Label: WOMENS MINOXIDIL HAIR REGROWTH- minoxidil kit
- NDC Code(s): 51326-161-01, 51326-166-01
- Packager: Topiderm, Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated June 20, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Use
-
Warnings
Do not use if
- your amount of hair loss is different than that shown on the side of this carton, because this product may not work for you
- you have no family history of hair loss
- your hair loss is sudden and/or patchy
- your hair loss is associated with childbirth
- you do not know the reason for your hair loss
- you are under 18 years of age. Do not use on babies and children
- your scalp is red, inflamed, irritated or painful
- you use other medicines on the scalp
When using this product
- do not apply on other parts of the body
- avoid contact with the eyes. In case of accidental contact, rinse eyes with large amounts of cool tap water
- some people have experienced changes in hair color and/or texture
- it takes time to regrow hair. Results may occur at 2 months with twice a day usage. For some women, you may need to use this product for at least 4 months before you see results.
- the amount of hair regrowth is different for each person. This product will not work for all women.
- Directions
-
Other information
- see hair loss pictures on opposite side
- before use, read all information on carton and enclosed leaflet
- keep the insert. It contains important information.
- In clinical studies of mostly white women aged 18-45 years with mild to moderate degrees of hair loss, the following response to 2% minoxidil topical solution was reported: 19% of women reported moderate hair regrowth after using 2% minoxidil topical solution for 8 months (19% had moderate regrowth; 40% had minimal regrowth). This compares with 7% of women reporting moderate hair regrowth after using placebo, the liquid without minoxidil in it, for 8 months (7% had moderate regrowth; 33% had minimal regrowth).
- store at USP controlled room temperature 20° to 25°C (68° to 77°F)
- Inactive ingredients
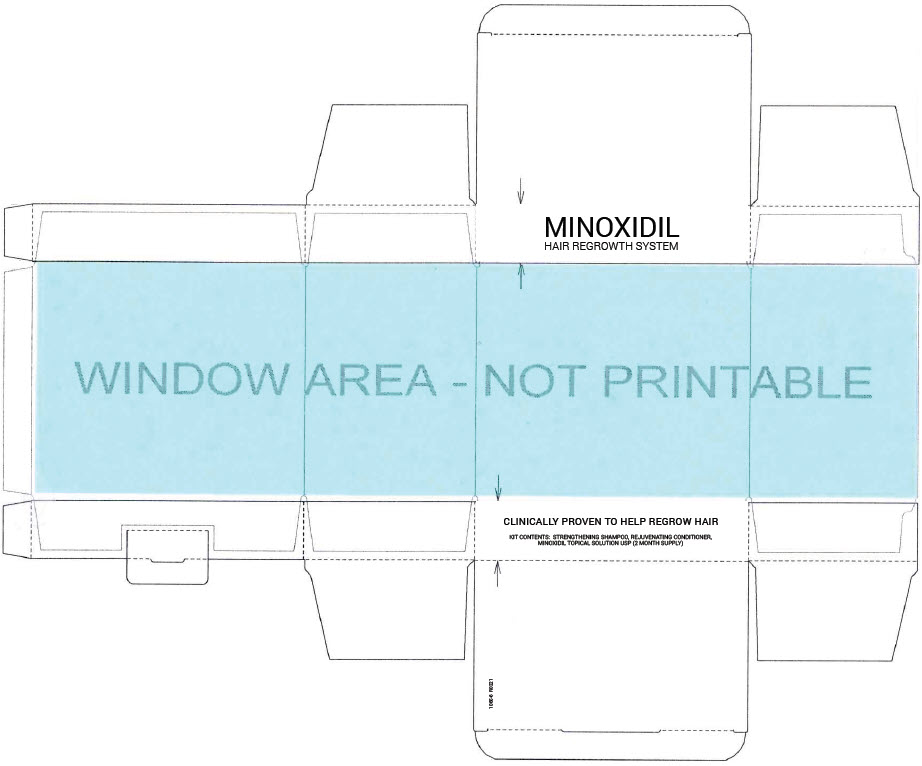
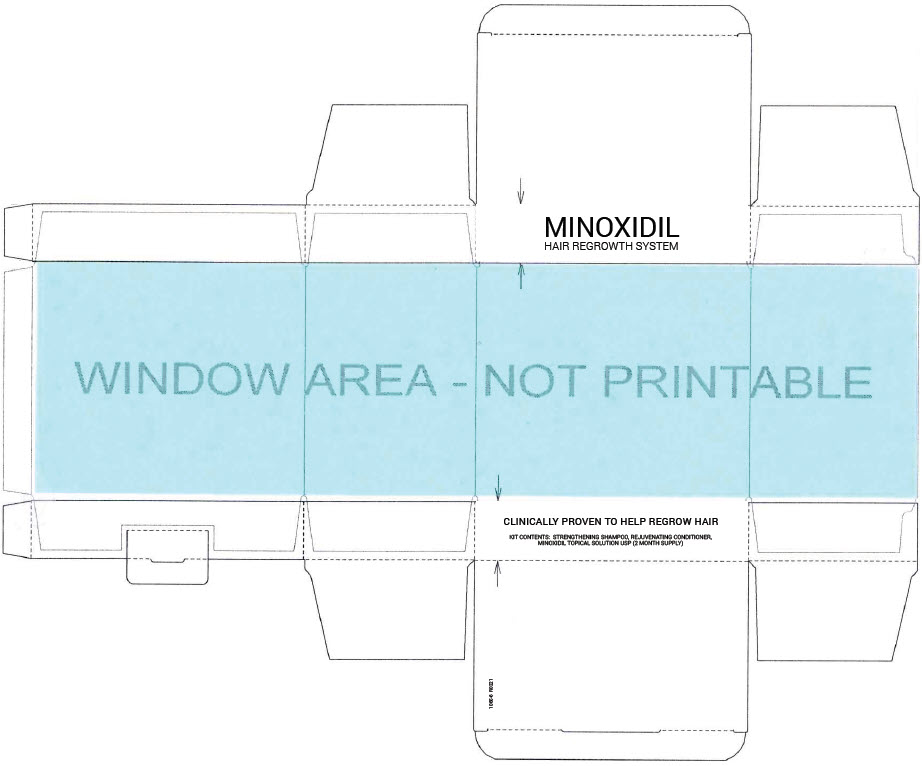
- PRINCIPAL DISPLAY PANEL - Kit Carton
-
PRINCIPAL DISPLAY PANEL - 2-60 mL Bottle Dropcard
WOMEN'S
MINOXIDIL
HAIR REGROWTH SYSTEM2% MINOXODIL TOPICAL SOLUTION, USP
Reactivates hair follicles to
stimulate regrowthClinically proven to help regrow hair
Topical Solution
Unscented
Minoxidil Topical Solution 2% is for women who have
a general thinning of hair on the top of the scalp
