Label: ICG- indocyanine green and water kit
-
Contains inactivated NDC Code(s)
NDC Code(s): 0409-4887-17, 70599-424-02 - Packager: KARL STORZ Endoscopy-America, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated November 13, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use ICG for Injection Set safely and effectively. See full prescribing information for Indocyanine Green for Injection, USP.
Initial U.S. Approval: 1959INDICATIONS AND USAGE
ICG for Injection Set, a tricarbocyanine dye, is indicated for use with the KARL STORZ ICG Imaging System to provide real-time endoscopic visible and near-infrared fluorescence imaging. ICG for Injection Set used in conjunction with the KARL STORZ ICG Imaging System enables surgeons to perform minimally invasive surgery using standard endoscopic visible light as well as:
- •
- Visual assessment of vessels, blood flow and related tissue perfusion. (1.1)
- •
- Visual assessment of at least one of the major extra-hepatic bile ducts (cystic duct, common bile duct and common hepatic duct), using near-infrared imaging. Fluorescence imaging of biliary ducts with the KARL STORZ ICG Imaging System is intended for use with standard of care white light and, when indicated, intraoperative cholangiography. The device is not intended for standalone use for biliary duct visualization. (1.2)
DOSAGE AND ADMINISTRATION
Perfusion Assessment (2.1)
Under sterile conditions, reconstitute one (1) 25 mg vial of Indocyanine Green for Injection, USP using one (1) 10 mL Sterile Water for Injection, USP vial located in the ICG for Injection Set. Shake the ICG vial gently to dissolve. After reconstitution, a 25 mg vial of ICG contains 2.5 mg of dye per mL of solution, so a 1.0 mL injection contains a 2.5 mg dose of ICG. ICG must be used within 6 hours after reconstitution.
A 3 mL (7.5 mg) dose, followed by a 10 mL bolus of saline, is recommended. Multiple doses can be administered as required.
Extra-Hepatic Biliary Anatomy (2.2)
Under sterile conditions, reconstitute one (1) 25 mg vial of Indocyanine Green for Injection, USP using one (1) 10 mL Sterile Water for Injection, USP vial located in the ICG for Injection Set. Shake the ICG vial gently to dissolve. After reconstitution, a 25 mg vial of ICG contains 2.5 mg of dye per mL of solution, so a 1.0 mL injection contains a 2.5 mg dose of ICG. ICG must be used within 6 hours after reconstitution.
A 0.02 mL/kg dose that is scaled to the patient’s weight, followed by a 10 mL bolus of saline, is recommended. This provides 0.05 mg/kg of ICG.
DOSAGE FORMS AND STRENGTHS
Indocyanine Green for Injection, USP is a sterile, lyophilized green powder containing 25 mg of indocyanine green with no more than 5% sodium iodide. (3)
CONTRAINDICATIONS
Indocyanine Green for Injection, USP contains sodium iodide and should be used with caution in patients who have a history of allergy to iodides because of the risk of anaphylaxis. (4)
WARNINGS AND PRECAUTIONS
- •
- Deaths due to anaphylaxis have been reported following Indocyanine Green for Injection, USP administration during cardiac catheterization. (5.1)
- •
- Indocyanine Green for Injection, USP is unstable in aqueous solution and must be used within 6 hours. (5.2)
- •
- Radioactive iodine uptake studies should not be performed for at least a week following the use of Indocyanine Green for Injection, USP. (5.3)
ADVERSE REACTIONS
Most common adverse reactions are anaphylactic or urticarial reactions. These have been reported in patients with and without a history of allergy to iodides. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Diagnostic Green LLC at 1-844-424-3784 (1-844-ICG-DRUG) or e-mail: drugsafety@diagnosticgreen.com; or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
Products containing sodium bisulfite reduce the absorption peak of Indocyanine Green for Injection, USP in blood. (7)
Revised: 6/2016
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Visual assessment of vessels, blood flow and related tissue perfusion
1.2 Visual assessment of at least one of the major extra-hepatic bile ducts (cystic duct, common bile duct and common hepatic duct), using near-infrared imaging.
