Label: RADIANCE SUPERBE DAY CREAM NUDE- ensulizole, octocrylene, titanium dioxide, zinc oxide cream
RADIANCE SUPERBE DAY CREAM NEUTRAL BRONZE- ensulizole, octocrylene, titanium dioxide, zinc oxide cream
RADIANCE SUPERBE DAY CREAM NEUTRAL LIGHT- ensulizole, octocrylene, titanium dioxide, zinc oxide cream
RADIANCE SUPERBE DAY CREAM TRANSLUCENT- ensulizole, octocrylene, titanium dioxide, zinc oxide cream
RADIANCE SUPERBE DAY CREAM NEUTRAL MEDIUM- ensulizole, octocrylene, titanium dioxide, zinc oxide cream
RADIANCE SUPERBE DAY CREAM NEUTRAL CARAMEL- ensulizole, octocrylene, titanium dioxide, zinc oxide cream
RADIANCE SUPERBE DAY CREAM NEUTRAL TAN- ensulizole, octocrylene, titanium dioxide, zinc oxide cream
-
NDC Code(s):
72414-000-15,
72414-000-50,
72414-001-15,
72414-001-50, view more72414-002-15, 72414-002-50, 72414-003-15, 72414-003-50, 72414-004-15, 72414-004-50, 72414-005-15, 72414-005-50, 72414-006-15, 72414-006-50
- Packager: Haute Custom Beauty
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated November 5, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
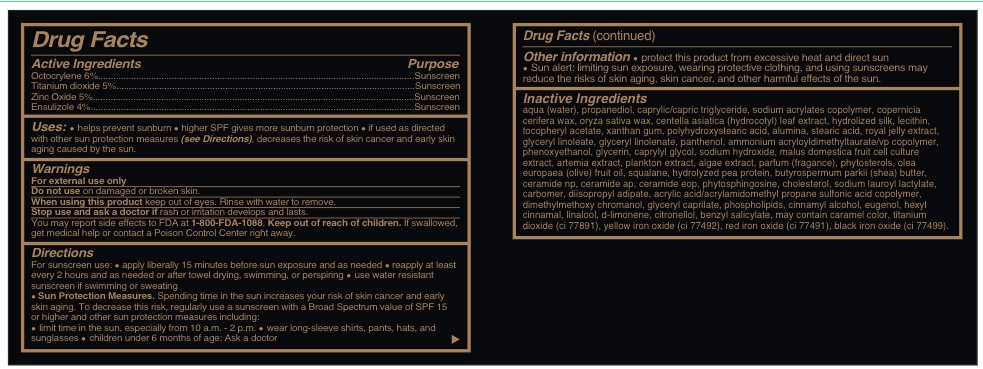
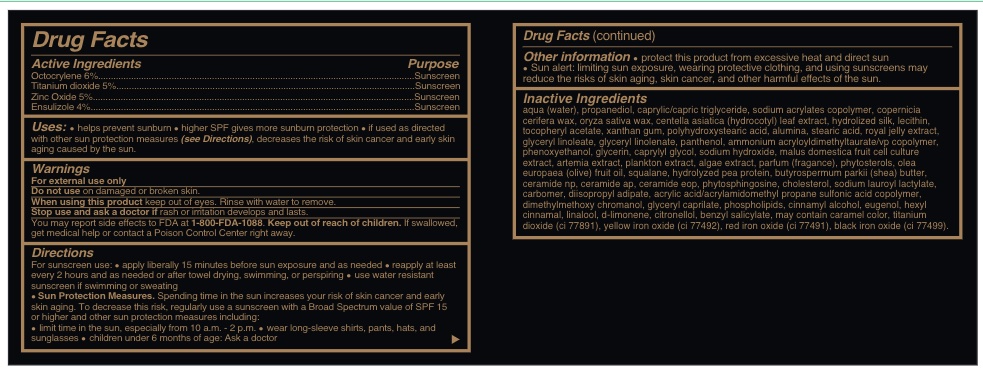
- Active Ingredients......sunscreen
-
Inactive Ingredients
AQUA (WATER), PROPANEDIOL, CAPRYLIC/CAPRIC TRIGLYCERIDE, SODIUM ACRYLATES COPOLYMER, COPERNICIA CERIFERA WAX, ORYZA SATIVA WAX, CENTELLA ASIATICA (HYDROCOTYL) LEAF EXTRACT, HYDROLIZED SILK, LECITHIN, TOCOPHERYL ACETATE, XANTHAN GUM, POLYHYDROXYSTEARIC ACID, ALUMINA, STEARIC ACID, ROYAL JELLY EXTRACT, GLYCERYL LINOLEATE, GLYCERYL LINOLENATE, PANTHENOL, AMMONIUM ACRYLOYLDIMETHYLTAURATE/VP COPOLYMER, PHENOXYETHANOL, GLYCERIN, CAPRYLYL GLYCOL, SODIUM HYDROXIDE, MALUS DOMESTICA FRUIT CELL CULTURE EXTRACT, ARTEMIA EXTRACT, PLANKTON EXTRACT, ALGAE EXTRACT, PARFUM (FRAGANCE), PHYTOSTEROLS, OLEA EUROPAEA (OLIVE) FRUIT OIL, SQUALANE, HYDROLYZED PEA PROTEIN, BUTYROSPERMUM PARKII (SHEA) BUTTER, CERAMIDE NP, CERAMIDE AP, CERAMIDE EOP, PHYTOSPHINGOSINE, CHOLESTEROL, SODIUM LAUROYL LACTYLATE, CARBOMER, DIISOPROPYL ADIPATE, ACRYLIC ACID/ACRYLAMIDOMETHYL PROPANE SULFONIC ACID COPOLYMER, DIMETHYLMETHOXY CHROMANOL, GLYCERYL CAPRILATE, PHOSPHOLIPIDS, CARAMEL, CI 77891, CI 77492, CI 77491 , CI 77499 , CINNAMYL ALCOHOL, EUGENOL, HEXYL CINNAMAL, LINALOOL, d-LIMONENE, CITRONELLOL, BENZYL SALICYLATE.