(vertex only, as shown below). Minoxidil Topical Solution
2% has been shown to regrow hair in women with the
following degrees of thinning hair of hair lossIf women have more hair loss than shown above,
Minoxidil Topi cal Solution 2% may not work.Do not use if you have patchy hair loss as shown above.
Two month supply
Two 60 ml (2 fl oz) bottles
- PRINCIPAL DISPLAY PANEL - 200 mL Bottle Label-Shampoo
- PRINCIPAL DISPLAY PANEL - 200 mL Bottle Label-Conditioner
-
INGREDIENTS AND APPEARANCE
WOMENS MINOXIDIL HAIR REGROWTH
minoxidil kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:51326-166 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51326-166-01 1 in 1 CARTON 06/16/2023 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 2 BOTTLE 120 mL Part 2 1 BOTTLE 200 mL Part 3 1 BOTTLE 200 mL Part 1 of 3 WOMENS MINOXIDIL HAIR GROWTH SYSTEM
minoxidil liquidProduct Information Item Code (Source) NDC:51326-161 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MINOXIDIL (UNII: 5965120SH1) (MINOXIDIL - UNII:5965120SH1) MINOXIDIL 20 mg in 1 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51326-161-01 60 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA078176 03/26/2021 Part 2 of 3 STRENGTHENING BASIL ROOT
shampoos (non-coloring) shampooProduct Information Route of Administration TOPICAL Other Ingredients Ingredient Kind Ingredient Name Quantity INGR WATER (UNII: 059QF0KO0R) INGR COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) INGR SODIUM LAUROYL SARCOSINATE (UNII: 632GS99618) INGR SODIUM COCOYL ISETHIONATE (UNII: 518XTE8493) INGR SODIUM LAURYL SULFOACETATE (UNII: D0Y70F2B9J) INGR GLYCERIN (UNII: PDC6A3C0OX) INGR PANTHENOL (UNII: WV9CM0O67Z) INGR PROPYLENE GLYCOL (UNII: 6DC9Q167V3) INGR .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) INGR DIOSCOREA VILLOSA TUBER (UNII: IWY3IWX2G8) INGR TRIFOLIUM PRATENSE FLOWER (UNII: 4JS0838828) INGR POTASSIUM SORBATE (UNII: 1VPU26JZZ4) INGR SPHINGANINE (UNII: YT0ZSD64HM) INGR CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) INGR ESCULIN (UNII: 1Y1L18LQAF) INGR GLYCOL DISTEARATE (UNII: 13W7MDN21W) INGR POLIDOCANOL (UNII: 0AWH8BFG9A) INGR C11-15 ALKETH-7 (UNII: 261HPE0IS3) INGR DIPROPYLENE GLYCOL (UNII: E107L85C40) INGR DECYL GLUCOSIDE (UNII: Z17H97EA6Y) INGR POLYQUATERNIUM-10 (400 MPA.S AT 2%) (UNII: HB1401PQFS) INGR POLYQUATERNIUM-7 (70/30 ACRYLAMIDE/DADMAC; 1600000 MW) (UNII: 0L414VCS5Y) INGR PEG-55 PROPYLENE GLYCOL OLEATE (UNII: 7RDE7PJS40) INGR MENTHONE 1,2-GLYCEROL