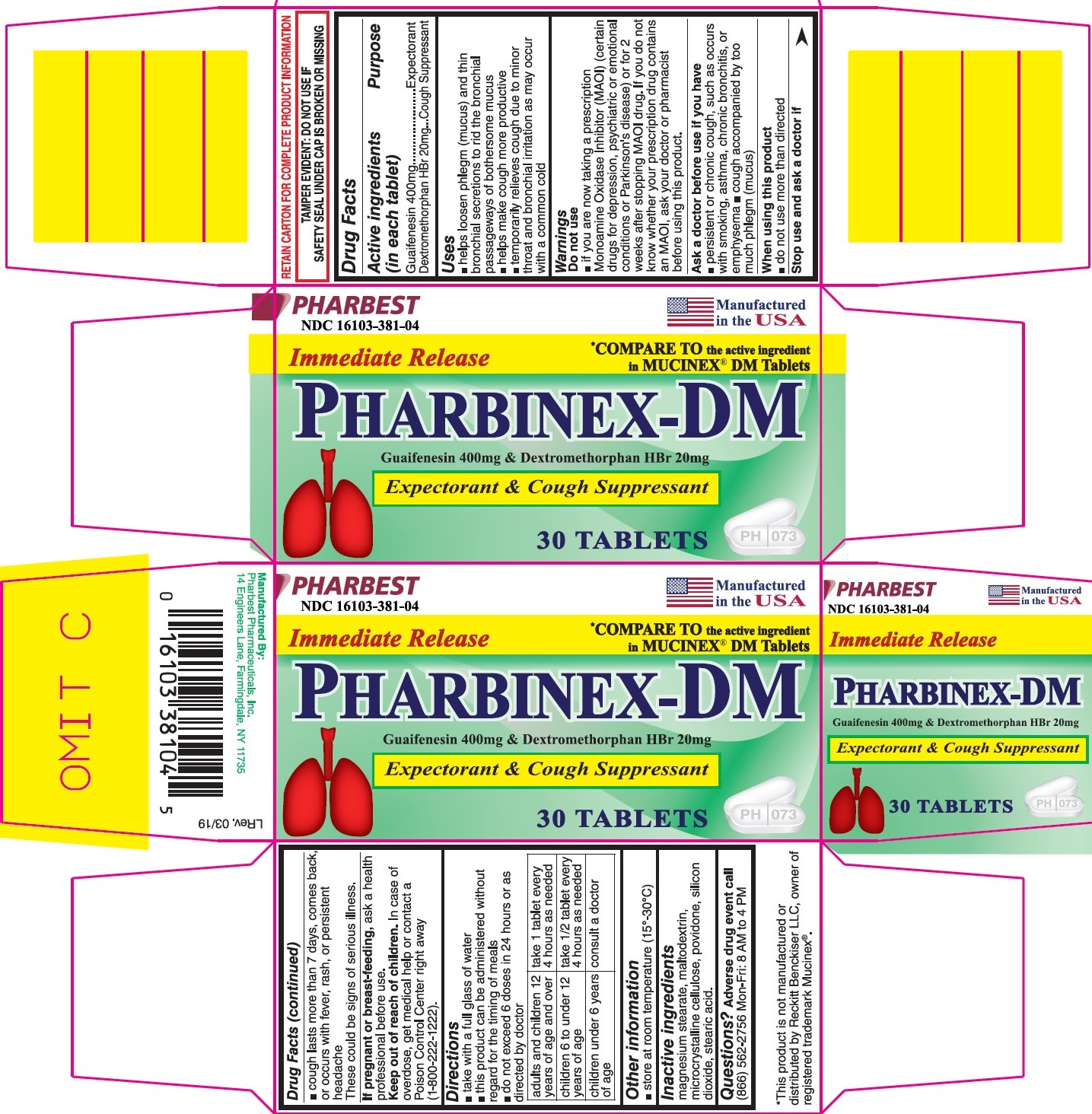
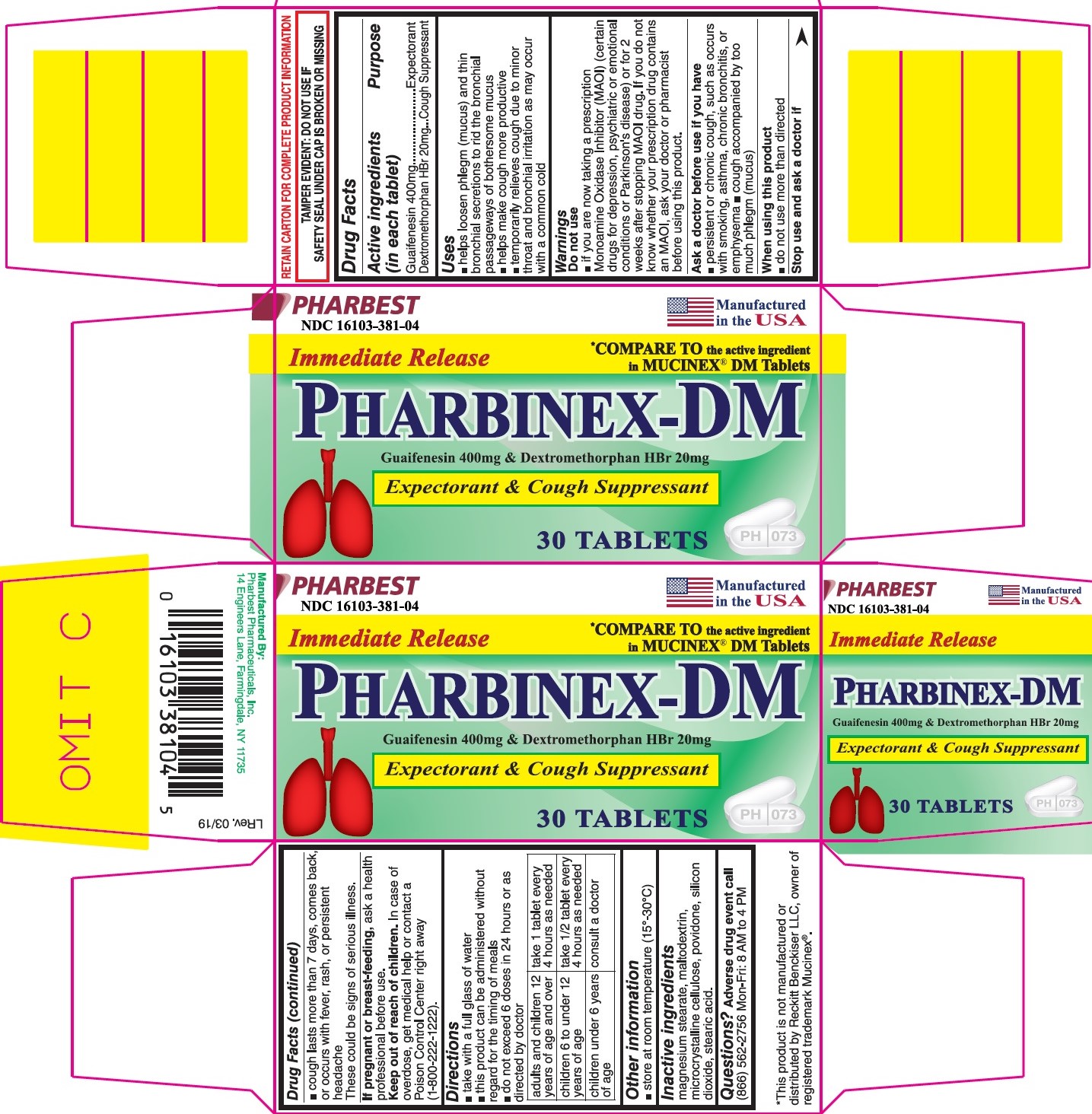
Label: PHARBINEX-DM- guaifenesin 400mg and dextromethorphan hbr 20mg tablet
- NDC Code(s): 16103-381-04
- Packager: Pharbest Pharmaceuticals, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 19, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredients (in each tablet)
- Purpose
- Uses
-
Warnings
Do not use
- if you are now taking a prescription Monoamine Oxidase Inhibitor (MAOI)(certain drugs for depression, psychiatric or emotional conditions or Parkinson's disease) or for 2 weeks after stopping MAOI drug. If you do not know whether your prescription drug contains MAOI, ask your doctor or pharmacist before using this product.
Ask a doctor before use if you have
- persistent or chronic cough, such as occurs with smoking, asthma, chronic bronchitis, or emphysema
- cough accompanied by too much phlegm (mucus)
Stop use and ask a doctor if
- cough lasts more than 7 days, comes back, or occure with fever, rash or persistent headache
These could be signs of serious illness.
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Directions
- take with a full glass of water
- this product can be administered without regard for the timing of meals
- do not exceed 6 doses in 24 hours or as directed by doctor
adults and children 12 years of age and over
take 1 tablet every 4 hours as needed
children 6 to under 12 years of age
take ½ tablet every 4 hours as needed