- Uses
-
Warnings
For external use only
Do not use on damaged or broken skin.
When using this product keep out of eyes. Rinse with water to remove.
Stop use and ask a doctor if rash or irratation develos and lasts.
You may report effects to FDA at 1-800-FDA-1088.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
- Keep out of reach of children
-
Directions
For suncreen use: *apply liberally 15 minutes before sun exposure and as needed * reapply at least every 2 hours and as needed or after towel drying, swimming, or perspiring. *. use water resistant sunscreen if swimming or sweating * Sun Protection measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum value of SPF 15 or higher and other sun protection measures including:
* limit time in th esun, especially from 10 a.m. - 2 p.m.
* wear long-sleeve shirts, pants, hats and sunglasses
*children under 6 months of age: Ask a doctor
- Other information
-
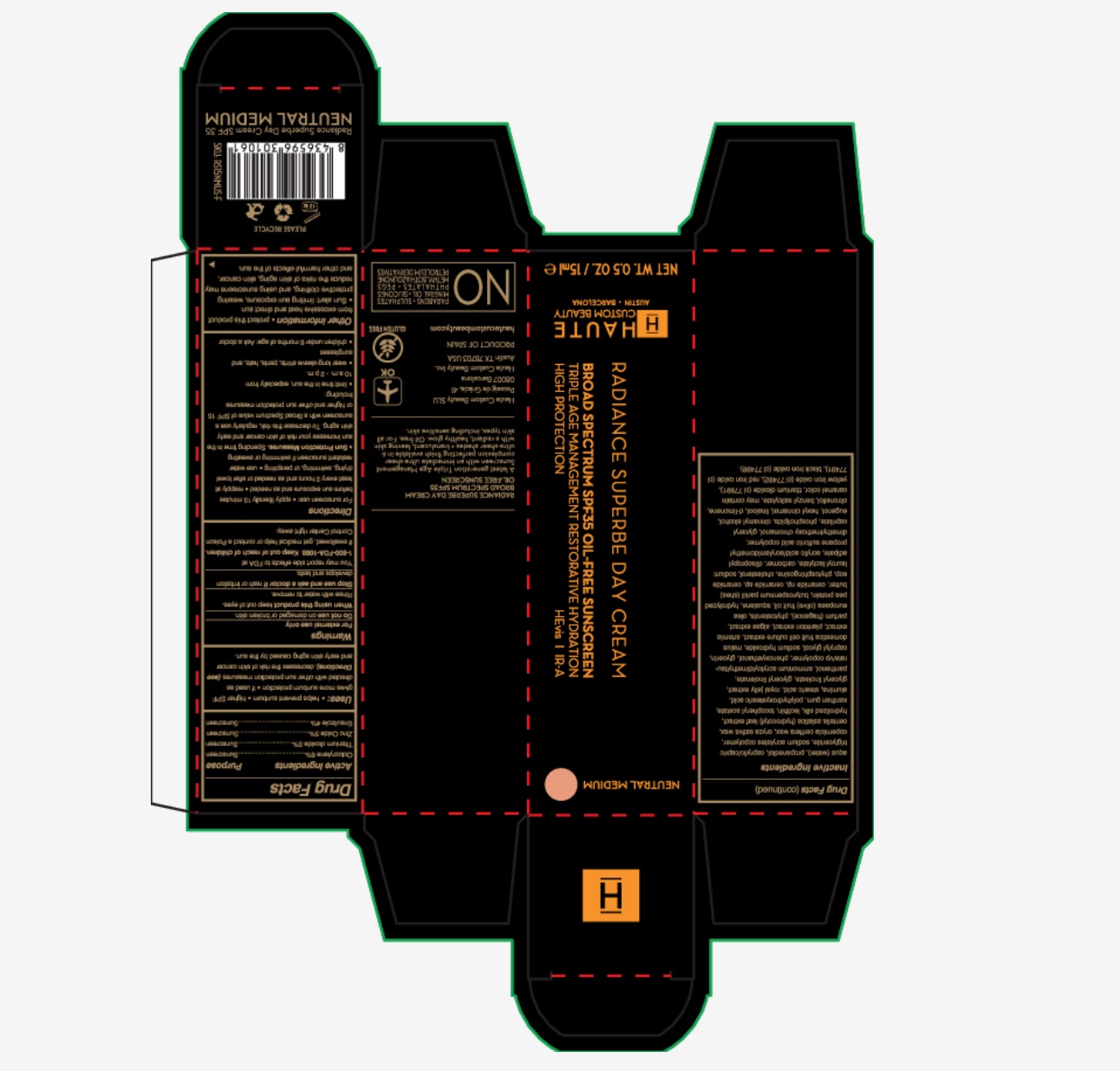
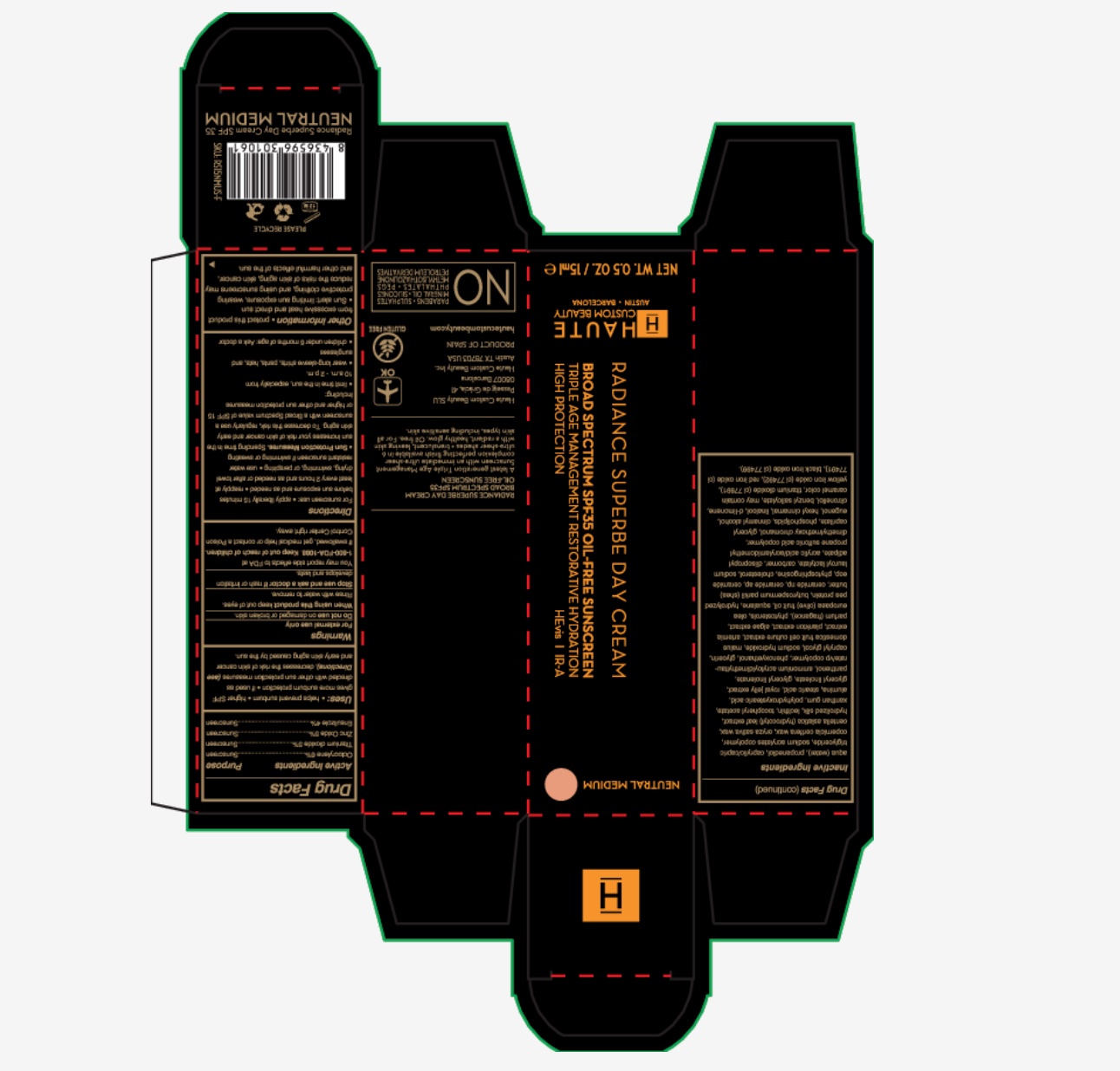
Description
RADIANCE SUPERBE DAY CREAM
BROAD SPECTRUM SPF 35
OIL-FREE SUNSCREEN
A latest generation Triple Age Management Sunscreen with an immediate ultra-sheer complexion perfecting finish availalbe in 6 ultra-sheer shades + translucent, leaving skin with a radiant, healthy glow. Oil free, for all skin types, including sensitive skin.
