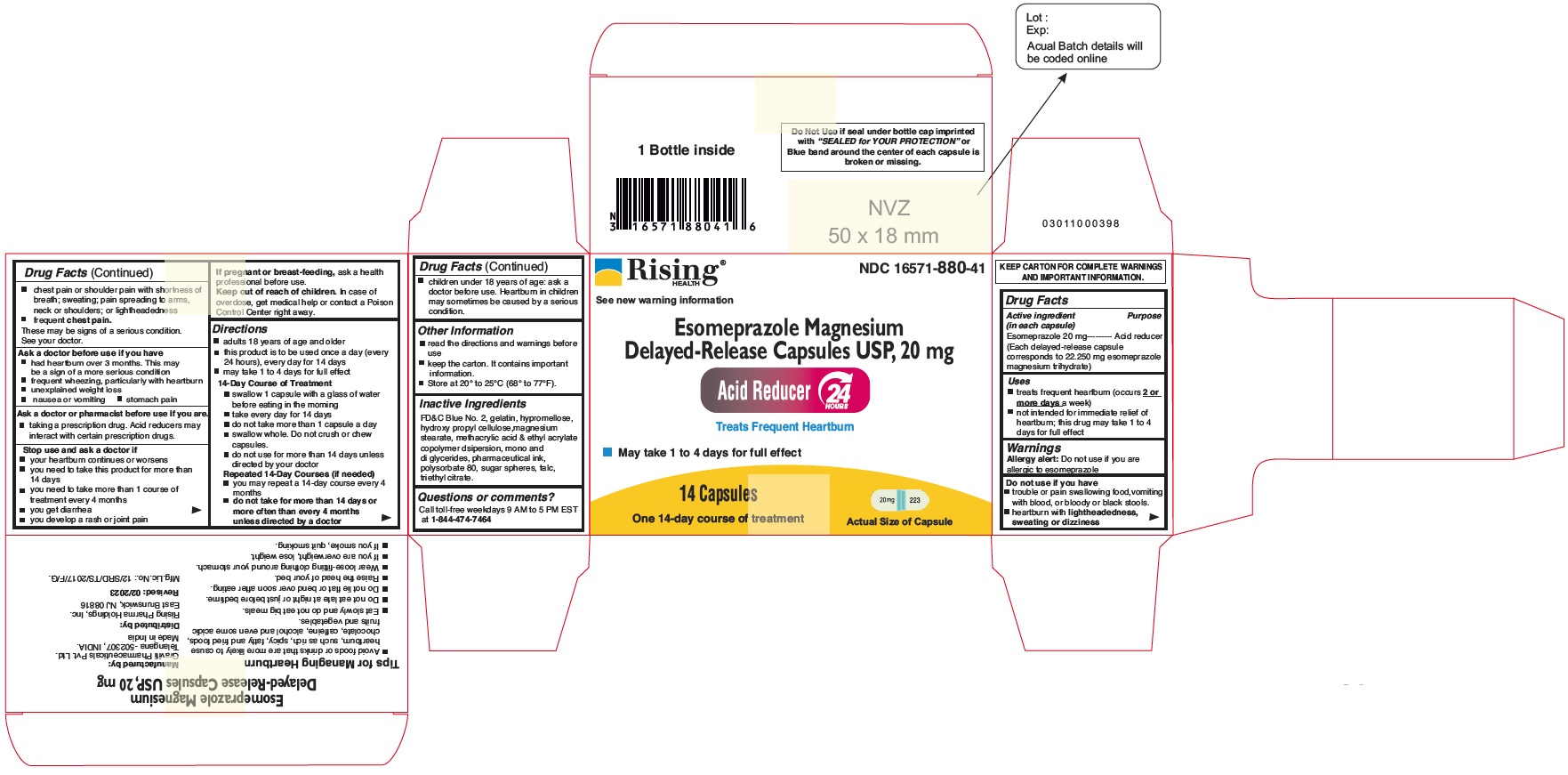
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Principal Display Panel - 20 mg Capsules: Container Label
NDC 16571-880-41
Esomeprazole Magnesium Delayed-Release Capsules USP, 20 mg
24 HR
Acid Reducer
Treats Frequent Heartburn
May take 1 to 4 Days for Full effect
14 Capsules
One 14-day Course of Treatment
Distributed by: Rising Pharma Holdings, Inc.

Principal Display Panel - 20 mg Capsules: Carton Label
NDC 16571-880-41
Esomeprazole Magnesium Delayed-Release Capsules USP, 20 mg
24 HR
Acid Reducer
Treats Frequent Heartburn
May take 1 to 4 Days for Full effect
14 Capsules
One 14-day Course of Treatment
Distributed by: Rising Pharma Holdings, Inc.