2 DOSAGE AND ADMINISTRATION
2.1 Perfusion Assessment
2.2 Extra-Hepatic Biliary Anatomy
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Anaphylaxis
5.2 Drug Instability
5.3 Drug/Laboratory Test Interactions
6 ADVERSE REACTIONS
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
16 HOW SUPPLIED/STORAGE AND HANDLING
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
ICG for Injection Set, a tricarbocyanine dye, is indicated for use with the KARL STORZ ICG Imaging System to provide real-time endoscopic visible and near-infrared fluorescence imaging. Indocyanine Green for Injection, USP used in conjunction with the KARL STORZ ICG Imaging System enables surgeons to perform minimally invasive surgery using standard endoscopic visible light as well as:
1.2 Visual assessment of at least one of the major extra-hepatic bile ducts (cystic duct, common bile duct and common hepatic duct), using near-infrared imaging.
Visual assessment of at least one of the major extra-hepatic bile ducts (cystic duct, common bile duct and common hepatic duct), using near-infrared imaging. Fluorescence imaging of biliary ducts with the KARL STORZ ICG Imaging System is intended for use with standard of care white light and, when indicated, intraoperative cholangiography. The device is not intended for standalone use for biliary duct visualization.
-
2 DOSAGE AND ADMINISTRATION
2.1 Perfusion Assessment
Preparation of ICG for Administration
Under sterile conditions, reconstitute one (1) 25 mg vial of Indocyanine Green for Injection, USP using one (1) 10 mL Sterile Water for Injection, USP vial located in the ICG for Injection Set. Shake the ICG vial gently to dissolve. After reconstitution, a 25 mg vial of ICG contains 2.5 mg of dye per mL of solution, so a 1.0 mL injection contains a 2.5 mg dose of ICG.
Indocyanine Green for Injection, USP must be used within 6 hours after reconstitution. If a precipitate is present, discard the solution.
Dosage
A 3 mL (7.5 mg) dose followed by a 10 mL bolus of saline is recommended. Multiple doses can be administered as required, up to the maximum recommended dose.
Maximum recommended dose
The total dose of dye injected should be kept below 2 mg/kg.
Timing of ICG Administration
ICG fluorescence is quickly visible within blood vessels, tissue and organs, and it does not last very long (refer to Table 1 below).
Table 1: ICG fluorescence visibility after intravenous injection Blood Vessels
Organs (Kidney, Liver, Adrenal Gland, Small Bowel)
See within:
5-30 seconds
1-2 minutes
Visibility lasts:
20-30 seconds
20-120 minutes
For fluorescence imaging of PERFUSION in blood vessels, administration of the ICG should be performed at the time fluorescence imaging is requested by the physician. Multiple imaging sequences may be performed as necessary [up to the maximum dose (2 mg/kg of patient body weight)], so it is recommended to withdraw the desired dosage of ICG solution for each planned imaging sequence into separate syringes ahead of time.
Method of Administration
ICG administration is to be performed via a central or peripheral venous line. Inject the prepared dose of ICG solution into the central or peripheral line as a tight bolus and immediately followed by a bolus of 10-12 mL of normal saline for injection.
2.2 Extra-Hepatic Biliary Anatomy
Preparation of ICG for Administration
Under sterile conditions, reconstitute one (1) 25 mg vial of Indocyanine Green for Injection, USP using one (1) 10 mL Sterile Water for Injection, USP vial located in the ICG for Injection Set. Shake the ICG vial gently to dissolve. After reconstitution, a 25 mg vial of ICG contains 2.5 mg of dye per mL of solution, so a 1.0 mL injection contains a 2.5 mg dose of ICG.
Indocyanine Green for Injection, USP must be used within 6 hours after reconstitution. If a precipitate is present, discard the solution.
