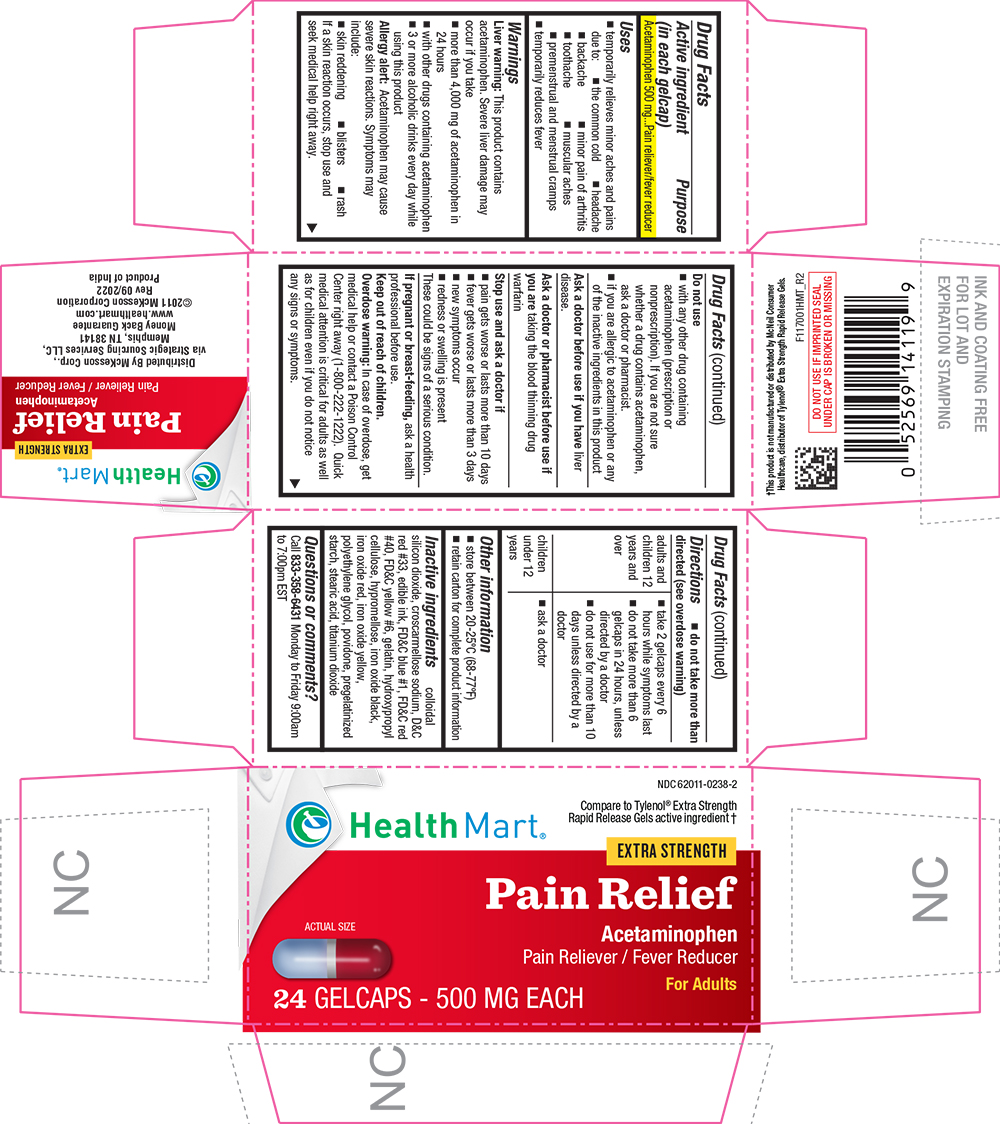
Label: PAIN RELIEVER RAPID RELEASE- acetaminophen tablet, coated
- NDC Code(s): 62011-0238-1, 62011-0238-2
- Packager: Strategic Sourcing Services LLC
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated October 30, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient (in each gelcap)
- Purpose
- Uses
-
Warnings
Liver warning
This product contains acetaminophen. Severe liver damage may occur if you take
- more than 4,000 mg of acetaminophen in 24 hours
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert
Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are allergic to acetaminophen or any of the inactive ingredients in this product
-
Directions
- do not take more than directed (see overdose warning)
adults and children 12 years and over - take 2 gelcaps every 6 hours while symptoms last
- do not take more than 6 gelcaps in 24 hours, unless directed by a doctor
- do not use for more than 10 days unless directed by a doctor
children under 12 years - ask a doctor
- Other information
-
Inactive ingredients
colloidal silicon dioxide, croscarmellose sodium, D&C red #33, edible ink, FD&C blue #1, FD&C red #40, FD&C yellow #6, gelatin, hydroxypropyl cellulose, hypromellose, iron oxide black, iron oxide red, iron oxide yellow, polyethylene glycol, povidone, pregelatinized starch, stearic acid, titanium dioxide
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
PAIN RELIEVER RAPID RELEASE
acetaminophen tablet, coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:62011-0238 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 500 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) D&C RED NO. 33 (UNII: 9DBA0SBB0L) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 40 (UNII: WZB9127XOA) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) GELATIN (UNII: 2G86QN327L) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) HYPROMELLOSES (UNII: 3NXW29V3WO) FERROSOFERRIC OXIDE (UNII: XM0M87F357) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) STARCH, CORN (UNII: O8232NY3SJ) STEARIC ACID (UNII: 4ELV7Z65AP) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color gray (red and light blue ends) Score no score Shape OVAL Size 19mm Flavor Imprint Code G1 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62011-0238-1 1 in 1 CARTON 10/24/2014 09/30/2024 1 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 2 NDC:62011-0238-2 1 in 1 CARTON 03/01/2018 01/31/2026 2 24 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M013 10/24/2014 01/31/2026 Labeler - Strategic Sourcing Services LLC (116956644)