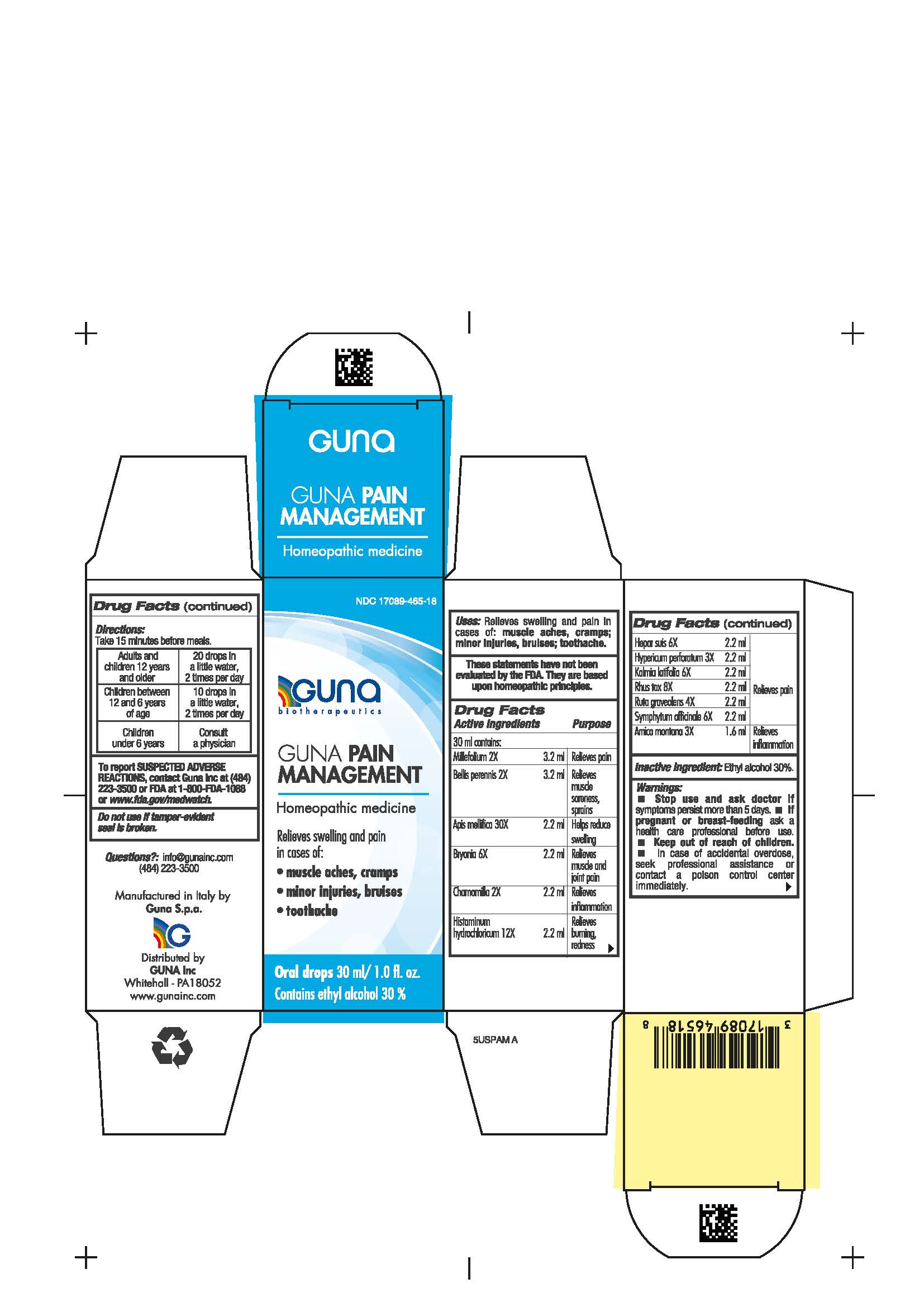
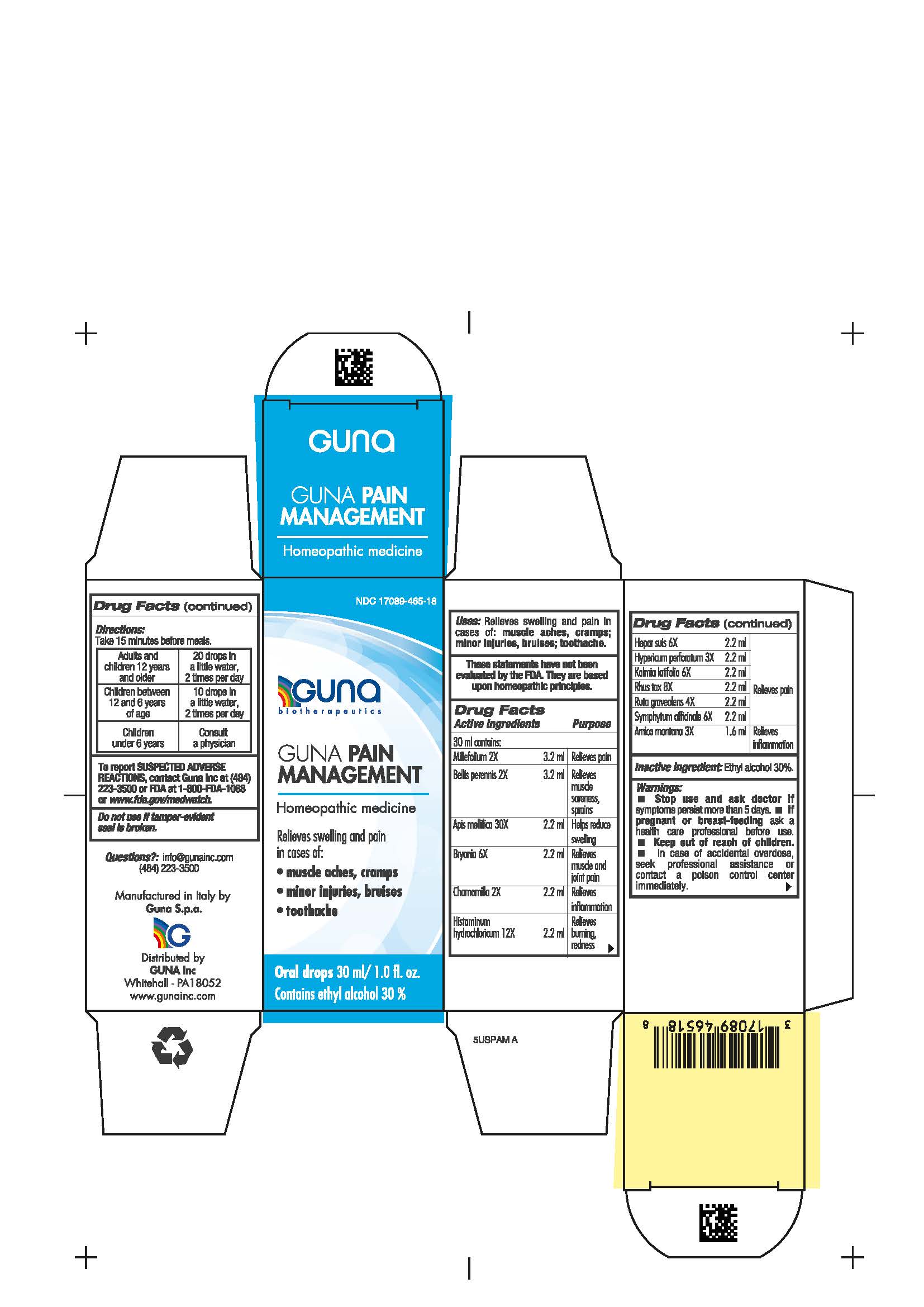
Label: GUNA PAIN MANAGEMENT- achillea millefolium whole - bellis perennis whole - matricaria chamomilla whole - toxicodendron pubescens whole - ruta graveolens flowering top - comfrey root - apis mellifera - bryonia alba root - histamine hydrochloride - hypericum perforatum - kalmia latifolia leaf - arnica montana whole - pork liver - solution/ drops
- NDC Code(s): 17089-465-18
- Packager: Guna spa
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated September 15, 2020
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- INACTIVE INGREDIENT
- QUESTIONS
- WARNINGS
- INDICATIONS & USAGE
- DIRECTIONS
- PREGNANCY
-
WARNINGS
- Stop use and ask doctor if symptoms worsen or persist more than 5 days.
- If pregnant or breast-feeding ask a health care professional before use.
- Keep out of reach of children.