Dosage
A 0.02 mL/kg dose that is scaled to the patient’s weight is recommended. This provides 0.05 mg/kg of ICG.
Maximum recommended dose
The total dose of dye injected should be kept below 2 mg/kg.
Timing of ICG Administration
Following intravenous injection, ICG is rapidly bound to plasma protein, of which albumin is the principle carrier (95%). ICG is taken up from the plasma almost exclusively by the hepatic parenchymal cells and is secreted entirely into the bile. For optimal fluorescence imaging of Extra-Hepatic Biliary Anatomy, ICG should be administered at least 45 minutes prior to the time fluorescence imaging is desired by the physician. If this preoperative administration is not performed, however, ICG can be administered once the patient is in the OR, as adequate fluorescence imaging is possible in as little as 15 minutes after IV injection.
Method of Administration
ICG administration is to be performed via a central or peripheral venous line. Inject the prepared weight-scaled dose of ICG solution into the central or peripheral line as a tight bolus and immediately followed by a bolus of 10-12 mL of normal saline for injection.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Anaphylaxis
Deaths from anaphylaxis have been reported following Indocyanine Green for Injection, USP administration during cardiac catheterization.
5.2 Drug Instability
Indocyanine Green for Injection, USP is unstable in aqueous solution and must be used within 6 hours. However, the dye is stable in plasma and whole blood so that samples obtained in discontinuous sampling techniques may be read hours later. Sterile techniques should be used in handling the dye solution as well as in the performance of the procedures. If a precipitate is present, discard the solution.
- 6 ADVERSE REACTIONS
- 7 DRUG INTERACTIONS
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Animal reproduction studies have not been conducted with Indocyanine Green for Injection, USP. It is also not known whether Indocyanine Green for Injection, USP can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Indocyanine Green for Injection, USP should be given to a pregnant woman only if clearly indicated.
- 10 OVERDOSAGE
-
11 DESCRIPTION
Indocyanine Green for Injection, USP is a sterile, lyophilized green powder containing 25 mg of indocyanine green with no more than 5% sodium iodide. It is packaged with Sterile Water for Injection, USP used to dissolve the indocyanine green. Indocyanine Green for Injection, USP is to be administered intravenously.
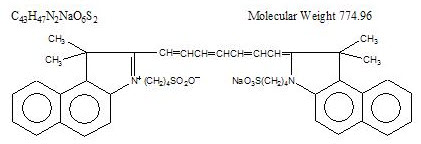
Indocyanine green is a water soluble, tricarbocyanine dye with a peak spectral absorption at 800 nm. The chemical name for Indocyanine Green is 1 H-Benz[e]indolium, 2-[7-[1,3-dihydro-1,1-dimethyl-3-(4-sulfobutyl)-2H-benz[e] indol-2-ylidene]-1,3,5-heptatrienyl]-1,1-dimethyl-3-(4-sulfobutyl)-,hydroxide, inner salt, sodium salt. Indocyanine Green for Injection, USP has a pH of approximately 6.5 when reconstituted. Each vial of Indocyanine Green for Injection, USP contains 25 mg of indocyanine green as a sterile lyophilized powder.
-
12 CLINICAL PHARMACOLOGY
Following intravenous injection, Indocyanine Green for Injection, USP is rapidly bound to plasma protein, of which albumin is the principle carrier (95%). Indocyanine Green for Injection, USP undergoes no significant extrahepatic or enterohepatic circulation; simultaneous arterial and venous blood estimations have shown negligible renal, peripheral, lung or cerebro-spinal uptake of the dye. Indocyanine Green for Injection, USP is taken up from the plasma almost exclusively by the hepatic parenchymal cells and is secreted entirely into the bile.
The peak absorption and emission of Indocyanine Green for Injection, USP lie in a region (800 to 850 nm) where transmission of energy by the pigment epithelium is more efficient than in the region of visible light energy. Indocyanine Green for Injection, USP also has the property of being nearly 98% bound to blood protein.
