Label: VILAZODONE- vilazodone hydrochloride tablet
VILAZODONE- vilazodone hydrochloride tablet
- NDC Code(s): 60505-4772-3, 60505-4773-3, 60505-4774-3
- Packager: Apotex Corp
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application Authorized Generic
Drug Label Information
Updated June 1, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Medication Guide: HTML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use VILAZODONE safely and effectively. See full prescribing information for VILAZODONE. VILAZODONE tablets, for oral ...
-
Table of ContentsTable of Contents
-
BOXED WARNING
(What is this?)
WARNING: SUICIDAL THOUGHTS AND BEHAVIORS
Antidepressants increased the risk of suicidal thoughts and behaviors in pediatric and young adult patients in short-term studies. Closely monitor all antidepressant-treated patients for clinical worsening and for emergence of suicidal thoughts and behaviors [see Warnings and Precautions (5.1)]. Vilazodone is not approved for use in pediatric patients [see Use in Specific Populations (8.4)].
Close -
1
INDICATIONS AND USAGE
Vilazodone is indicated for the treatment of major depressive disorder (MDD) in adults [see Clinical Studies (14)].
-
2
DOSAGE AND ADMINISTRATION
2.1 - Dosage for Treatment of Major Depressive Disorder - The recommended target dosage for vilazodone is 20 mg to 40 mg orally once daily with food [see Clinical Pharmacology (12.3) ...
-
3
DOSAGE FORMS AND STRENGTHS
Vilazodone tablets are available as 10 mg, 20 mg and 40 mg film-coated tablets. 10 mg pink, oval tablet, debossed with 10 on one side - 20 mg orange, oval tablet, debossed with 20 on one side - 40 ...
-
4
CONTRAINDICATIONS
Vilazodone is contraindicated in: Patients taking, or within 14 days of stopping, monoamine oxidase inhibitors (MAOIs), including MAOIs such as linezolid or intravenous methylene blue, because of ...
-
5
WARNINGS AND PRECAUTIONS
5.1 - Suicidal Thoughts and Behavior in Adolescents and Young Adults - In pooled analyses of placebo-controlled trials of antidepressant drugs (SSRIs and other antidepressant classes ...
-
6
ADVERSE REACTIONS
The following adverse reactions are discussed in greater detail in other sections of the labeling: Suicidal Thoughts and Behaviors in Adolescents and Young Adults [see Warnings and Precautions ...
-
7
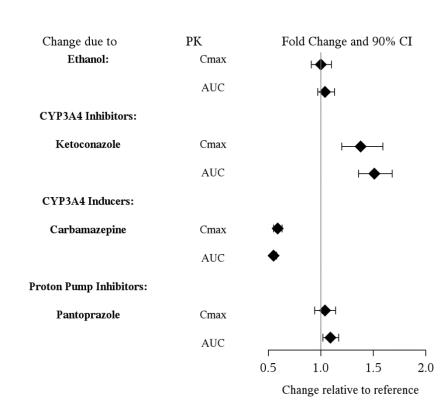
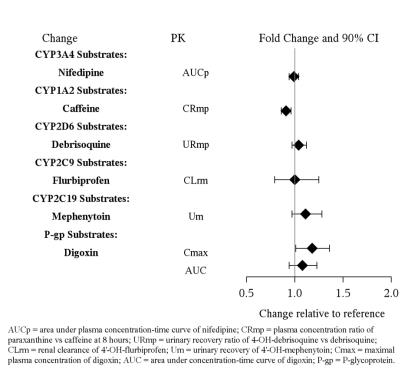
DRUG INTERACTIONS
7.1 - Drugs Having Clinically Important Interactions With Vilazodone - Table 4: Clinically Important Drug Interactions with Vilazodone - Concomitant Drug Name or Drug ...
-
8
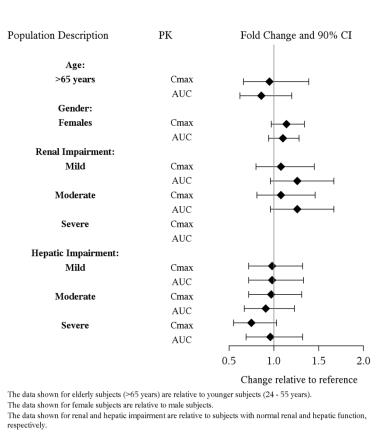
USE IN SPECIFIC POPULATIONS
8.1 - Pregnancy - Pregnancy Exposure Registry - There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to antidepressants during pregnancy. Healthcare ...
-
9
DRUG ABUSE AND DEPENDENCE
9.1 - Controlled Substance - Vilazodone is not a controlled substance. 9.2 - Abuse and Dependence - Vilazodone has been systematically studied in animals and did not ...
-
10
OVERDOSAGE
There is limited clinical trial experience regarding human overdose with vilazodone. The adverse reactions associated with overdose of vilazodone at doses of 200-280 mg (5 to 7 times the ...
-
11
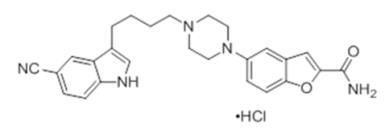
DESCRIPTION
VILAZODONE tablets for oral administration contain polymorph Form IV vilazodone hydrochloride (HCl), a selective serotonin reuptake inhibitor and a 5HT1A receptor partial agonist. Vilazodone HCl ...
-
12
CLINICAL PHARMACOLOGY
12.1 - Mechanism of action - The mechanism of action of vilazodone in the treatment of major depressive disorder is not fully understood, but is thought to be related to its enhancement ...
-
13
NONCLINICAL TOXICOLOGY
13.1 - Carcinogenesis, Mutagenesis, Impairment of Fertility - Carcinogenesis - Carcinogenicity studies were conducted in which B6C3F1mice and Wistar rats were given oral doses of ...
-
14
CLINICAL STUDIES
The efficacy of vilazodone as a treatment for major depressive disorder was demonstrated in four multicenter, randomized, double-blind, placebo-controlled studies in adult (18-70 years of age ...
-
16
HOW SUPPLIED/STORAGE AND HANDLING
Vilazodone tablets are supplied in the following configurations: Tablet - StrengthTablet - Color/ShapeTablet - MarkingsPackage - ConfigurationNDC Code - 10 mgpink, oval tablet debossed with ...
-
17
PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide). Suicidal Thoughts and Behaviors - Advise patients and caregivers to look for the emergence of suicidality ...
-
MEDICATION GUIDEMEDICATION GUIDE - Vilazodone - Tablets, for oral use - What is the most important information I should know about Vilazodone? Vilazodone may cause serious side effects ...
-
PRINCIPAL DISPLAY PANEL
NDC 60505-4772-3 - 30 Tablets - Vilazodone HCI Tablets 10 mg - PHARMACIST: Dispense with - Medication Guide available at - www1.apotex.come/products/us - Rx Only - APOTEX CORP.
-
PRINCIPAL DISPLAY PANEL
NDC 60505-4773-3 - 30 Tablets - Vilazodone HCI Tablets 20 mg - PHARMACIST: Dispense with - Medication Guide available at - www1.apotex.come/products/us - Rx Only - APOTEX CORP.
-
PRINCIPAL DISPLAY PANEL
NDC 60505-4774-3 - 30 Tablets - Vilazodone HCI Tablets 40 mg - PHARMACIST: Dispense with - Medication Guide available at - www1.apotex.come/products/us - Rx Only - APOTEX CORP.
-
INGREDIENTS AND APPEARANCEProduct Information