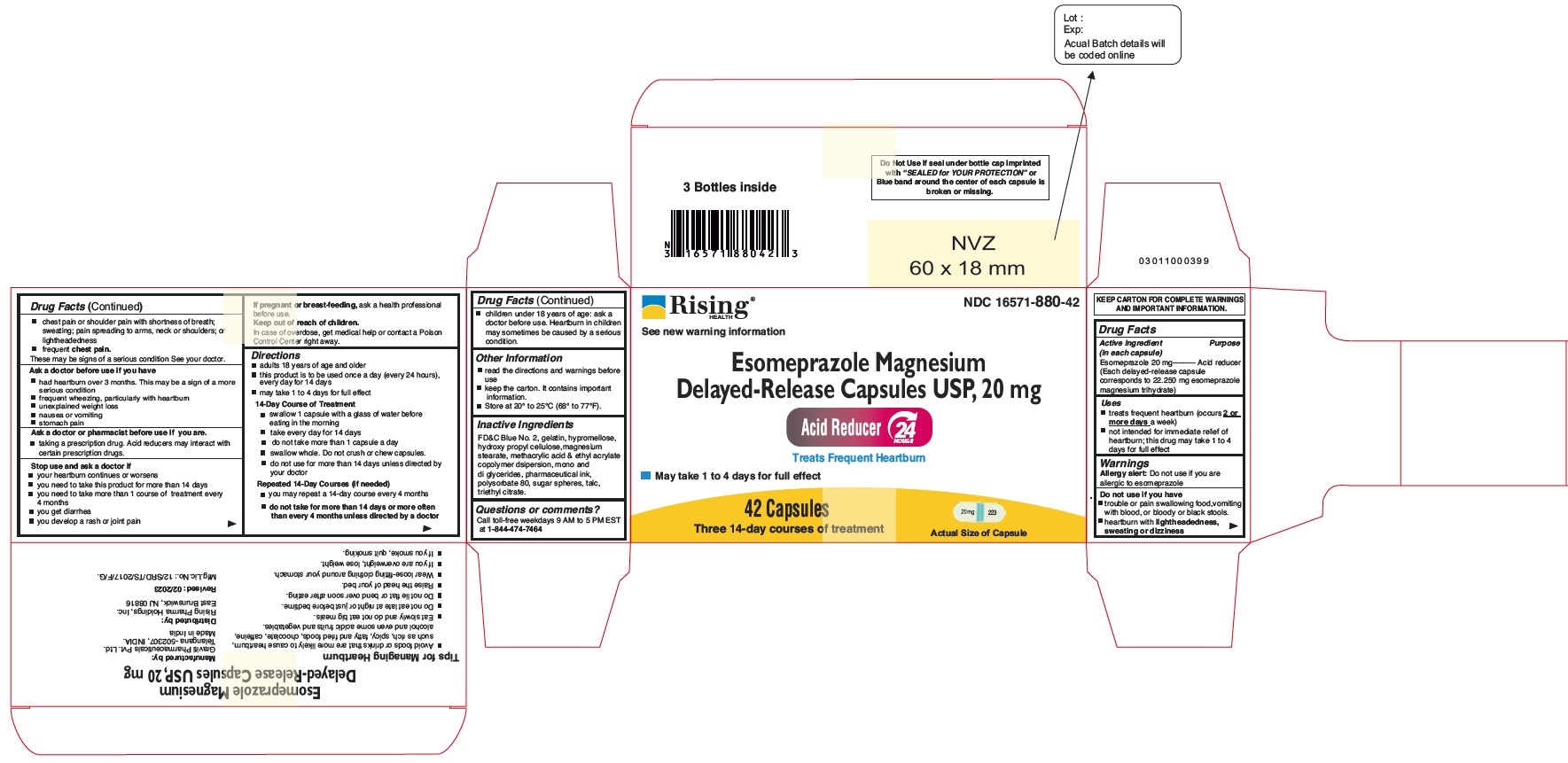
Principal Display Panel - 20 mg Capsules: Carton Label
NDC 16571-880-42
Esomeprazole Magnesium Delayed-Release Capsules USP, 20 mg
24 HR
Acid Reducer
Treats Frequent Heartburn
May take 1 to 4 Days for Full effect
42 Capsules
Three 14-day Courses of Treatment
Distributed by: Rising Pharma Holdings, Inc.
