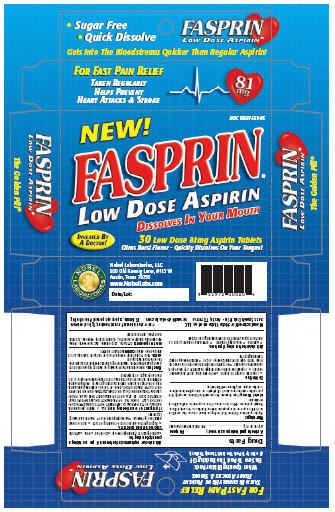
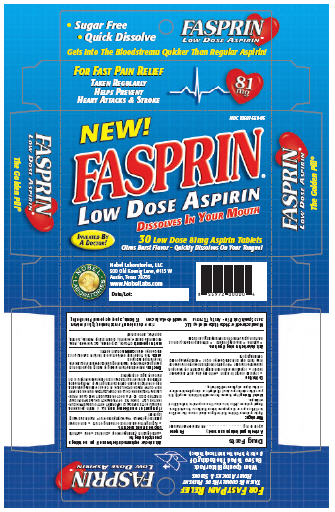
Label: FASPRIN- aspirin tablet, orally disintegrating
-
Contains inactivated NDC Code(s)
NDC Code(s): 10597-559-05, 10597-559-06 - Packager: Nobel Laboratories, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated November 30, 2010
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient (In Each Tablet)
- Purpose
- Uses
-
Warnings:
Reye's syndrome: Children and teenagers should not use this medicine
for chicken pox or flu symptoms before a doctor is consulted about
Reye's syndrome, a rare but serious illness reported to be associated
with aspirin.
Alcohol Warning: If you consume 3 or more alcoholic drinks every day, ask
your doctor whether you should take aspirin or other pain relievers/fever
reducers. Aspirin may cause stomach bleeding.- If you are allergic to aspirin
- With any other pain reliever/fever reducer
- If you have ever had an allergic reaction to any other pain reliever/fever reducer
- For pain for more than 10 days or for fever for more than 3 days unless directed by a doctor
- With any other product containing aspirin
Ask doctor before using if you have:
- Asthma
- Bleeding problems
- Stomach problems (such as heartburn, upset stomach or stomach pain) gastric ulcers
Ask a doctor or pharmacist before use if you are taking a prescription drug for:
- Anticoagulation (thinning of blood)
- Diabetes
- Gout
- Arthritis
- KEEP OUT OF REACH OF CHILDREN
- Directions
- Inactive Ingredients:
- SPL UNCLASSIFIED SECTION
- STORAGE AND HANDLING
- Package Label Section
-
INGREDIENTS AND APPEARANCE
FASPRIN
aspirin tablet, orally disintegratingProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10597-559 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ASPIRIN (UNII: R16CO5Y76E) (ASPIRIN - UNII:R16CO5Y76E) ASPIRIN 81 mg Inactive Ingredients Ingredient Name Strength CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) CROSPOVIDONE (UNII: 68401960MK) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) GLUCOSAMINE (UNII: N08U5BOQ1K) MANNITOL (UNII: 3OWL53L36A) SODIUM STEARYL FUMARATE (UNII: 7CV7WJK4UI) SORBITOL (UNII: 506T60A25R) ZINC GLUCONATE (UNII: U6WSN5SQ1Z) Product Characteristics Color yellow (yellow) Score no score Shape ROUND (Round) Size 9mm Flavor Imprint Code F Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10597-559-05 30 in 1 BOX 2 NDC:10597-559-06 100 in 1 BOX Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part343 09/20/2006 Labeler - Nobel Laboratories, LLC (022735189) Establishment Name Address ID/FEI Business Operations Avema Pharma Solutions 804087794 manufacture