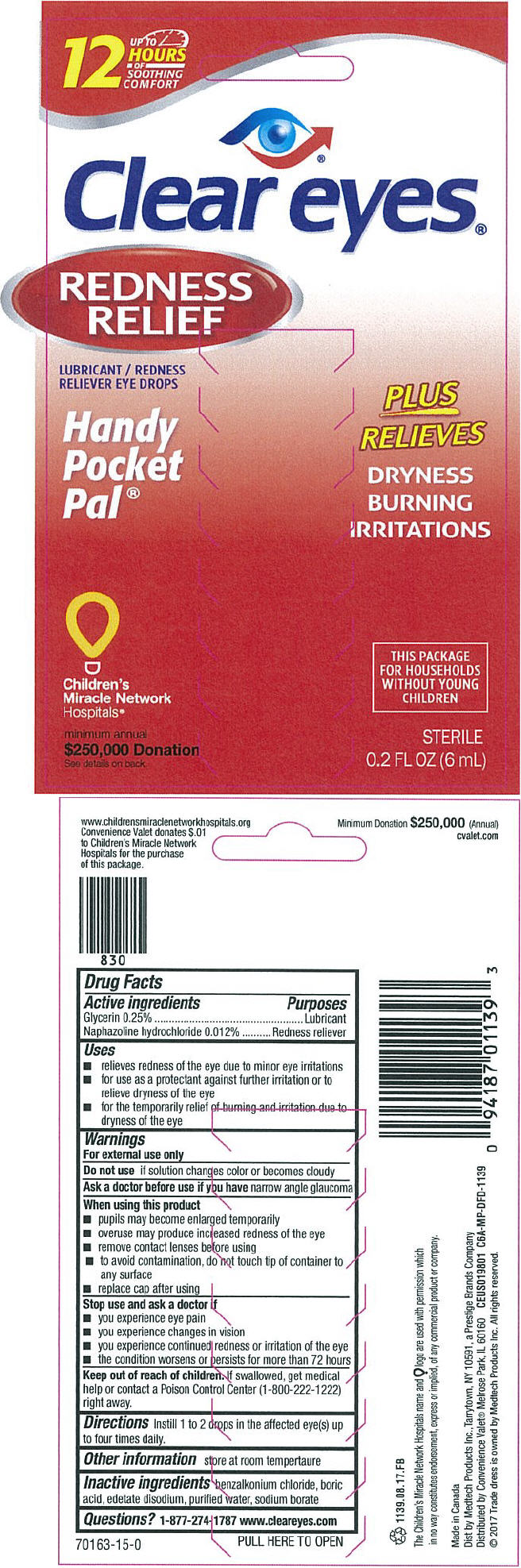
Label: CLEAR EYES REDNESS RELIEF HANDY POCKET PAL- naphazoline hydrochloride and glycerin solution/ drops
-
Contains inactivated NDC Code(s)
NDC Code(s): 29485-1139-1 - Packager: Mechanical Servants LLC
- This is a repackaged label.
- Source NDC Code(s): 67172-796
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated July 18, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- Uses
-
Warnings
For external use only
When using this product
- pupils may become enlarged temporarily
- overuse may produce increased redness of the eye
- remove contact lenses before using
- to avoid contamination, do not touch tip of container to any surface
- replace cap after using
- Directions
- Other information
- Inactive ingredients
- Questions?
