Label: FORMULATION R- phenylephrine hydrochloride, hydrogenate palm kernel oil suppository
-
Contains inactivated NDC Code(s)
NDC Code(s): 0713-0535-12, 0713-0535-24 - Packager: G&W Laboratories, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 30, 2012
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
-
USES
• helps relieve the local itching and discomfort associated with hemorrhoids
• temporarily relieves burning and shrinks hemorrhoidal tissue
• temporarily provides a coating for relief of anorectal discomforts
• temporarily protects the inflamed, irritated anorectal surface to help make bowel movements less painful -
WARNINGS
For rectal use only
Ask a doctor before use if you have
• heart disease
• high blood pressure
• thyroid disease
• diabetes
• difficulty in urination due to an enlarged prostate
Ask a doctor or pharmacist before use if you are
presently taking a prescription drug for high blood pressure or depression
When using this product
• do not use more than directed
Stop use and ask a doctor if
• symptoms get worse or do not get better in 7 days
• bleeding occurs - IF PREGNANT OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
-
DIRECTIONS
adults: when practical, clean the affected area by patting or blotting with an appropriate cleansing wipe. Gently dry by patting or blotting with tissue or a soft cloth before insertion of the product.
• remove wrapper before inserting into the rectum as follows:
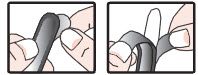
• hold suppository with pointed end up
• carefully separate tabs with fingernail
• slowly pull apart by pulling tabs down on both sides
• remove suppository from wrapper
• insert one suppository into the rectum up to 4 times daily, especially at night, in the morning or after each bowel movement
• children under 12 years: ask a doctor - OTHER INFORMATION
- INACTIVE INGREDIENT
-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
NDC 0713-0535-24
G&W Formulation RTM HEMORRHOIDAL SUPPOSITORIES
Prompt Soothing Relief from Painful Burning, Itching and Discomfort
24 ADULT SUPPOSITORIES
Tamper-Evident: For your safety suppositories are packaged in "Tamper-Evident" sealed foil. Do not use if foil is torn or open
Compare to active ingredients in Preparation-H® Hemorrhoidal Suppositories
FOR TEMPORARY RELIEF FROM PAIN AND ITCHING OF HEMORRHOIDS
-
INGREDIENTS AND APPEARANCE
FORMULATION R
phenylephrine hydrochloride, hydrogenate palm kernel oil suppositoryProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0713-0535 Route of Administration RECTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Phenylephrine Hydrochloride (UNII: 04JA59TNSJ) (Phenylephrine - UNII:1WS297W6MV) Phenylephrine Hydrochloride 2.5 mg Hydrogenated Palm Kernel Oil (UNII: FM8D1RE2VP) (Hydrogenated Palm Kernel Oil - UNII:FM8D1RE2VP) Hydrogenated Palm Kernel Oil 855 mg Inactive Ingredients Ingredient Name Strength Cod Liver Oil (UNII: BBL281NWFG) Starch, Corn (UNII: O8232NY3SJ) Methylparaben (UNII: A2I8C7HI9T) Propylparaben (UNII: Z8IX2SC1OH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0713-0535-24 24 in 1 CARTON 2 NDC:0713-0535-12 12 in 1 CARTON Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part346 06/24/2002 Labeler - G&W Laboratories, Inc. (001271188) Registrant - G&W Laboratories, Inc. (001271188) Establishment Name Address ID/FEI Business Operations G&W Laboratories, Inc. 001271188 MANUFACTURE