- In case of accidental overdose, seek professional assistance or contact a poison control center immediately
- Contains ethyl alcohol 30%
-
ACTIVE INGREDIENTS/PURPOSE
Millefolium 2X Relieves pain
Bellis perennis 2X Relieves muscle soreness, sprains
Apis mellifica 30X Helps reduce swelling
Bryonia 6X Relieves muscle and joint pain
Chamomilla 2X Relieves inflammation
Histaminum hydroc. 12X Relieves burning, redness
Hepar suis 6X Relieves pain
Hypericum perf. 3X Relieves pain
Kalmia latifolia 6X Relieves pain
Rhus tox 8X Relieves pain
Ruta graveolens 4X Relieves pain
Symphytum officinale 6X Relieves pain
Arnica montana 3X Relieves inflammation
- USES
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
GUNA PAIN MANAGEMENT
achillea millefolium whole - bellis perennis whole - matricaria chamomilla whole - toxicodendron pubescens whole - ruta graveolens flowering top - comfrey root - apis mellifera - bryonia alba root - histamine hydrochloride - hypericum perforatum - kalmia latifolia leaf - arnica montana whole - pork liver - solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:17089-465 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BRYONIA ALBA ROOT (UNII: T7J046YI2B) (BRYONIA ALBA ROOT - UNII:T7J046YI2B) BRYONIA ALBA ROOT 6 [hp_X] in 30 mL HISTAMINE DIHYDROCHLORIDE (UNII: 3POA0Q644U) (HISTAMINE - UNII:820484N8I3) HISTAMINE DIHYDROCHLORIDE 12 [hp_X] in 30 mL HYPERICUM PERFORATUM (UNII: XK4IUX8MNB) (HYPERICUM PERFORATUM - UNII:XK4IUX8MNB) HYPERICUM PERFORATUM 3 [hp_X] in 30 mL ARNICA MONTANA WHOLE (UNII: O80TY208ZW) (ARNICA MONTANA WHOLE - UNII:O80TY208ZW) ARNICA MONTANA WHOLE 3 [hp_X] in 30 mL PORK LIVER (UNII: 6EC706HI7F) (PORK LIVER - UNII:6EC706HI7F) PORK LIVER 6 [hp_X] in 30 mL BELLIS PERENNIS WHOLE (UNII: 2HU33I03UY) (BELLIS PERENNIS WHOLE - UNII:2HU33I03UY) BELLIS PERENNIS WHOLE 2 [hp_X] in 30 mL MATRICARIA CHAMOMILLA WHOLE (UNII: G0R4UBI2ZZ) (MATRICARIA CHAMOMILLA WHOLE - UNII:G0R4UBI2ZZ) MATRICARIA CHAMOMILLA WHOLE 2 [hp_X] in 30 mL TOXICODENDRON PUBESCENS LEAF (UNII: 6IO182RP7A) (TOXICODENDRON PUBESCENS LEAF - UNII:6IO182RP7A) TOXICODENDRON PUBESCENS LEAF 8 [hp_X] in 30 mL RUTA GRAVEOLENS FLOWERING TOP (UNII: N94C2U587S) (RUTA GRAVEOLENS FLOWERING TOP - UNII:N94C2U587S) RUTA GRAVEOLENS FLOWERING TOP 4 [hp_X] in 30 mL KALMIA LATIFOLIA LEAF (UNII: 79N6542N18) (KALMIA LATIFOLIA LEAF - UNII:79N6542N18) KALMIA LATIFOLIA LEAF 6 [hp_X] in 30 mL ACHILLEA MILLEFOLIUM WHOLE (UNII: 2FXJ6SW4PK) (ACHILLEA MILLEFOLIUM WHOLE - UNII:2FXJ6SW4PK) ACHILLEA MILLEFOLIUM WHOLE 2 [hp_X] in 30 mL COMFREY ROOT (UNII: M9VVZ08EKQ) (COMFREY ROOT - UNII:M9VVZ08EKQ) COMFREY ROOT 6 [hp_X] in 30 mL APIS MELLIFERA (UNII: 7S82P3R43Z) (APIS MELLIFERA - UNII:7S82P3R43Z) APIS MELLIFERA 30 [hp_X] in 30 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) 9 mL in 30 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:17089-465-18 1 in 1 BOX 09/24/2020 1 30 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 09/24/2020 Labeler - Guna spa (430538264) Establishment Name Address ID/FEI Business Operations Guna spa 338587646 manufacture(17089-465)