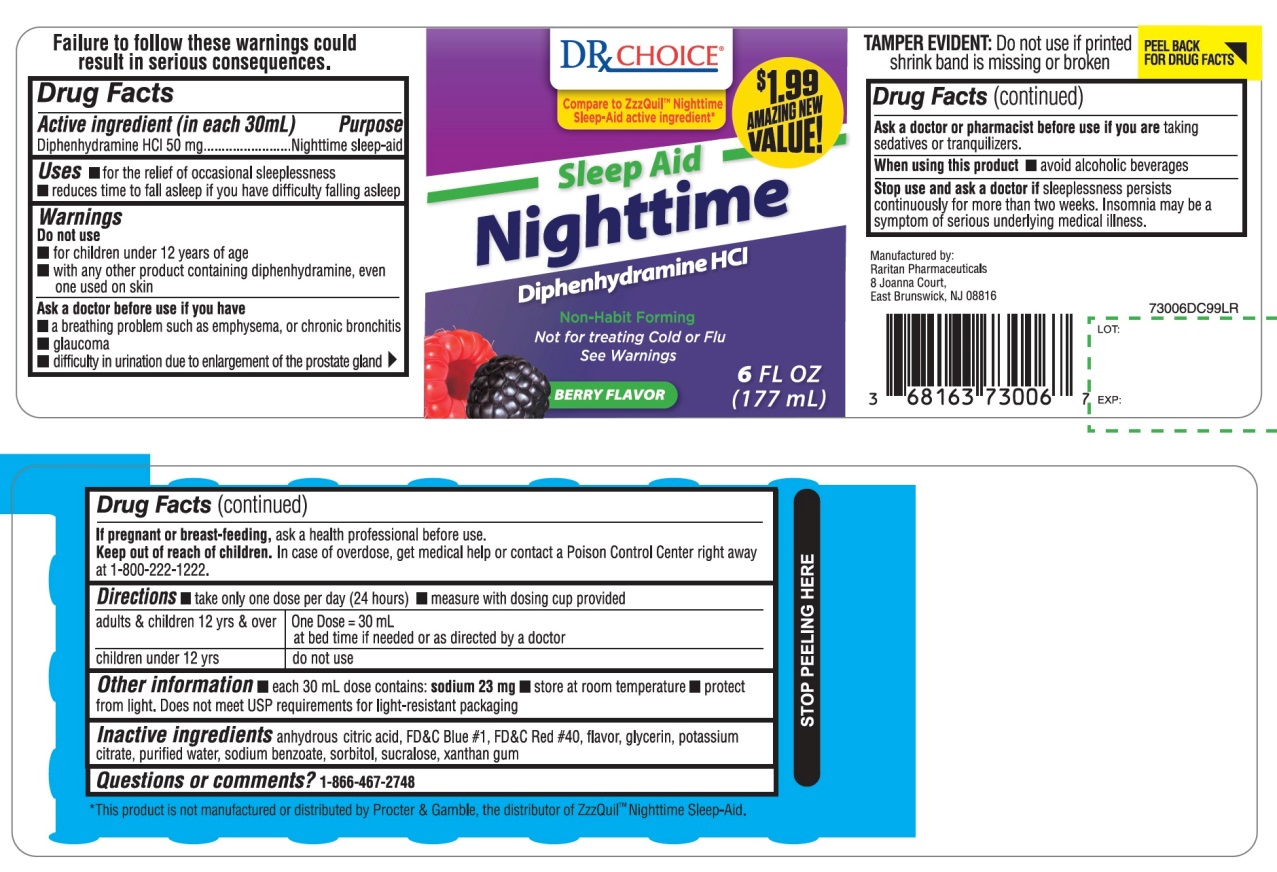
Label: NIGHTTIME SLEEP AID BERRY FLAVOR- diphenhydramine hydrochloride liquid
- NDC Code(s): 68163-730-06
- Packager: Raritan Pharmaceuticals Inc
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated September 2, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient (in each 30 mL)
- Purpose
- Uses
-
Warnings
Do not use
- ▪
- for children under 12 years of age
- ▪
- with any other product containing diphenhydramine, even one used on skin
Ask a doctor before use if you have
- ▪
- a breathing problem such as emphysema, or chronic bronchitis
- ▪
- glaucoma
- ▪
- difficulty in urination due to enlargement of the prostate gland
- Directions
- Other information
- Inactive ingredients
-
PRINCIPAL DISPLAY PANEL
DRx CHOICE®
NDC 68163-730-06
Compare to in ZzzQuil® Nighttime Sleep-Aid active ingredient *
Sleep-Aid
Nighttime
Diphenhydramine HCl
Non-Habit Forming
BERRY FLAVOR
Naturally and Artificially Flavored
Not for treating Cold or Flu See Warnings
6 FL OZ (177 mL)
$1.99 AMAZING NEW VALUE!
TAMPER EVIDENT: DO NOT USE IF PRINTED SHRINK BAND IS MISSING OR BROKEN
*This product is not manufactured or distributed by Procter & Gamble, the distributor of ZzzQuil™ Nighttime Sleep-Aid.
Manufactured by:
Raritan Pharmaceuticals
8 Joanna Court,
East Brunswick, NJ 08816
-
INGREDIENTS AND APPEARANCE
NIGHTTIME SLEEP AID BERRY FLAVOR
diphenhydramine hydrochloride liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68163-730 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIPHENHYDRAMINE HYDROCHLORIDE (UNII: TC2D6JAD40) (DIPHENHYDRAMINE - UNII:8GTS82S83M) DIPHENHYDRAMINE HYDROCHLORIDE 50 mg in 30 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 40 (UNII: WZB9127XOA) GLYCERIN (UNII: PDC6A3C0OX) POTASSIUM CITRATE (UNII: EE90ONI6FF) WATER (UNII: 059QF0KO0R) SODIUM BENZOATE (UNII: OJ245FE5EU) SORBITOL (UNII: 506T60A25R) SUCRALOSE (UNII: 96K6UQ3ZD4) XANTHAN GUM (UNII: TTV12P4NEE) Product Characteristics Color PURPLE Score Shape Size Flavor BERRY (Naturally and Artificially) Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68163-730-06 177 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/17/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 04/17/2019 Labeler - Raritan Pharmaceuticals Inc (127602287)