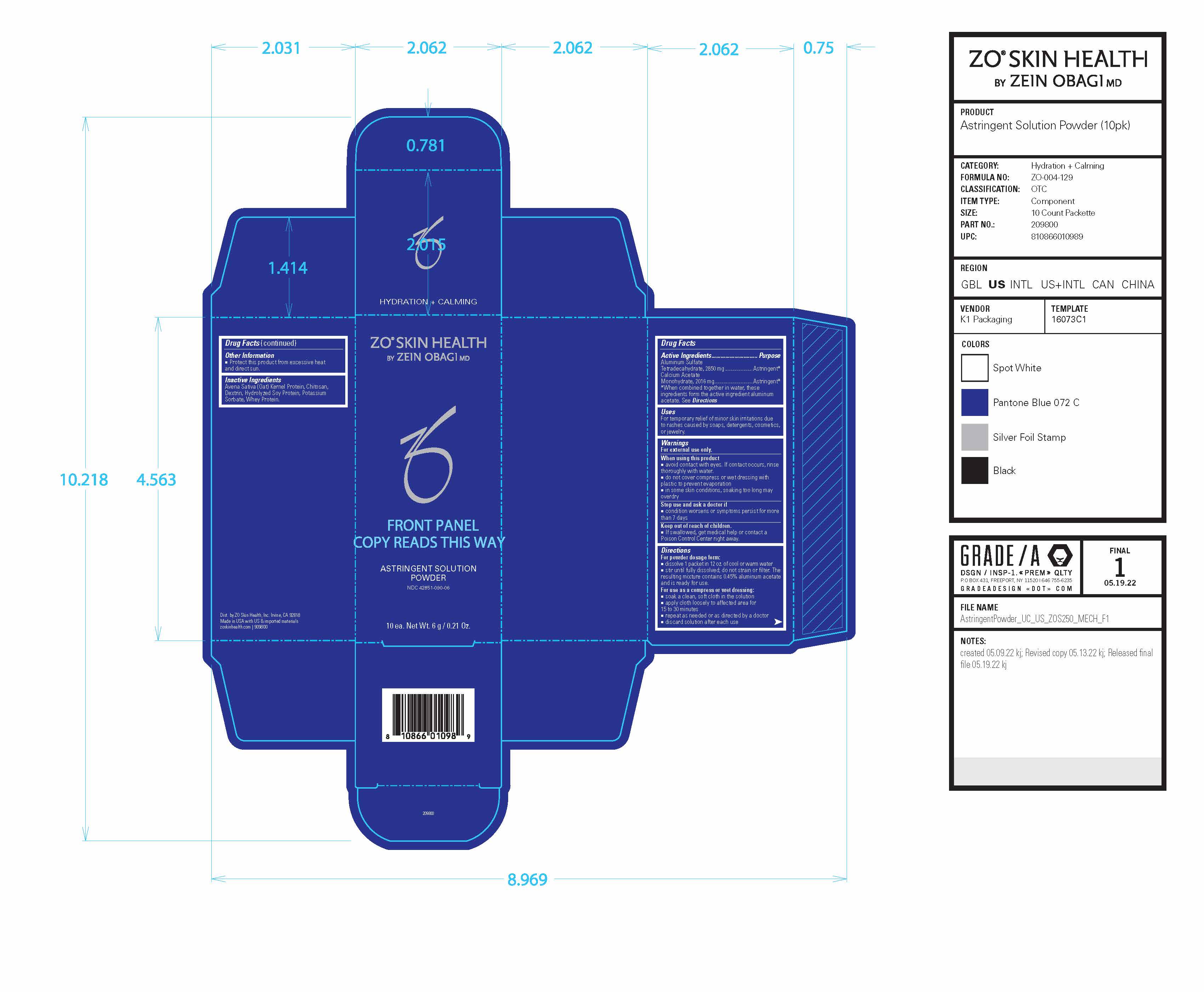
Label: ZO SKIN HEALTH ASTRINGENT SOLUTION- aluminum acetate powder
- NDC Code(s): 42851-090-06
- Packager: ZO Skin Health, Inc.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 5, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
Drug Facts
Active Ingredients Purpose Aluminum Sulfate Tetradecahydrate, 2850 mg Astringent* Calcium AcetateMonohydrate, 2016 mg Astringent* *When combined together in water, these ingredients form the active ingredient aluminum acetate. See Directions
Uses
For temporary relief of minor skin irritations due to rashes caused by soaps, detergents, cosmetics, or jewelry.
Warnings
For external use only.
Directions
For powder dosage form:
■ dissolve 1 packet in 12 oz. of cool or warm water
■ stir until fully dissolved; do not strain or filter. The resulting mixture contains 0.45% aluminum acetate and is ready for use.
For use as a compress or wet dressing:
■ soak a clean, soft cloth in the solution
■ apply cloth loosely to affected area for
15 to 30 minutes
■ repeat as needed or as directed by a doctor
■ discard solution after each use - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ZO SKIN HEALTH ASTRINGENT SOLUTION
aluminum acetate powderProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:42851-090 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALUMINUM ACETATE (UNII: 80EHD8I43D) (ALUMINUM CATION - UNII:3XHB1D032B) ALUMINUM ACETATE 4866 mg in 6 g Inactive Ingredients Ingredient Name Strength WHEY (UNII: 8617Z5FMF6) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) HYDROLYZED SOY PROTEIN (ENZYMATIC; 2000 MW) (UNII: 1394NXB9L6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:42851-090-06 6 g in 1 PACKET; Type 0: Not a Combination Product 02/21/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 02/21/2022 Labeler - ZO Skin Health, Inc. (826468527)