Label: CENESTIN- synthetic conjugated estrogens, a tablet, film coated
-
Contains inactivated NDC Code(s)
NDC Code(s): 54868-1432-0, 54868-1432-1, 54868-4879-0, 54868-4879-1, view more - Packager: Physicians Total Care, Inc.
- This is a repackaged label.
- Source NDC Code(s): 51285-442, 51285-444, 51285-446
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated September 1, 2004
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
SPL UNCLASSIFIED SECTIONRx only - Revised SEPTEMBER 2004 - 11000422506 - ESTROGENS INCREASE THE RISK OF ENDOMETRIAL CANCER - Close clinical surveillance of all women taking estrogens is important. Adequate diagnostic ...
-
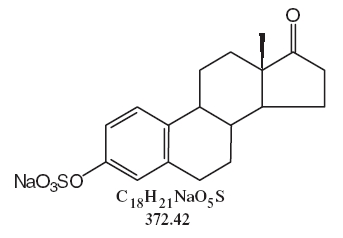
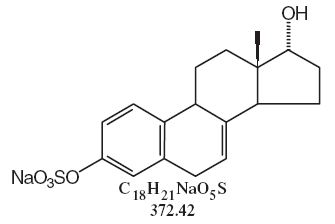
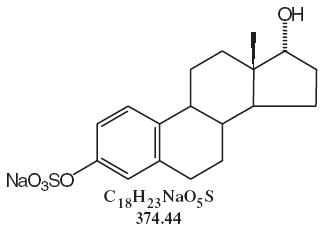
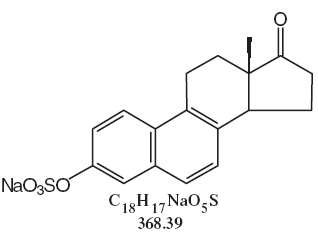
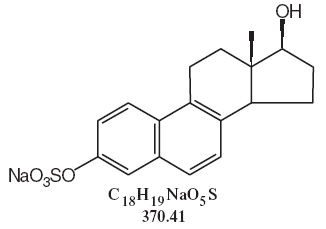
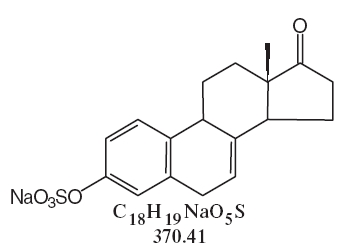
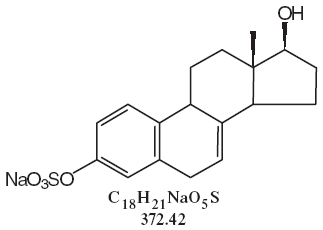
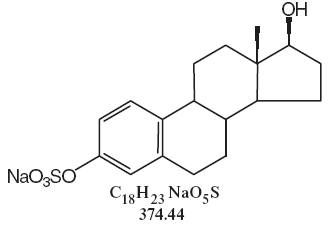
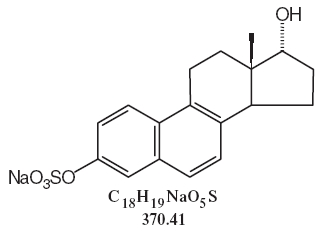
DESCRIPTIONSynthetic conjugated estrogens, A tablets contain a blend of nine (9) synthetic estrogenic substances. The estrogenic substances are sodium estrone sulfate, sodium equilin sulfate, sodium ...
-
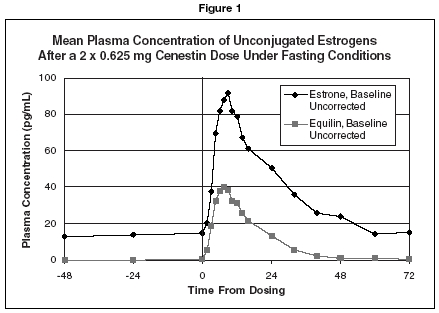
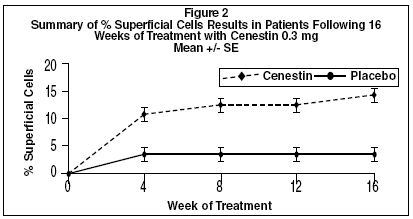
CLINICAL PHARMACOLOGYEndogenous estrogens are largely responsible for the development and maintenance of the female reproductive system and secondary sexual characteristics. Although circulating estrogens exist in a ...
-
INDICATIONS AND USAGECenestin therapy is indicated for the: 1. Treatment of moderate-to-severe vasomotor symptoms associated with the menopause. 0.45 mg Cenestin - 0.625 mg Cenestin - 0.9 mg Cenestin - 1.25 mg Cenestin - 2 ...
-
CONTRAINDICATIONSCenestin should not be used in women with any of the following conditions: Undiagnosed abnormal genital bleeding. Known, suspected, or history of cancer of the breast. Known or suspected ...
-
WARNINGSSee BOXED WARNINGS. 1. Cardiovascular disorders. Estrogen and estrogen/progestin therapy has been associated with an increased risk of cardiovascular events such as myocardial infarction and ...
-
PRECAUTIONSA. General - Addition of a progestin when a woman has not had a hysterectomy. Studies of the addition of a progestin for 10 or more days of a cycle of estrogen administration, or daily with ...
-
ADVERSE REACTIONSSee BOXED WARNINGS, WARNINGS and PRECAUTIONS. Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be ...
-
OVERDOSAGESerious ill effects have not been reported following acute ingestion of large doses of estrogen containing drug products by young children. Overdosage of estrogen may cause nausea and vomiting ...
-
DOSAGE AND ADMINISTRATIONWhen estrogen is prescribed for a postmenopausal woman with a uterus, progestin should also be initiated to reduce the risk of endometrial cancer. A woman without a uterus does not need progestin ...
-
HOW SUPPLIEDCenestin (synthetic conjugated estrogens, A) Tablets are available as: 0.45 mg: Round, orange, film-coated, and are debossed with letters, dp, and number, 46.Available in bottles ...
-
PATIENT INFORMATIONCenestin® (synthetic conjugated estrogens, A) Tablets - Read this PATIENT INFORMATION before you start taking Cenestin and read what you get each time you refill Cenestin. There may be new ...
-
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL - 0.45 mgCenestin® (synthetic conjugated estrogens, A) Tablets - 0.45 mg - Rx only - Each tablet contains: Synthetic conjugated estrogens, A..........0.45 mg - Usual Dosage: See accompanying prescribing ...
-
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL - 0.625 mgCenestin® (synthetic conjugated estrogens, A) Tablets - 0.625 mg - Each tablet contains: Synthetic conjugated estrogens, A..........0.625 mg - Usual Dosage: See accompanying prescribing ...
-
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL - 1.25 mgCenestin® (synthetic conjugated estrogens, A) Tablets - 1.25 mg - Rx only - Each tablet contains: Synthetic conjugated estrogens, A..........1.25 mg - Usual Dosage: See accompanying prescribing ...
-
INGREDIENTS AND APPEARANCEProduct Information