Haute Custom Beauty SLU
Passeig de Gracia 41
08047 Barcelona
Haute Custom Beauty INC
Austin, TX 78703 USA
PRODUCT OF SPAIN
www.hautecustombeauty.com
Airplane image OK
Gluten Free
No: parabens, sulphates, mineral oil, silicons, phthalates, PEGS, methysilothazzolinone, petroleum derivatives
- PRINCIPAL DISPLAY PANEL
- Radiance Superbe Day Cream Nude 72414-001
- Radiance Superbe Day Cream Neutral Light 72414-002
- Radiance Superbe Day Cream Neutral Medium 72414-003
- Radiance Superbe Day Cream Neutral Caramel 72414-004
- Radiance Superbe Day Cream Neutral Tan 72414-005
- Radiance Superbe Day Cream Neutral Bronze 72414-006
-
INGREDIENTS AND APPEARANCE
RADIANCE SUPERBE DAY CREAM NUDE
ensulizole, octocrylene, titanium dioxide, zinc oxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72414-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ENSULIZOLE (UNII: 9YQ9DI1W42) (ENSULIZOLE - UNII:9YQ9DI1W42) ENSULIZOLE 4 g in 100 g TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 5 g in 100 g OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 6 g in 100 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 5 g in 100 g Inactive Ingredients Ingredient Name Strength CERAMIDE AP (UNII: F1X8L2B00J) PHYTOSPHINGOSINE (UNII: GIN46U9Q2Q) GLYCERYL LINOLENATE (UNII: SGB6X4G86K) PEA PROTEIN (UNII: 7Q50F46595) PHENOXYETHANOL (UNII: HIE492ZZ3T) GLYCERIN (UNII: PDC6A3C0OX) CERAMIDE NP (UNII: 4370DF050B) CINNAMYL ALCOHOL (UNII: SS8YOP444F) BENZYL SALICYLATE (UNII: WAO5MNK9TU) OLEA EUROPAEA FRUIT VOLATILE OIL (UNII: 8E7358CX1J) MALUS DOMESTICA WHOLE (UNII: 04W636S1V3) ROYAL JELLY (UNII: L497I37F0C) PANTHENOL (UNII: WV9CM0O67Z) CAPRYLIC/CAPRIC/LAURIC TRIGLYCERIDE (UNII: FJ1H6M2JG9) DIISOPROPYL ADIPATE (UNII: P7E6YFV72X) LINALOOL, (+)- (UNII: F4VNO44C09) PROPANEDIOL (UNII: 5965N8W85T) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) AMMONIUM ACRYLOYLDIMETHYLTAURATE/VP COPOLYMER (UNII: W59H9296ZG) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CARNAUBA WAX (UNII: R12CBM0EIZ) CHOLESTEROL (UNII: 97C5T2UQ7J) CENTELLA ASIATICA LEAF (UNII: 6810070TYD) SILK, ENZYME HYDROLYZED (1000 MW) (UNII: 960MDR3Z07) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) XANTHAN GUM (UNII: TTV12P4NEE) STEARIC ACID (UNII: 4ELV7Z65AP) GLYCERYL LINOLEATE (UNII: 7B8K5VPO5S) SODIUM 2-ACRYLAMIDO-2-METHYL-1-PROPANE SULFONATE/SODIUM ACRYLATE/N-ISOPROPYLACRYLAMIDE/TRIMETHYL(3-METHACRYLAMIDOPROPYL)AMMONIUM CHLORIDE COPOLYMER (UNII: R9E4IU0HDO) DIMETHYLMETHOXY CHROMANOL (UNII: XBH432G01F) GLYCERYL CAPRYLATE (UNII: TM2TZD4G4A) EGG PHOSPHOLIPIDS (UNII: 1Z74184RGV) LIMONENE, (+/-)- (UNII: 9MC3I34447) .BETA.-CITRONELLOL, (+/-)- (UNII: 565OK72VNF) SODIUM HYDROXIDE (UNII: 55X04QC32I) SQUALANE (UNII: GW89575KF9) SHEA BUTTER (UNII: K49155WL9Y) CERAMIDE EOS (UNII: CR0J8RN66K) SODIUM LAUROYL LACTYLATE (UNII: 7243K85WFO) CARBOMER 980 (UNII: 4Q93RCW27E) EUGENOL (UNII: 3T8H1794QW) HEXYL CINNAMATE (UNII: 532UJ0Y6RI) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72414-001-50 50 g in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 12/28/2021 2 NDC:72414-001-15 15 g in 1 APPLICATOR; Type 0: Not a Combination Product 12/28/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 12/28/2021 12/28/2021 RADIANCE SUPERBE DAY CREAM NEUTRAL BRONZE
