Label: METOPROLOL TARTRATE- metoprolol tablet
- NDC Code(s): 62332-112-30, 62332-112-31, 62332-112-91, 62332-113-30, view more
- Packager: Alembic Pharmaceuticals Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated August 22, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use METOPROLOL TARTRATE TABLETS safely and effectively. See full prescribing information for METOPROLOL TARTRATE TABLETS. METOPROLOL ...
-
Table of ContentsTable of Contents
-
1 INDICATIONS AND USAGE1.1 Hypertension - Metoprolol tartrate tablets are indicated for the treatment of hypertension in adult patients, to lower blood pressure. Lowering blood pressure lowers the risk of fatal and ...
-
2 DOSAGE AND ADMINISTRATION2.1 Hypertension - Individualize the dosage of metoprolol tartrate tablets. Metoprolol tartrate tablets should be taken with or immediately following meals. The usual initial dosage is 100 mg ...
-
3 DOSAGE FORMS AND STRENGTHSMetoprolol tartrate tablets, USP are supplied as: 25 mg tablets having functional scoring are white to off white film coated, round, biconvex tablet debossed with ‘L150’ on one side and ...
-
4 CONTRAINDICATIONSMetoprolol tartrate tablets are contraindicated in severe bradycardia, second-or third-degree heart block, cardiogenic shock, systolic blood pressure <100, decompensated heart failure, sick sinus ...
-
5 WARNINGS AND PRECAUTIONS5.1 Abrupt Cessation of Therapy - Following abrupt cessation of therapy with certain beta-blocking agents, exacerbations of angina pectoris and, in some cases, myocardial infarction have ...
-
6 ADVERSE REACTIONSThe following adverse reactions are described elsewhere in labeling: Worsening angina or myocardial infarction [see Warnings and Precautions (5)] Worsening heart failure [see Warnings and ...
-
7 DRUG INTERACTIONS7.1 Catecholamine Depleting Drugs - Catecholamine depleting drugs (e.g., reserpine, monoamine oxidase (MAO) inhibitors) may have an additive effect when given with beta-blocking agents. Observe ...
-
8 USE IN SPECIFIC POPULATIONS8.1 Pregnancy - Risk Summary - Available data from published observational studies have not demonstrated an association of adverse developmental outcomes with maternal use of metoprolol during ...
-
10 OVERDOSAGESigns and Symptoms – Overdosage of metoprolol tartrate may lead to severe bradycardia, hypotension, and cardiogenic shock. Clinical presentation can also include: atrioventricular block, heart ...
-
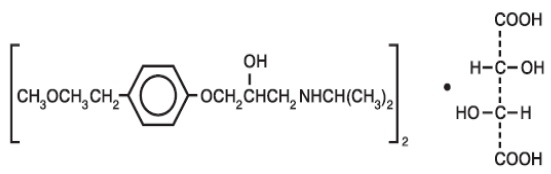
11 DESCRIPTIONMetoprolol tartrate tablets, USP contain metoprolol tartrate, USP a selective beta1-adrenoreceptor blocking agent. Metoprolol tartrate, USP is (±)-1-(Isopropylamino)-3-[p-(2-methoxyethyl ...
-
12 CLINICAL PHARMACOLOGY12.1 Mechanism of Action - Metoprolol is a beta1-selective (cardioselective) adrenergic receptor blocking agent. This preferential effect is not absolute, however, and at higher plasma ...
-
13 NONCLINICAL TOXICOLOGY13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility - Long-term studies in animals have been conducted to evaluate the carcinogenic potential of metoprolol tartrate. In 2-year studies in ...
-
14 CLINICAL STUDIES14.1 Hypertension - In controlled clinical studies, metoprolol tartrate has been shown to be an effective antihypertensive agent when used alone or as concomitant therapy with thiazide-type ...
-
16 HOW SUPPLIED/STORAGE AND HANDLINGMetoprolol Tartrate Tablets, USP are available containing 25 mg, 50 mg or 100 mg of metoprolol tartrate, USP. The 25 mg tablets having functional scoring are white to off white film coated ...
-
17 PATIENT COUNSELING INFORMATIONAdvise patients to take metoprolol tartrate tablets regularly and continuously, as directed, preferably with or immediately following meals. If a dose is missed, the patient should take only the ...
-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL 25 mg strengthNDC 62332-112-30 - Metoprolol Tartrate - Tablets, USP - 25 mg - Rx only - 30 Tablets - Alembic
-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL 50 mg strengthNDC 62332-113-30 - Metoprolol Tartrate - Tablets, USP - 50 mg - Rx only - 30 Tablets - Alembic
-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL 100 mg strengthNDC 62332-114-30 - Metoprolol Tartrate - Tablets, USP - 25 mg - Rx only - 30 Tablets - Alembic
-
INGREDIENTS AND APPEARANCEProduct Information