- 13 NONCLINICAL TOXICOLOGY
-
16 HOW SUPPLIED/STORAGE AND HANDLING
ICG for Injection Set is a kit (NDC 70599-424-02) containing one Indocyanine Green for Injection, USP kit (NDC 70100-424-02) and these Instructions For Use with the KARL STORZ ICG Imaging System.
The Indocyanine Green for Injection, USP kit (NDC 70100-424-02) contains six 25 mg Indocyanine Green for Injection, USP vials and six 10 mL Sterile Water for Injection, USP plastic vials:
NDC 70100-424-01 Indocyanine Green for Injection, USP vial. 25 mg fill in 25 mL vial.
NDC 63323-185-10 (or NDC 0409-4887-17) Sterile Water for Injection, USP, 10 mL fill in 10 mL plastic vials.
ICG for Injection Set
Distributed by:
KARL STORZ Endoscopy-America, Inc.
El Segundo, CA 90245 USA50441
-
PRINCIPAL DISPLAY PANEL - KIT CARTON
Front Panel
NDC 70599-424-02 PN: VTG0001
ICG for Injection Set
For Intravenous Administration 25 mg/Vial Kit Rx Only - Sterile
STORZ
KARL STORZ-ENDOSKOPEBack Panel
NDC 70599-424-02 PN: VTG0001
ICG for Injection Set
Contents:
Six Indocyanine Green for Injection, USP vials (25 mg each)
Six Sterile Water for Injection, USP Vials (10 mL each)Lot Code Area
No Print / No CoatingSTORZ
KARL STORZ-ENDOSKOPEFor Intravenous Administration 25 mg/Vial Kit Rx Only - Sterile 06/2016
Left Panel
ICG for Injection Set
DIRECTIONS FOR USE ENCLOSED
CAUTION: To ensure accurate readings,
Indocyanine Green, USP dissolved in Sterile Water
for Injection, USP must be used within 6 hours.STORAGE: Store at 20° to 25°C (68° to 77°F)
[See USP Controlled Room Temperature].USAGE: See package insert for dosage information.
25 mg/Vial Kit
Right Panel
ICG for Injection Set
Distributed by:
KARL STORZ
Endoscopy-America, Inc.
2151 E. Grand Ave.
El Segundo, CA 90245
Phone: (800) 421-0837
Fax: (800) 321-1304
E-Mail: communications@karlstorz.com
Web: www.karlstorz.comRx Only
25 mg/Vial Kit
- PRINCIPAL DISPLAY PANEL - VIAL
- PRINCIPAL DISPLAY PANEL - STERILE WATER VIAL
-
INGREDIENTS AND APPEARANCE
ICG
indocyanine green and water kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70599-424 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70599-424-02 6 in 1 CARTON 06/01/2016 06/01/2020 1 1 in 1 PACKAGE; Type 0: Not a Combination Product Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 VIAL 1 Part 2 1 VIAL, PLASTIC 10 mL Part 1 of 2 INDOCYANINE GREEN
indocyanine green injection, powder, lyophilized, for solutionProduct Information Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength INDOCYANINE GREEN (UNII: IX6J1063HV) (INDOCYANINE GREEN ACID FORM - UNII:C4V974V932) INDOCYANINE GREEN 25 mg Inactive Ingredients Ingredient Name Strength SODIUM IODIDE (UNII: F5WR8N145C) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 1 in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA040811 01/01/2008 Part 2 of 2 STERILE WATER
water injectionProduct Information Item Code (Source) NDC:0409-4887 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength WATER (UNII: 059QF0KO0R) (WATER - UNII:059QF0KO0R) WATER 1 mL in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0409-4887-17 10 mL in 1 VIAL, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA018801 10/27/1982 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA040811 06/01/2016 Labeler - KARL STORZ Endoscopy-America, Inc. (075293423)