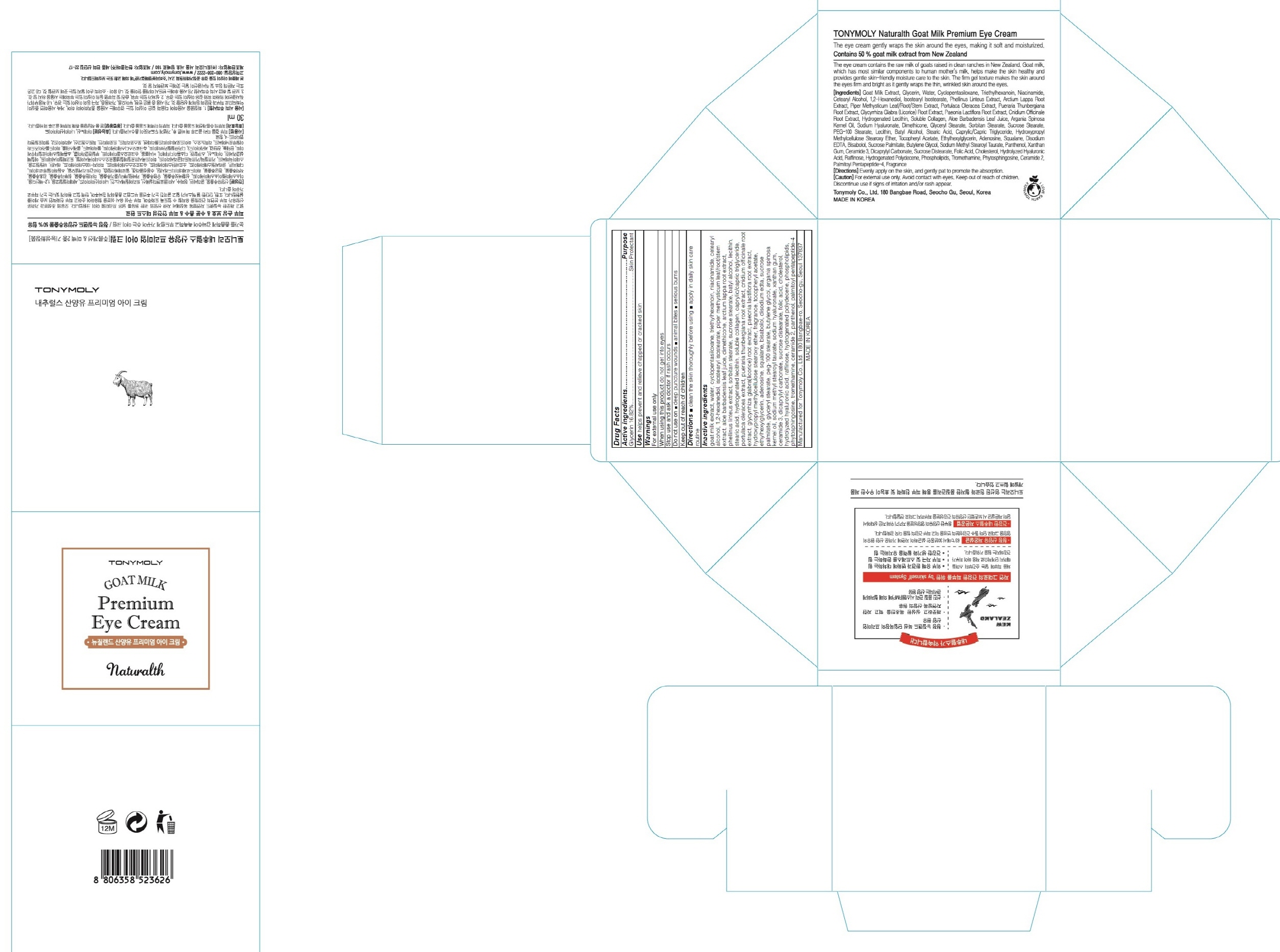
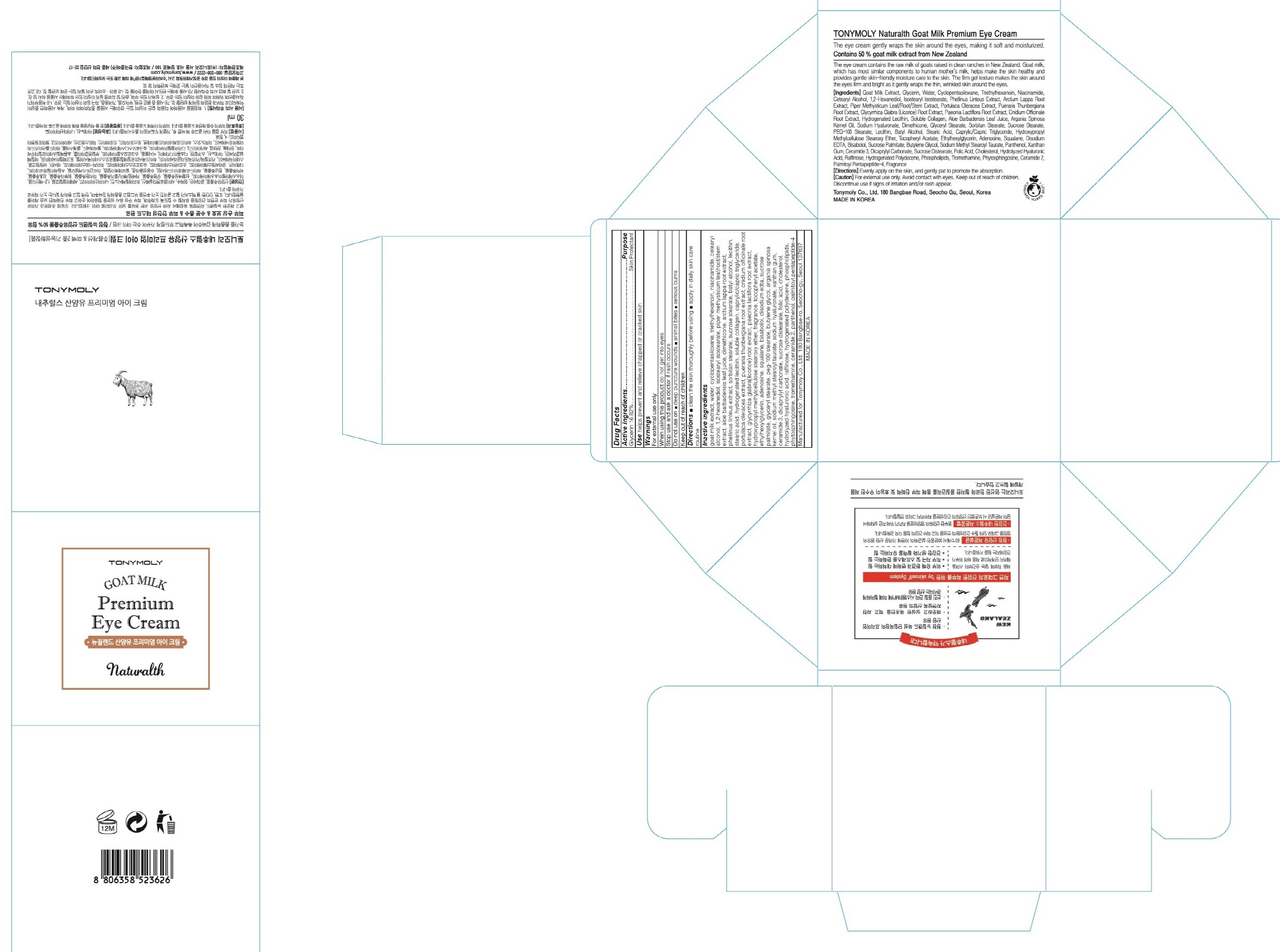
Label: TONYMOLY NATURALTH GOAT MILK PREMIUM EYE- glycerin cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 59078-240-01 - Packager: TONYMOLY CO.,LTD
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated January 20, 2016
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
-
INACTIVE INGREDIENT
Inactive ingredients: goat milk extract, water, cyclopentasiloxane, triethylhexanoin, niacinamide, cetearyl alcohol, 1,2-hexanediol, isostearyl isostearate, piper methysticum leaf/root/stem extract, aloe barbadensis leaf juice, dimethicone, arctium lappa root extract, phellinus linteus extract, sorbitan stearate, sucrose stearate, batyl alcohol, lecithin, stearic acid, hydrogenated lecithin, soluble collagen, caprylic/capric triglyceride, portulaca oleracea extract, pueraria thunbergiana root extract, cnidium officinale root extract, glycyrrhiza glabra(licorice) root extract, paeonia lactiflora root extract, hydroxypropyl methylcellulose stearoxy ether, fragrance, tocopheryl acetate, ethylhexylglycerin, adenosine, squalane, bisabolol, disodium edta, sucrose palmitate, glyceryl stearate, peg-100 stearate, butylene glycol, argania spinosa kernel oil, sodium methyl stearoyl taurate, sodium hyaluronate, xanthan gum, ceramide 3, dicaprylyl carbonate, sucrose distearate, folic acid, cholesterol, hydrolyzed hyaluronic acid, raffinose, hydrogenated polydecene, phospholipids, phytosphingosine, tromethamine, ceramide 2, panthenol, palmitoyl pentapeptide-4
- PURPOSE
- WARNINGS
- KEEP OUT OF REACH OF CHILDREN
- Use
- Directions
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
TONYMOLY NATURALTH GOAT MILK PREMIUM EYE
glycerin creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:59078-240 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Glycerin (UNII: PDC6A3C0OX) (GLYCERIN - UNII:PDC6A3C0OX) Glycerin 5.04 g in 30 mL Inactive Ingredients Ingredient Name Strength goat milk (UNII: XE5K5I4RP7) water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59078-240-01 30 mL in 1 CARTON; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 12/01/2015 Labeler - TONYMOLY CO.,LTD (688216798) Registrant - TONYMOLY CO.,LTD (688216798) Establishment Name Address ID/FEI Business Operations TONYMOLY CO.,LTD 688216798 manufacture(59078-240)