children under 6 years of age
consult a doctor
- Other information
- Inactive ingredients
- Questions?
-
PRINCIPAL DISPLAY PANEL
PHARBEST
NDC 16103-381-04
Manufactured in the USA
Immediate Release
*Compare to the active ingredient in MUCINEX® DM Tablets
PHARBINEX-DM
Guaifenesin 400mg & Dextromethorphan HBr 20mg
Expectorant & Cough Suppressant
30 TABLETS
RETAIN CARTON FOR COMPLETE PRODUCT INFORMATION
TAMPER EVIDENT: DO NOT USE IF SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
-
INGREDIENTS AND APPEARANCE
PHARBINEX-DM
guaifenesin 400mg and dextromethorphan hbr 20mg tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:16103-381 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 400 mg DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 20 mg Inactive Ingredients Ingredient Name Strength MAGNESIUM STEARATE (UNII: 70097M6I30) MALTODEXTRIN (UNII: 7CVR7L4A2D) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STEARIC ACID (UNII: 4ELV7Z65AP) Product Characteristics Color white Score 2 pieces Shape CAPSULE (CAPSULE SHAPED TABLET) Size 18mm Flavor Imprint Code PH073 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:16103-381-04 1 in 1 CARTON 04/02/2018 1 30 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M 04/02/2018 Labeler - Pharbest Pharmaceuticals, Inc. (557054835) Establishment Name Address ID/FEI Business Operations Pharbest Pharmaceuticals, Inc. 557054835 manufacture(16103-381) , analysis(16103-381) , pack(16103-381) , label(16103-381)