ensulizole, octocrylene, titanium dioxide, zinc oxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72414-006 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ENSULIZOLE (UNII: 9YQ9DI1W42) (ENSULIZOLE - UNII:9YQ9DI1W42) ENSULIZOLE 4 g in 100 g TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 5 g in 100 g OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 6 g in 100 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 5 g in 100 g Inactive Ingredients Ingredient Name Strength CERAMIDE AP (UNII: F1X8L2B00J) PHYTOSPHINGOSINE (UNII: GIN46U9Q2Q) GLYCERYL LINOLENATE (UNII: SGB6X4G86K) PEA PROTEIN (UNII: 7Q50F46595) PHENOXYETHANOL (UNII: HIE492ZZ3T) GLYCERIN (UNII: PDC6A3C0OX) CERAMIDE NP (UNII: 4370DF050B) CINNAMYL ALCOHOL (UNII: SS8YOP444F) BENZYL SALICYLATE (UNII: WAO5MNK9TU) OLEA EUROPAEA FRUIT VOLATILE OIL (UNII: 8E7358CX1J) MALUS DOMESTICA WHOLE (UNII: 04W636S1V3) ROYAL JELLY (UNII: L497I37F0C) PANTHENOL (UNII: WV9CM0O67Z) CAPRYLIC/CAPRIC/LAURIC TRIGLYCERIDE (UNII: FJ1H6M2JG9) DIISOPROPYL ADIPATE (UNII: P7E6YFV72X) LINALOOL, (+)- (UNII: F4VNO44C09) PROPANEDIOL (UNII: 5965N8W85T) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) AMMONIUM ACRYLOYLDIMETHYLTAURATE/VP COPOLYMER (UNII: W59H9296ZG) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CARNAUBA WAX (UNII: R12CBM0EIZ) CHOLESTEROL (UNII: 97C5T2UQ7J) CENTELLA ASIATICA LEAF (UNII: 6810070TYD) SILK, ENZYME HYDROLYZED (1000 MW) (UNII: 960MDR3Z07) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) XANTHAN GUM (UNII: TTV12P4NEE) STEARIC ACID (UNII: 4ELV7Z65AP) GLYCERYL LINOLEATE (UNII: 7B8K5VPO5S) SODIUM 2-ACRYLAMIDO-2-METHYL-1-PROPANE SULFONATE/SODIUM ACRYLATE/N-ISOPROPYLACRYLAMIDE/TRIMETHYL(3-METHACRYLAMIDOPROPYL)AMMONIUM CHLORIDE COPOLYMER (UNII: R9E4IU0HDO) DIMETHYLMETHOXY CHROMANOL (UNII: XBH432G01F) GLYCERYL CAPRYLATE (UNII: TM2TZD4G4A) EGG PHOSPHOLIPIDS (UNII: 1Z74184RGV) LIMONENE, (+/-)- (UNII: 9MC3I34447) .BETA.-CITRONELLOL, (+/-)- (UNII: 565OK72VNF) SODIUM HYDROXIDE (UNII: 55X04QC32I) SQUALANE (UNII: GW89575KF9) SHEA BUTTER (UNII: K49155WL9Y) CERAMIDE EOS (UNII: CR0J8RN66K) SODIUM LAUROYL LACTYLATE (UNII: 7243K85WFO) CARBOMER 980 (UNII: 4Q93RCW27E) EUGENOL (UNII: 3T8H1794QW) HEXYL CINNAMATE (UNII: 532UJ0Y6RI) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72414-006-50 50 g in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 07/01/2022 2 NDC:72414-006-15 15 g in 1 APPLICATOR; Type 0: Not a Combination Product 07/01/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 07/01/2022 RADIANCE SUPERBE DAY CREAM NEUTRAL LIGHT
ensulizole, octocrylene, titanium dioxide, zinc oxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72414-002 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ENSULIZOLE (UNII: 9YQ9DI1W42) (ENSULIZOLE - UNII:9YQ9DI1W42) ENSULIZOLE 4 g in 100 g TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 5 g in 100 g OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 6 g in 100 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 5 g in 100 g Inactive Ingredients Ingredient Name Strength POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) AMMONIUM ACRYLOYLDIMETHYLTAURATE/VP COPOLYMER (UNII: W59H9296ZG) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CARNAUBA WAX (UNII: R12CBM0EIZ) CHOLESTEROL (UNII: 97C5T2UQ7J) CERAMIDE AP (UNII: F1X8L2B00J) PHYTOSPHINGOSINE (UNII: GIN46U9Q2Q) GLYCERYL LINOLENATE (UNII: SGB6X4G86K) PEA PROTEIN (UNII: 7Q50F46595) PHENOXYETHANOL (UNII: HIE492ZZ3T) GLYCERIN (UNII: PDC6A3C0OX) CERAMIDE NP (UNII: 4370DF050B) CINNAMYL ALCOHOL (UNII: SS8YOP444F) BENZYL SALICYLATE (UNII: WAO5MNK9TU) OLEA EUROPAEA FRUIT VOLATILE OIL (UNII: 8E7358CX1J) MALUS DOMESTICA WHOLE (UNII: 04W636S1V3) ROYAL JELLY (UNII: L497I37F0C) PANTHENOL (UNII: WV9CM0O67Z) CAPRYLIC/CAPRIC/LAURIC TRIGLYCERIDE (UNII: FJ1H6M2JG9) DIISOPROPYL ADIPATE (UNII: P7E6YFV72X) LINALOOL, (+)- (UNII: F4VNO44C09) PROPANEDIOL (UNII: 5965N8W85T) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) CENTELLA ASIATICA LEAF (UNII: 6810070TYD) SILK, ENZYME HYDROLYZED (1000 MW) (UNII: 960MDR3Z07) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) XANTHAN GUM (UNII: TTV12P4NEE) STEARIC ACID (UNII: 4ELV7Z65AP) GLYCERYL LINOLEATE (UNII: 7B8K5VPO5S) SODIUM 2-ACRYLAMIDO-2-METHYL-1-PROPANE SULFONATE/SODIUM ACRYLATE/N-ISOPROPYLACRYLAMIDE/TRIMETHYL(3-METHACRYLAMIDOPROPYL)AMMONIUM CHLORIDE COPOLYMER (UNII: R9E4IU0HDO) DIMETHYLMETHOXY CHROMANOL (UNII: XBH432G01F) GLYCERYL CAPRYLATE (UNII: TM2TZD4G4A) EGG PHOSPHOLIPIDS (UNII: 1Z74184RGV) LIMONENE, (+/-)- (UNII: 9MC3I34447) .BETA.