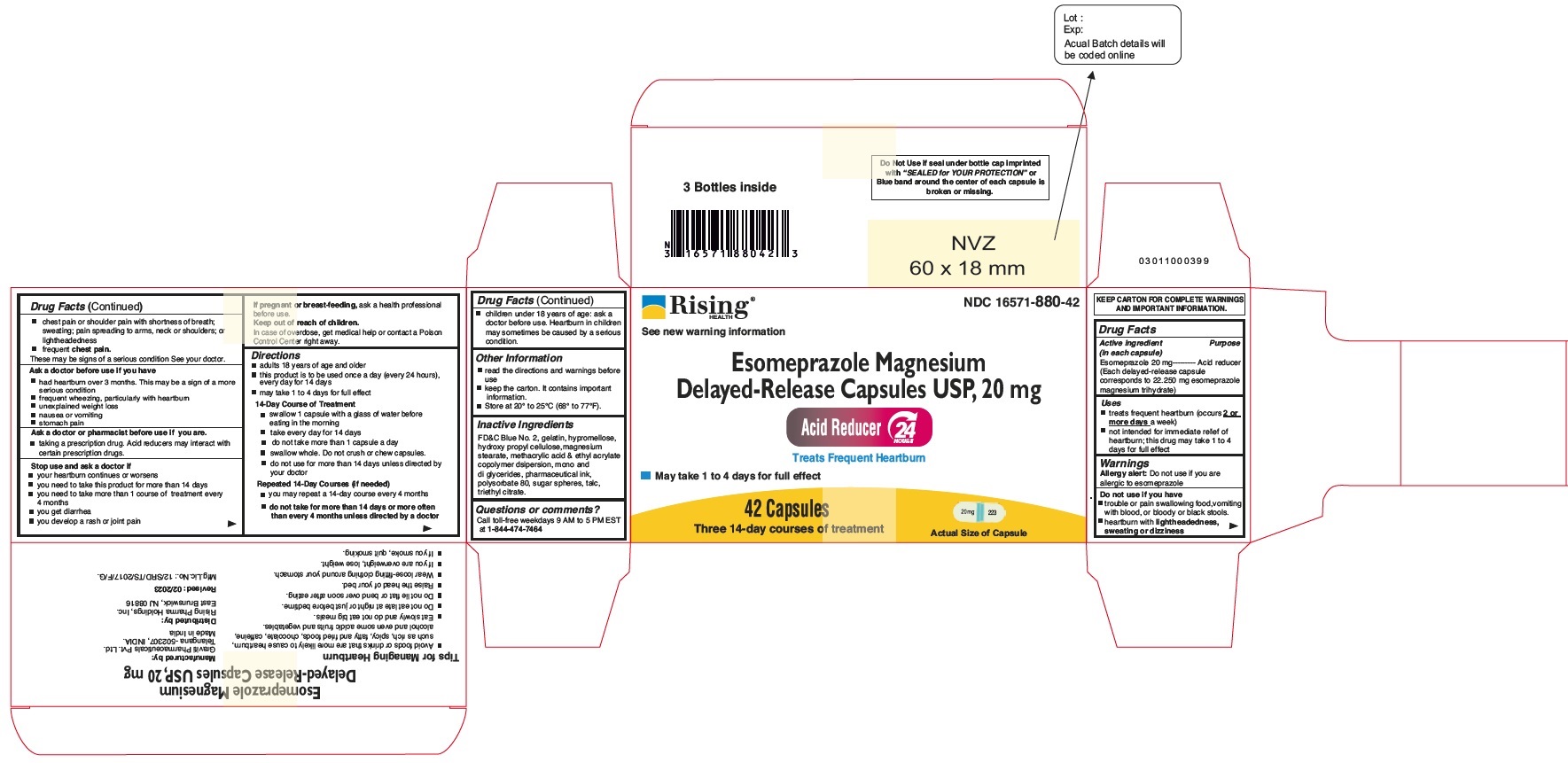
-
INGREDIENTS AND APPEARANCE
ESOMEPRAZOLE MAGNESIUM
esomeprazole magnesium capsule, delayed releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:16571-880 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ESOMEPRAZOLE MAGNESIUM (UNII: R6DXU4WAY9) (ESOMEPRAZOLE - UNII:N3PA6559FT) ESOMEPRAZOLE 20 mg Inactive Ingredients Ingredient Name Strength RAW SUGAR (UNII: 8M707QY5GH) HYPROMELLOSE 2910 (5 MPA.S) (UNII: R75537T0T4) POLYSORBATE 80 (UNII: 6OZP39ZG8H) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) TALC (UNII: 7SEV7J4R1U) MAGNESIUM STEARATE (UNII: 70097M6I30) METHACRYLIC ACID-ETHYL ACRYLATE COPOLYMER (1:1) TYPE A (UNII: NX76LV5T8J) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) GLYCERYL DISTEARATE (UNII: 73071MW2KM) Product Characteristics Color WHITE (white opaque colored) Score no score Shape CAPSULE Size 14mm Flavor Imprint Code 20mg;223 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:16571-880-41 1 in 1 CARTON 12/01/2022 1 14 in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:16571-880-42 3 in 1 CARTON 12/01/2022 2 14 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA216349 12/01/2022 Labeler - Rising Pharma Holdings, Inc. (116880195) Establishment Name Address ID/FEI Business Operations Graviti Pharmaceuticals Private Limited 650884781 MANUFACTURE(16571-880) , ANALYSIS(16571-880)