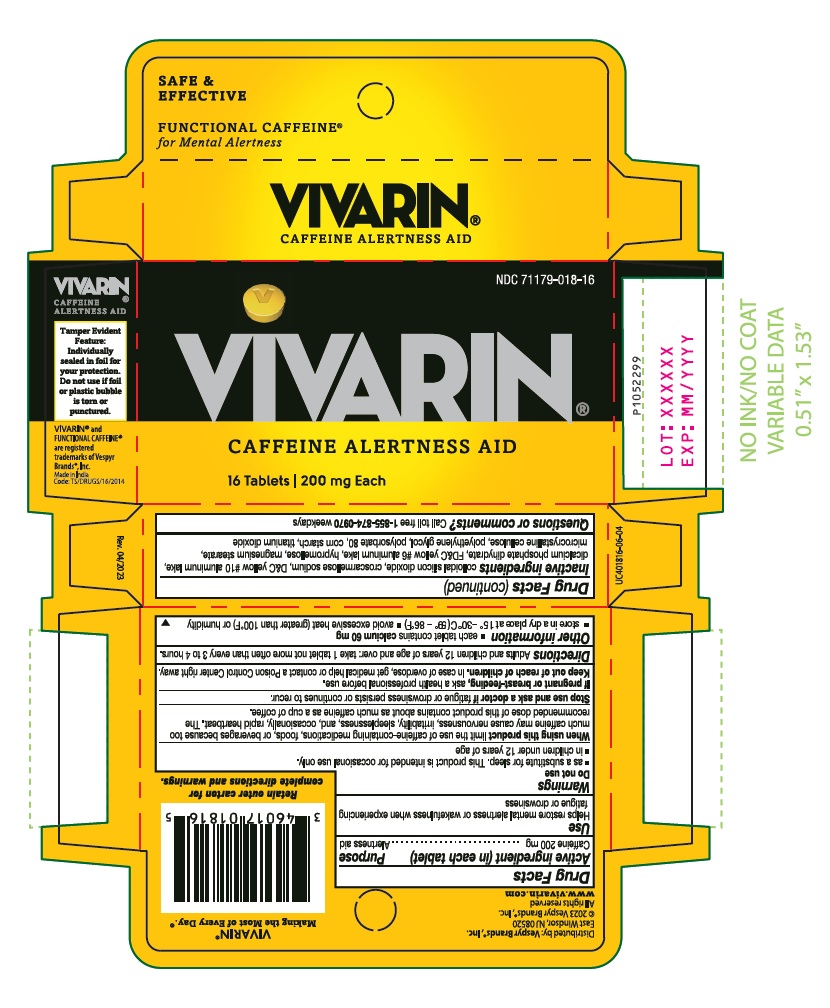
Label: VIVARIN- caffeine tablet
- NDC Code(s): 71179-018-16, 71179-018-40, 71179-018-60
- Packager: VESPYR BRANDS, INC.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 31, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Purpose
- Use
- Warnings
- When using this product
- Stop use and ask a doctor if
- If pregnant or breast-feeding,
- Keep out of reach of children.
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL 200 mg (16 Tablets Container Carton)
-
INGREDIENTS AND APPEARANCE
VIVARIN
caffeine tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71179-018 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAFFEINE (UNII: 3G6A5W338E) (CAFFEINE - UNII:3G6A5W338E) CAFFEINE 200 mg Inactive Ingredients Ingredient Name Strength CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) D&C YELLOW NO. 10 ALUMINUM LAKE (UNII: CQ3XH3DET6) DIBASIC CALCIUM PHOSPHATE DIHYDRATE (UNII: O7TSZ97GEP) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYSORBATE 80 (UNII: 6OZP39ZG8H) STARCH, CORN (UNII: O8232NY3SJ) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color yellow Score no score Shape ROUND Size 11mm Flavor Imprint Code V;V Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71179-018-16 2 in 1 CARTON 06/19/2023 1 8 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC:71179-018-40 5 in 1 CARTON 06/19/2023 2 8 in 1 BLISTER PACK; Type 0: Not a Combination Product 3 NDC:71179-018-60 1 in 1 CARTON 01/02/2024 3 60 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M011 06/19/2023 Labeler - VESPYR BRANDS, INC. (118094893)