-CITRONELLOL, (+/-)- (UNII: 565OK72VNF) SODIUM HYDROXIDE (UNII: 55X04QC32I) SQUALANE (UNII: GW89575KF9) SHEA BUTTER (UNII: K49155WL9Y) CERAMIDE EOS (UNII: CR0J8RN66K) SODIUM LAUROYL LACTYLATE (UNII: 7243K85WFO) CARBOMER 980 (UNII: 4Q93RCW27E) EUGENOL (UNII: 3T8H1794QW) HEXYL CINNAMATE (UNII: 532UJ0Y6RI) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72414-002-50 50 g in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 07/01/2022 2 NDC:72414-002-15 15 g in 1 APPLICATOR; Type 0: Not a Combination Product 07/01/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 12/28/2021 RADIANCE SUPERBE DAY CREAM TRANSLUCENT
ensulizole, octocrylene, titanium dioxide, zinc oxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72414-000 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 5 g in 100 g ENSULIZOLE (UNII: 9YQ9DI1W42) (ENSULIZOLE - UNII:9YQ9DI1W42) ENSULIZOLE 4 g in 100 g TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 5 g in 100 g OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 6 g in 100 g Inactive Ingredients Ingredient Name Strength CERAMIDE AP (UNII: F1X8L2B00J) PHYTOSPHINGOSINE (UNII: GIN46U9Q2Q) GLYCERYL LINOLENATE (UNII: SGB6X4G86K) PEA PROTEIN (UNII: 7Q50F46595) PHENOXYETHANOL (UNII: HIE492ZZ3T) GLYCERIN (UNII: PDC6A3C0OX) CERAMIDE NP (UNII: 4370DF050B) CINNAMYL ALCOHOL (UNII: SS8YOP444F) BENZYL SALICYLATE (UNII: WAO5MNK9TU) POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) OLEA EUROPAEA FRUIT VOLATILE OIL (UNII: 8E7358CX1J) MALUS DOMESTICA WHOLE (UNII: 04W636S1V3) ROYAL JELLY (UNII: L497I37F0C) PANTHENOL (UNII: WV9CM0O67Z) CAPRYLIC/CAPRIC/LAURIC TRIGLYCERIDE (UNII: FJ1H6M2JG9) DIISOPROPYL ADIPATE (UNII: P7E6YFV72X) LINALOOL, (+)- (UNII: F4VNO44C09) PROPANEDIOL (UNII: 5965N8W85T) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) AMMONIUM ACRYLOYLDIMETHYLTAURATE/VP COPOLYMER (UNII: W59H9296ZG) CAPRYLYL GLYCOL (UNII: 00YIU5438U) GLYCERYL LINOLEATE (UNII: 7B8K5VPO5S) CARNAUBA WAX (UNII: R12CBM0EIZ) CHOLESTEROL (UNII: 97C5T2UQ7J) CENTELLA ASIATICA LEAF (UNII: 6810070TYD) SILK, ENZYME HYDROLYZED (1000 MW) (UNII: 960MDR3Z07) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) XANTHAN GUM (UNII: TTV12P4NEE) STEARIC ACID (UNII: 4ELV7Z65AP) SODIUM 2-ACRYLAMIDO-2-METHYL-1-PROPANE SULFONATE/SODIUM ACRYLATE/N-ISOPROPYLACRYLAMIDE/TRIMETHYL(3-METHACRYLAMIDOPROPYL)AMMONIUM CHLORIDE COPOLYMER (UNII: R9E4IU0HDO) DIMETHYLMETHOXY CHROMANOL (UNII: XBH432G01F) GLYCERYL CAPRYLATE (UNII: TM2TZD4G4A) EGG PHOSPHOLIPIDS (UNII: 1Z74184RGV) LIMONENE, (+/-)- (UNII: 9MC3I34447) .BETA.-CITRONELLOL, (+/-)- (UNII: 565OK72VNF) CERAMIDE EOS (UNII: CR0J8RN66K) SODIUM HYDROXIDE (UNII: 55X04QC32I) SQUALANE (UNII: GW89575KF9) SHEA BUTTER (UNII: K49155WL9Y) SODIUM LAUROYL LACTYLATE (UNII: 7243K85WFO) CARBOMER 980 (UNII: 4Q93RCW27E) EUGENOL (UNII: 3T8H1794QW) HEXYL CINNAMATE (UNII: 532UJ0Y6RI) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72414-000-50 50 g in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 07/01/2022 2 NDC:72414-000-15 15 g in 1 APPLICATOR; Type 0: Not a Combination Product 07/01/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 07/01/2022 RADIANCE SUPERBE DAY CREAM NEUTRAL MEDIUM