- SPL UNCLASSIFIED SECTION
-
PRINCIPAL DISPLAY PANEL - 6 mL Bottle Box
UP TO
12
HOURS
OF
SOOTHING
COMFORTClear eyes®
REDNESS
RELIEFLUBRICANT / REDNESS
RELIEVER EYE DROPSHandy
Pocket
Pal®PLUS
RELIEVESDRYNESS
BURNING
IRRITATIONSChildren's
Miracle Network
Hospitals®minimum annual
$250,000 Donation
See details on backTHIS PACKAGE
FOR HOUSEHOLDS
WITHOUT YOUNG
CHILDRENSTERILE
0.2 FL OZ (6 mL)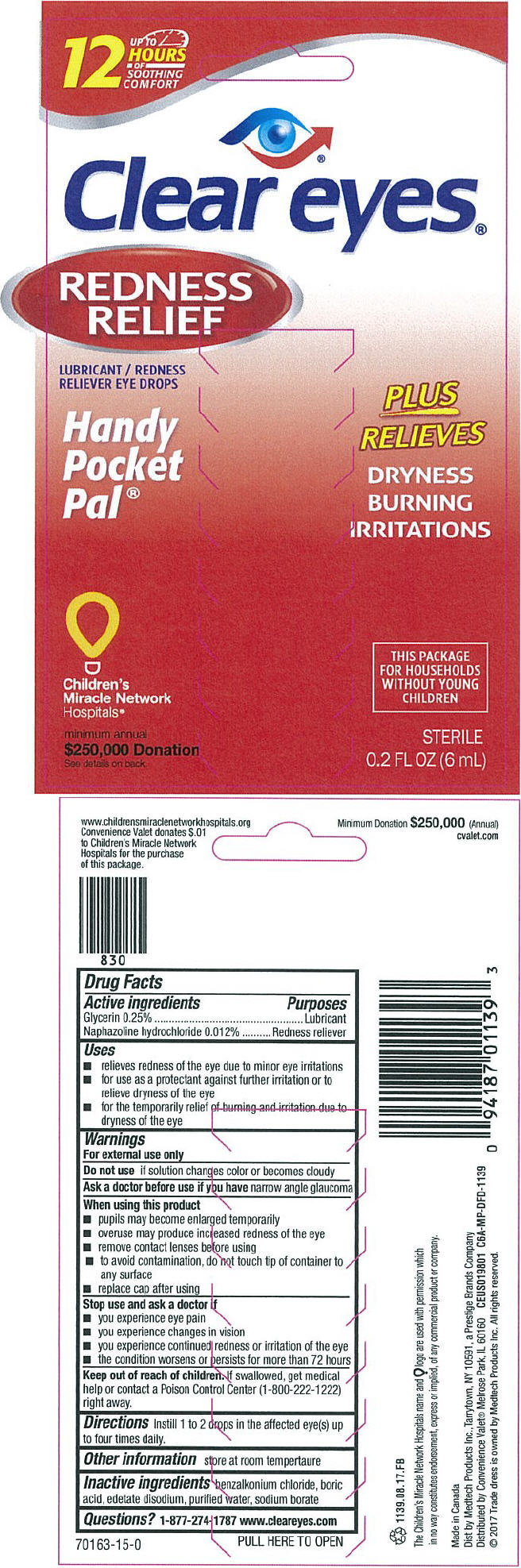
-
INGREDIENTS AND APPEARANCE
CLEAR EYES REDNESS RELIEF HANDY POCKET PAL
naphazoline hydrochloride and glycerin solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:29485-1139(NDC:67172-796) Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NAPHAZOLINE HYDROCHLORIDE (UNII: MZ1131787D) (NAPHAZOLINE - UNII:H231GF11BV) NAPHAZOLINE HYDROCHLORIDE 0.00012 mg in 1 mL GLYCERIN (UNII: PDC6A3C0OX) (GLYCERIN - UNII:PDC6A3C0OX) GLYCERIN 0.0025 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) BORIC ACID (UNII: R57ZHV85D4) EDETATE DISODIUM (UNII: 7FLD91C86K) WATER (UNII: 059QF0KO0R) SODIUM BORATE (UNII: 91MBZ8H3QO) Product Characteristics Color WHITE Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:29485-1139-1 1 in 1 BOX 11/01/2012 1 6 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part349 11/01/2012 Labeler - Mechanical Servants LLC (005530951) Establishment Name Address ID/FEI Business Operations Mechanical Servants LLC 005530951 REPACK(29485-1139) , RELABEL(29485-1139)