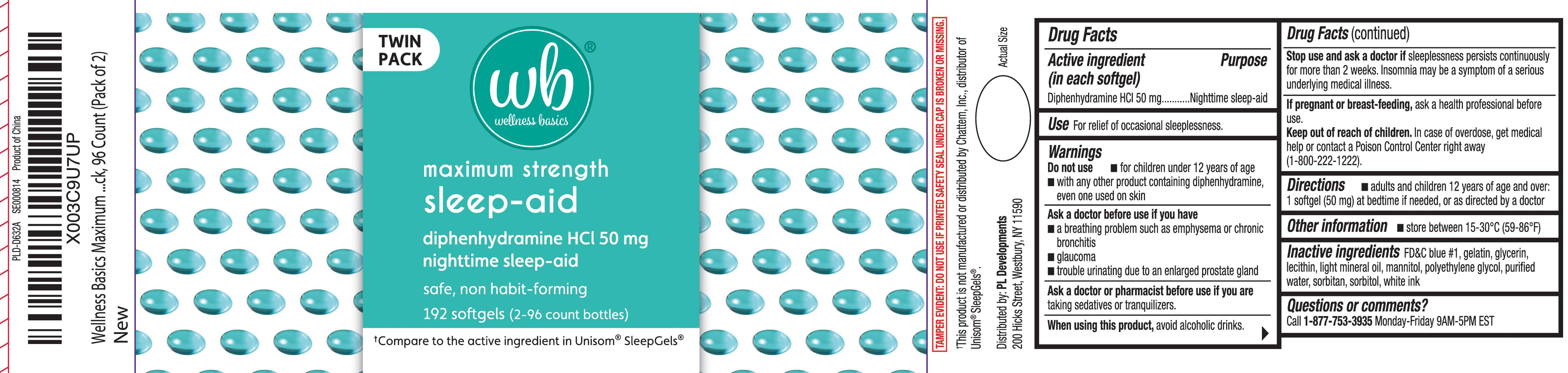
Label: SLEEP AID MAXIMUM STRENGTH- diphenhydramine hcl capsule
- NDC Code(s): 59726-816-29, 59726-816-64, 59726-816-92
- Packager: P & L Development, LLC
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated October 23, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each softgel)
- Purpose
- Use
-
Warnings
Do not use
- for children under 12 years of age
- with any other product containing diphenhydramine, even one used on skin
Ask a doctor before use if you have
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- trouble urinating due to an enlarged prostate gland
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
Maximum Strength
Sleep-Aid
diphenhydramine HCl 50 mg
nighttime sleep-aid
safe, non-habit forming
softgels
One Softgel Per Dose
†Compare to the active ingredient in Unisom® SleepGels®
†This product is not manufactured or distributed by Chattem, Inc., distributor of Unisom® SleepGels®.
TAMPER EVIDENT: DO NOT USE IF PRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING.
Distributed by: PL Developments
200 Hicks Street
Westbury, NY 11590
- Package Label
-
INGREDIENTS AND APPEARANCE
SLEEP AID MAXIMUM STRENGTH
diphenhydramine hcl capsuleProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:59726-816 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIPHENHYDRAMINE HYDROCHLORIDE (UNII: TC2D6JAD40) (DIPHENHYDRAMINE - UNII:8GTS82S83M) DIPHENHYDRAMINE HYDROCHLORIDE 50 mg Inactive Ingredients Ingredient Name Strength FD&C BLUE NO. 1 (UNII: H3R47K3TBD) GELATIN (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) WATER (UNII: 059QF0KO0R) SORBITAN (UNII: 6O92ICV9RU) SORBITOL (UNII: 506T60A25R) LIGHT MINERAL OIL (UNII: N6K5787QVP) MANNITOL (UNII: 3OWL53L36A) Product Characteristics Color blue Score no score Shape CAPSULE Size 13mm Flavor Imprint Code PC5 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59726-816-92 96 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 11/29/2019 2 NDC:59726-816-64 1 in 1 BOX 11/29/2019 2 64 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 3 NDC:59726-816-29 2 in 1 BOX 11/29/2019 3 96 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 11/29/2019 Labeler - P & L Development, LLC (800014821)