ensulizole, octocrylene, titanium dioxide, zinc oxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72414-003 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ENSULIZOLE (UNII: 9YQ9DI1W42) (ENSULIZOLE - UNII:9YQ9DI1W42) ENSULIZOLE 4 g in 100 g TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 5 g in 100 g OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 6 g in 100 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 5 g in 100 g Inactive Ingredients Ingredient Name Strength CERAMIDE AP (UNII: F1X8L2B00J) PHYTOSPHINGOSINE (UNII: GIN46U9Q2Q) GLYCERYL LINOLENATE (UNII: SGB6X4G86K) PEA PROTEIN (UNII: 7Q50F46595) PHENOXYETHANOL (UNII: HIE492ZZ3T) GLYCERIN (UNII: PDC6A3C0OX) CERAMIDE NP (UNII: 4370DF050B) CINNAMYL ALCOHOL (UNII: SS8YOP444F) BENZYL SALICYLATE (UNII: WAO5MNK9TU) OLEA EUROPAEA FRUIT VOLATILE OIL (UNII: 8E7358CX1J) MALUS DOMESTICA WHOLE (UNII: 04W636S1V3) ROYAL JELLY (UNII: L497I37F0C) PANTHENOL (UNII: WV9CM0O67Z) CAPRYLIC/CAPRIC/LAURIC TRIGLYCERIDE (UNII: FJ1H6M2JG9) DIISOPROPYL ADIPATE (UNII: P7E6YFV72X) LINALOOL, (+)- (UNII: F4VNO44C09) PROPANEDIOL (UNII: 5965N8W85T) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) AMMONIUM ACRYLOYLDIMETHYLTAURATE/VP COPOLYMER (UNII: W59H9296ZG) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CARNAUBA WAX (UNII: R12CBM0EIZ) CHOLESTEROL (UNII: 97C5T2UQ7J) CENTELLA ASIATICA LEAF (UNII: 6810070TYD) SILK, ENZYME HYDROLYZED (1000 MW) (UNII: 960MDR3Z07) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) XANTHAN GUM (UNII: TTV12P4NEE) STEARIC ACID (UNII: 4ELV7Z65AP) GLYCERYL LINOLEATE (UNII: 7B8K5VPO5S) SODIUM 2-ACRYLAMIDO-2-METHYL-1-PROPANE SULFONATE/SODIUM ACRYLATE/N-ISOPROPYLACRYLAMIDE/TRIMETHYL(3-METHACRYLAMIDOPROPYL)AMMONIUM CHLORIDE COPOLYMER (UNII: R9E4IU0HDO) DIMETHYLMETHOXY CHROMANOL (UNII: XBH432G01F) GLYCERYL CAPRYLATE (UNII: TM2TZD4G4A) EGG PHOSPHOLIPIDS (UNII: 1Z74184RGV) LIMONENE, (+/-)- (UNII: 9MC3I34447) .BETA.-CITRONELLOL, (+/-)- (UNII: 565OK72VNF) SODIUM HYDROXIDE (UNII: 55X04QC32I) SQUALANE (UNII: GW89575KF9) SHEA BUTTER (UNII: K49155WL9Y) CERAMIDE EOS (UNII: CR0J8RN66K) SODIUM LAUROYL LACTYLATE (UNII: 7243K85WFO) CARBOMER 980 (UNII: 4Q93RCW27E) EUGENOL (UNII: 3T8H1794QW) HEXYL CINNAMATE (UNII: 532UJ0Y6RI) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72414-003-50 50 g in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 07/01/2022 2 NDC:72414-003-15 15 g in 1 APPLICATOR; Type 0: Not a Combination Product 07/01/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 07/01/2022 RADIANCE SUPERBE DAY CREAM NEUTRAL CARAMEL
ensulizole, octocrylene, titanium dioxide, zinc oxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72414-004 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ENSULIZOLE (UNII: 9YQ9DI1W42) (ENSULIZOLE - UNII:9YQ9DI1W42) ENSULIZOLE 4 g in 100 g TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 5 g in 100 g OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 6 g in 100 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 5 g in 100 g Inactive Ingredients Ingredient Name Strength CERAMIDE AP (UNII: F1X8L2B00J) PHYTOSPHINGOSINE (UNII: GIN46U9Q2Q) GLYCERYL LINOLENATE (UNII: SGB6X4G86K) PEA PROTEIN (UNII: 7Q50F46595) PHENOXYETHANOL (UNII: HIE492ZZ3T) GLYCERIN (UNII: PDC6A3C0OX) CERAMIDE NP (UNII: 4370DF050B) CINNAMYL ALCOHOL (UNII: SS8YOP444F) BENZYL SALICYLATE (UNII: WAO5MNK9TU) OLEA EUROPAEA FRUIT VOLATILE OIL (UNII: 8E7358CX1J) MALUS DOMESTICA WHOLE (UNII: 04W636S1V3) ROYAL JELLY (UNII: L497I37F0C) PANTHENOL (UNII: WV9CM0O67Z) CAPRYLIC/CAPRIC/LAURIC TRIGLYCERIDE (UNII: FJ1H6M2JG9) DIISOPROPYL ADIPATE (UNII: P7E6YFV72X) LINALOOL, (+)- (UNII: F4VNO44C09) PROPANEDIOL (UNII: 5965N8W85T) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) AMMONIUM ACRYLOYLDIMETHYLTAURATE/VP COPOLYMER (UNII: W59H9296ZG) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CARNAUBA WAX (UNII: R12CBM0EIZ) CHOLESTEROL (UNII: 97C5T2UQ7J) CENTELLA ASIATICA LEAF (UNII: 6810070TYD) SILK, ENZYME HYDROLYZED (1000 MW) (UNII: 960MDR3Z07) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) XANTHAN GUM (UNII: TTV12P4NEE) STEARIC ACID (UNII: 4ELV7Z65AP) GLYCERYL LINOLEATE (UNII: 7B8K5VPO5S) SODIUM 2-ACRYLAMIDO-2-METHYL-1-PROPANE SULFONATE/SODIUM ACRYLATE/N-ISOPROPYLACRYLAMIDE/TRIMETHYL(3-METHACRYLAMIDOPROPYL)AMMONIUM CHLORIDE COPOLYMER (UNII: R9E4IU0HDO) DIMETHYLMETHOXY CHROMANOL (UNII: XBH432G01F) GLYCERYL CAPRYLATE (UNII: TM2TZD4G4A) EGG PHOSPHOLIPIDS (UNII: 1Z74184RGV) LIMONENE, (+/-)- (UNII: 9MC3I34447) .BETA.