Label: INMAZEB- atoltivimab, maftivimab, and odesivimab-ebgn injection, solution
- NDC Code(s): 61755-018-00, 61755-018-01, 61755-019-00, 61755-019-01
- Packager: Regeneron Pharmaceuticals, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Biologic Licensing Application
Drug Label Information
Updated December 23, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use INMAZEB safely and effectively. See full prescribing information for INMAZEB. INMAZEB® (atoltivimab, maftivimab, and ...
-
Table of ContentsTable of Contents
-
1 INDICATIONS AND USAGEINMAZEB is indicated for the treatment of infection caused by Orthoebolavirus zairense in adult and pediatric patients, including neonates born to a mother who is RT-PCR positive for ...
-
2 DOSAGE AND ADMINISTRATION2.1 Recommended Dosage - INMAZEB is a combination of three human monoclonal antibodies co-formulated in a 1:1:1 ratio of atoltivimab, maftivimab, and odesivimab. INMAZEB is available as two ...
-
3 DOSAGE FORMS AND STRENGTHSINMAZEB is a clear to slightly opalescent and colorless to pale yellow solution available as: Injection: 241.7 mg of atoltivimab, 241.7 mg of maftivimab, and 241.7 mg of odesivimab per 14.5 mL ...
-
4 CONTRAINDICATIONSNone.
-
5 WARNINGS AND PRECAUTIONS5.1 Hypersensitivity Reactions Including Infusion-Associated Events - Hypersensitivity reactions including infusion-associated events have been reported during and post-infusion with INMAZEB ...
-
6 ADVERSE REACTIONSThe following clinically significant adverse reactions are described elsewhere in the labeling: Hypersensitivity Reactions Including Infusion-Associated Events [see Warnings and Precautions ...
-
7 DRUG INTERACTIONS7.1 Vaccine Interactions - No vaccine-therapeutic interaction studies have been performed in human subjects using INMAZEB. However, because of the potential for INMAZEB to inhibit replication of ...
-
8 USE IN SPECIFIC POPULATIONS8.1 Pregnancy - Risk Summary - Orthoebolavirus zairense infection is life-threatening for both the mother and fetus and treatment should not be withheld due to pregnancy (see Clinical ...
-
11 DESCRIPTIONAtoltivimab, maftivimab, and odesivimab-ebgn is a combination of Orthoebolavirus zairense glycoprotein (GP) directed recombinant human IgG1 human monoclonal antibodies of similar structure. The ...
-
12 CLINICAL PHARMACOLOGY12.1 Mechanism of Action - INMAZEB is an antiviral drug combination of three recombinant human IgG1κ monoclonal antibodies (atoltivimab, maftivimab, and odesivimab) that inhibit Orthoebolavirus ...
-
13 NONCLINICAL TOXICOLOGY13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility - Carcinogenicity, genotoxicity, and fertility studies have not been conducted with INMAZEB.
-
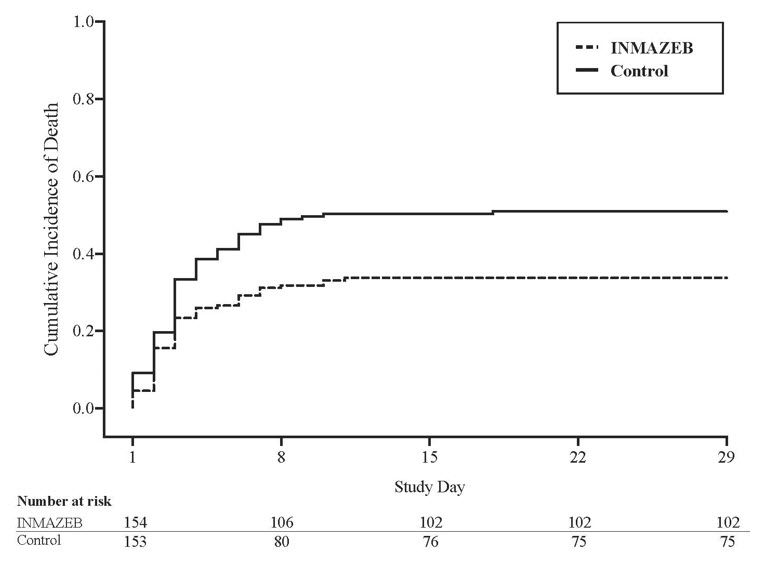
14 CLINICAL STUDIESThe efficacy of INMAZEB was evaluated in PALM, a multi-center, open-label, randomized controlled trial sponsored by the National Institute of Allergy and Infectious Diseases (NIAID; NCT03719586) ...
-
16 HOW SUPPLIED/STORAGE AND HANDLINGINMAZEB (atoltivimab, maftivimab, and odesivimab-ebgn) injection is a clear to slightly opalescent and colorless to pale yellow solution. It is supplied in a carton containing one single dose vial ...
-
17 PATIENT COUNSELING INFORMATIONHypersensitivity Reactions Including Infusion-Associated Events - Inform patients that hypersensitivity reactions including infusion-associated events have been reported during and ...
-
SPL UNCLASSIFIED SECTIONManufactured by: Regeneron Pharmaceuticals, Inc. 777 Old Saw Mill River Road - Tarrytown, NY 10591-6707 - U.S. License No. 1760 - INMAZEB is a registered trademark owned by Regeneron ...
-
PRINCIPAL DISPLAY PANEL - 14.5 mL Vial Carton - 61755-018NDC 61755-018-01 - Rx only - Inmazeb® (atoltivimab, maftivimab, and odesivimab – ebgn) Injection - 241.7 mg / 241.7 mg / 241.7 mg per 14.5 mL - (16.67 mg / 16.67 mg / 16.67 mg per mL) For ...
-
PRINCIPAL DISPLAY PANEL - 14.5 mL Vial Carton - 61755-019NDC 61755-019-01 - Rx only - Inmazeb® (atoltivimab, maftivimab, and odesivimab – ebgn) Injection - 483.3 mg / 483.3 mg / 483.3 mg per 14.5 mL - (33.33 mg / 33.33 mg / 33.33 mg per mL) For ...
-
INGREDIENTS AND APPEARANCEProduct Information