Label: AZITHROMYCIN DIHYDRATE tablet, film coated
- NDC Code(s): 68094-900-06, 68094-900-18, 68094-900-30, 68094-900-50
- Packager: Precision Dose, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated November 29, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use AZITHROMYCIN TABLETS safely and effectively. See full prescribing information for AZITHROMYCIN TABLETS. AZITHROMYCIN tablets, for ...
-
Table of ContentsTable of Contents
-
1 INDICATIONS AND USAGEAzithromycin is a macrolide antibacterial drug indicated for the treatment of patients with mild to moderate infections caused by susceptible strains of the designated microorganisms in the ...
-
2 DOSAGE AND ADMINISTRATION2.1 Adult Patients - [see Indications and Usage (1.1) and Clinical Pharmacology (12.3)] Infection*Recommended Dose/Duration of Therapy - * DUE TO THE INDICATED ORGANISMS [see ...
-
3 DOSAGE FORMS AND STRENGTHSAzithromycin Tablets USP, 250 mg (debossed "OE" on one side and "250" on the other side) are supplied as red, oval, film coated tablets containing azithromycin dihydrate equivalent to 250 mg ...
-
4 CONTRAINDICATIONS4.1 Hypersensitivity - Azithromycin is contraindicated in patients with known hypersensitivity to azithromycin, erythromycin, any macrolide, or ketolide drug. 4.2 Hepatic ...
-
5 WARNINGS AND PRECAUTIONS5.1 Hypersensitivity - Serious allergic reactions, including angioedema, anaphylaxis, and dermatologic reactions including Acute Generalized Exanthematous Pustulosis (AGEP), Stevens-Johnson ...
-
6 ADVERSE REACTIONS6.1 Clinical Trials Experience - Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly ...
-
7 DRUG INTERACTIONS7.1 Nelfinavir - Co-administration of nelfinavir at steady-state with a single oral dose of azithromycin resulted in increased azithromycin serum concentrations. Although a dose adjustment of ...
-
8 USE IN SPECIFIC POPULATIONS8.1 Pregnancy - Risk Summary - Available data from published literature and postmarketing experience over several decades with azithromycin use in pregnant women have not identified any ...
-
10 OVERDOSAGEAdverse reactions experienced at higher than recommended doses were similar to those seen at normal doses particularly nausea, diarrhea, and vomiting. In the event of overdosage, general ...
-
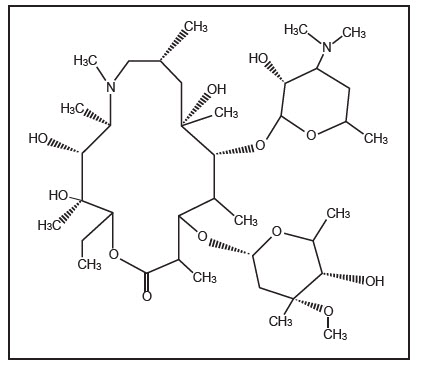
11 DESCRIPTIONAzithromycin Tablets, USP contain the active ingredient azithromycin, a macrolide antibacterial drug, for oral administration. Azithromycin has the chemical name ...
-
12 CLINICAL PHARMACOLOGY12.1 Mechanism of Action - Azithromycin is a macrolide antibacterial drug. [see Microbiology (12.4)] 12.2 Pharmacodynamics - Based on animal models of infection, the antibacterial activity of ...
-
13 NONCLINICAL TOXICOLOGY13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility - Long-term studies in animals have not been performed to evaluate carcinogenic potential. Azithromycin has shown no mutagenic potential ...
-
14 CLINICAL STUDIES14.1 Adult Patients - Acute Bacterial Exacerbations of Chronic Bronchitis - In a randomized, double-blind controlled clinical trial of acute exacerbation of chronic bronchitis (AECB) ...
-
16 HOW SUPPLIED/STORAGE AND HANDLINGAzithromycin Tablets USP, 250 mg are supplied as red, oval, film coated tablets containing azithromycin dihydrate equivalent to 250 mg of azithromycin. Azithromycin Tablets USP, 250 mg are ...
-
17 PATIENT COUNSELING INFORMATIONAdvise the patient to read the FDA-approved patient labeling (Patient Information). General Patient Counseling - Azithromycin tablets can be taken with or without food. Patients should also be ...
-
SPL UNCLASSIFIED SECTIONSee FDA-approved Patient Labeling - Manufactured by: CSPC Ouyi Pharmaceutical Co., Ltd. Shijiazhuang, Hebei, China, 052160 - Manufactured for: Precision Dose, Inc. South Beloit, IL 61080
-
Patient InformationAzithromycin Tablets, USP[ay zith" roe mye' sin]Read this Patient Information leaflet before you start taking azithromycin tablets and each time you get a refill. There may be new information. This information does not take the place of talking ...
-
PRINCIPAL DISPLAY PANEL - 250 mg Tablet Bottle Label - 30 TabletsNDC 68094-900-30 - Azithromycin - Tablets, USP - 250 mg - CSPC® PrecisionDose - 30 Tablets - Rx only
-
PRINCIPAL DISPLAY PANEL - 250 mg Tablet Bottle Label - 100 TabletsNDC 68094-900-50 - Azithromycin - Tablets, USP - 250 mg - CSPC® PrecisionDose - 100 Tablets - Rx only
-
PRINCIPAL DISPLAY PANEL - 250 mg Tablet Blister Pack CartonNDC 68094-900-06 - 1 Card x 6 Tablets - Azithromycin Tablets, USP - 250 mg - A full course of antibiotic therapy in 5 daily doses - Rx only - CSPC® PrecisionDose - Manufactured by: CSPC Ouyi Pharmaceutical ...
-
PRINCIPAL DISPLAY PANEL - 250 mg Tablet Blister Pack Carton BoxNDC 68094-900-18 - 3 Cartons x 6 Tablets - Azithromycin Tablets, USP - 250 mg - A full course of antibiotic therapy in 5 daily doses - Rx only - CSPC® PrecisionDose
-
INGREDIENTS AND APPEARANCEProduct Information