-CITRONELLOL, (+/-)- (UNII: 565OK72VNF) SODIUM HYDROXIDE (UNII: 55X04QC32I) SQUALANE (UNII: GW89575KF9) SHEA BUTTER (UNII: K49155WL9Y) CERAMIDE EOS (UNII: CR0J8RN66K) SODIUM LAUROYL LACTYLATE (UNII: 7243K85WFO) CARBOMER 980 (UNII: 4Q93RCW27E) EUGENOL (UNII: 3T8H1794QW) HEXYL CINNAMATE (UNII: 532UJ0Y6RI) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72414-004-50 50 g in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 07/01/2022 2 NDC:72414-004-15 15 g in 1 APPLICATOR; Type 0: Not a Combination Product 07/01/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 07/01/2022 RADIANCE SUPERBE DAY CREAM NEUTRAL TAN
ensulizole, octocrylene, titanium dioxide, zinc oxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72414-005 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ENSULIZOLE (UNII: 9YQ9DI1W42) (ENSULIZOLE - UNII:9YQ9DI1W42) ENSULIZOLE 4 g in 100 g TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 5 g in 100 g OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 6 g in 100 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 5 g in 100 g Inactive Ingredients Ingredient Name Strength CERAMIDE AP (UNII: F1X8L2B00J) PHYTOSPHINGOSINE (UNII: GIN46U9Q2Q) GLYCERYL LINOLENATE (UNII: SGB6X4G86K) PEA PROTEIN (UNII: 7Q50F46595) PHENOXYETHANOL (UNII: HIE492ZZ3T) GLYCERIN (UNII: PDC6A3C0OX) CERAMIDE NP (UNII: 4370DF050B) CINNAMYL ALCOHOL (UNII: SS8YOP444F) BENZYL SALICYLATE (UNII: WAO5MNK9TU) OLEA EUROPAEA FRUIT VOLATILE OIL (UNII: 8E7358CX1J) MALUS DOMESTICA WHOLE (UNII: 04W636S1V3) ROYAL JELLY (UNII: L497I37F0C) PANTHENOL (UNII: WV9CM0O67Z) CAPRYLIC/CAPRIC/LAURIC TRIGLYCERIDE (UNII: FJ1H6M2JG9) DIISOPROPYL ADIPATE (UNII: P7E6YFV72X) LINALOOL, (+)- (UNII: F4VNO44C09) PROPANEDIOL (UNII: 5965N8W85T) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) AMMONIUM ACRYLOYLDIMETHYLTAURATE/VP COPOLYMER (UNII: W59H9296ZG) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CARNAUBA WAX (UNII: R12CBM0EIZ) CHOLESTEROL (UNII: 97C5T2UQ7J) CENTELLA ASIATICA LEAF (UNII: 6810070TYD) SILK, ENZYME HYDROLYZED (1000 MW) (UNII: 960MDR3Z07) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) XANTHAN GUM (UNII: TTV12P4NEE) STEARIC ACID (UNII: 4ELV7Z65AP) GLYCERYL LINOLEATE (UNII: 7B8K5VPO5S) SODIUM 2-ACRYLAMIDO-2-METHYL-1-PROPANE SULFONATE/SODIUM ACRYLATE/N-ISOPROPYLACRYLAMIDE/TRIMETHYL(3-METHACRYLAMIDOPROPYL)AMMONIUM CHLORIDE COPOLYMER (UNII: R9E4IU0HDO) DIMETHYLMETHOXY CHROMANOL (UNII: XBH432G01F) GLYCERYL CAPRYLATE (UNII: TM2TZD4G4A) EGG PHOSPHOLIPIDS (UNII: 1Z74184RGV) LIMONENE, (+/-)- (UNII: 9MC3I34447) .BETA.-CITRONELLOL, (+/-)- (UNII: 565OK72VNF) SODIUM HYDROXIDE (UNII: 55X04QC32I) SQUALANE (UNII: GW89575KF9) SHEA BUTTER (UNII: K49155WL9Y) CERAMIDE EOS (UNII: CR0J8RN66K) SODIUM LAUROYL LACTYLATE (UNII: 7243K85WFO) CARBOMER 980 (UNII: 4Q93RCW27E) EUGENOL (UNII: 3T8H1794QW) HEXYL CINNAMATE (UNII: 532UJ0Y6RI) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72414-005-50 50 g in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 07/01/2022 2 NDC:72414-005-15 15 g in 1 APPLICATOR; Type 0: Not a Combination Product 07/01/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 07/01/2022 Labeler - Haute Custom Beauty (024442805) Registrant - Haute Custom Beauty (466493781) Establishment Name Address ID/FEI Business Operations Haute Custom Beauty SLU 466493781 manufacture(72414-000, 72414-001, 72414-002, 72414-003, 72414-004, 72414-005, 72414-006) , label(72414-000, 72414-001, 72414-002, 72414-003, 72414-004, 72414-005, 72414-006)