KETAL, (+/-)- (UNII: 7QQ1EE6RCP) INGR TRIDECETH-12 (UNII: YFY3KG5Y7O) INGR ACETYL TETRAPEPTIDE-3 (UNII: D1HW9N9QBX) INGR BUTYLENE GLYCOL (UNII: 3XUS85K0RA) INGR BENZYL ALCOHOL (UNII: LKG8494WBH) INGR CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) INGR SODIUM BENZOATE (UNII: OJ245FE5EU) INGR SODIUM HYDROXIDE (UNII: 55X04QC32I) INGR BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) INGR EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) INGR PHENOXYETHANOL (UNII: HIE492ZZ3T) INGR BASIL (UNII: 2U0KZP0FDW) INGR LOQUAT (UNII: DM9CY56A36) INGR PEA (UNII: W4X7H8GYFM) INGR AMODIMETHICONE (1300 CST) (UNII: 3V7U636DWN) INGR DEXTRAN 110 (UNII: 05Q25F6XJ3) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 200 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date COSMETIC 03/26/2021 Part 3 of 3 REJUVENATING BASIL ROOT CONDITIONER
shampoos (non-coloring) shampooProduct Information Route of Administration TOPICAL Other Ingredients Ingredient Kind Ingredient Name Quantity INGR WATER (UNII: 059QF0KO0R) INGR STEARALKONIUM CHLORIDE (UNII: 0OUO26BB88) INGR CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) INGR STEARYL ALCOHOL (UNII: 2KR89I4H1Y) INGR TRIFOLIUM PRATENSE FLOWER (UNII: 4JS0838828) INGR POTASSIUM SORBATE (UNII: 1VPU26JZZ4) INGR SPHINGANINE (UNII: YT0ZSD64HM) INGR CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) INGR RESVERATROL (UNII: Q369O8926L) INGR SILIBININ (UNII: 4RKY41TBTF) INGR CAFFEINE (UNII: 3G6A5W338E) INGR ESCULIN (UNII: 1Y1L18LQAF) INGR POLYOXYL 40 HYDROGENATED CASTOR OIL (UNII: 7YC686GQ8F) INGR CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) INGR ACETYL TETRAPEPTIDE-3 (UNII: D1HW9N9QBX) INGR BUTYLENE GLYCOL (UNII: 3XUS85K0RA) INGR ZINC PIDOLATE (UNII: C32PQ86DH4) INGR COPPER PIDOLATE (UNII: 497G7G1SL1) INGR SODIUM CHLORIDE (UNII: 451W47IQ8X) INGR SODIUM BENZOATE (UNII: OJ245FE5EU) INGR EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) INGR PHENOXYETHANOL (UNII: HIE492ZZ3T) INGR BASIL (UNII: 2U0KZP0FDW) INGR LOQUAT (UNII: DM9CY56A36) INGR PEA (UNII: W4X7H8GYFM) INGR TEA LEAF OIL (UNII: VC855RRT77) INGR DEXTRAN 110 (UNII: 05Q25F6XJ3) INGR LECITHIN, SOYBEAN (UNII: 1DI56QDM62) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 200 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date COSMETIC 03/26/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA078176 06/16/2023 Labeler - Topiderm, Inc (049121643) Establishment Name Address ID/FEI Business Operations Topix Pharmaceuticals, Inc. 117745066 PACK(51326-166)