Label: MEDROXYPROGESTERONE ACETATE tablet
- NDC Code(s): 59762-0055-1, 59762-0055-2, 59762-0056-1, 59762-0056-2, view more
- Packager: Greenstone LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application Authorized Generic
Drug Label Information
Updated August 28, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
BOXED WARNING
(What is this?)Cardiovascular Disorders and Probable Dementia - Estrogen plus progestin therapy should not be used for the prevention of cardiovascular disease or dementia. (See CLINICAL STUDIES and WARNINGS ...
WARNING: CARDIOVASCULAR DISORDERS, BREAST CANCER AND PROBABLE DEMENTIA FOR ESTROGEN PLUS PROGESTIN THERAPY
Cardiovascular Disorders and Probable Dementia
Estrogen plus progestin therapy should not be used for the prevention of cardiovascular disease or dementia. (See CLINICAL STUDIES and WARNINGS, Cardiovascular Disorders and Probable Dementia.)
The Women's Health Initiative (WHI) estrogen plus progestin substudy reported an increased risk of deep vein thrombosis (DVT), pulmonary embolism (PE), stroke and myocardial infarction (MI) in postmenopausal women (50 to 79 years of age) during 5.6 years of treatment with daily oral conjugated estrogens (CE) [0.625] mg combined with medroxyprogesterone acetate (MPA) [2.5 mg], relative to placebo. (See CLINICAL STUDIES and WARNINGS, Cardiovascular Disorders.)
The WHI Memory Study (WHIMS) estrogen plus progestin ancillary study reported an increased risk of developing probable dementia in postmenopausal women 65 years of age or older during 4 years of treatment with daily CE (0.625 mg) combined with MPA (2.5 mg), relative to placebo. It is unknown whether this finding applies to younger postmenopausal women. (See CLINICAL STUDIES and WARNINGS, Probable Dementia and PRECAUTIONS, Geriatric Use.)
CloseBreast Cancer
The WHI estrogen plus progestin substudy demonstrated an increased risk of invasive breast cancer. (See CLINICAL STUDIES and WARNINGS, Malignant Neoplasm, Breast Cancer.)
In the absence of comparable data, these risks should be assumed to be similar for other doses of CE and MPA, and other combinations and dosage forms of estrogens and progestins.
Progestins with estrogens should be prescribed at the lowest effective doses and for the shortest duration consistent with treatment goals and risks for the individual woman.
-
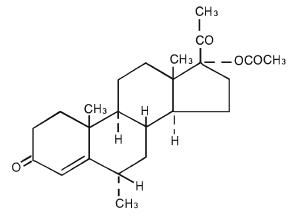
DESCRIPTIONMedroxyprogesterone acetate tablets contain medroxyprogesterone acetate, which is a derivative of progesterone. It is a white to off-white, odorless crystalline powder, stable in air, melting ...
-
CLINICAL PHARMACOLOGYMedroxyprogesterone acetate (MPA) administered orally or parenterally in the recommended doses to women with adequate endogenous estrogen, transforms proliferative into secretory endometrium ...
-
CLINICAL STUDIESEffects on the Endometrium - In a 3-year, double-blind, placebo-controlled study of 356 nonhysterectomized, postmenopausal women between 45 and 64 years of age randomized to receive placebo ...
-
INDICATIONS AND USAGEMedroxyprogesterone acetate tablets are indicated for the treatment of secondary amenorrhea and abnormal uterine bleeding due to hormonal imbalance in the absence of organic pathology, such as ...
-
CONTRAINDICATIONSMPA tablets is contraindicated in women with any of the following conditions: 1. Undiagnosed abnormal genital bleeding. 2. Known, suspected, or history of breast cancer. 3. Known or suspected ...
-
WARNINGSSee BOXED WARNINGS. 1. Cardiovascular Disorders - An increased risk of PE, DVT, stroke, and MI has been reported with estrogen plus progestin therapy. Should any of these events occur or be ...
-
PRECAUTIONSA .General - 1. Addition of a progestin when a woman has not had a hysterectomy - Studies of the addition of a progestin for 10 or more days of a cycle of estrogen administration, or daily ...
-
ADVERSE REACTIONSSee BOXED WARNINGS, WARNINGS, and PRECAUTIONS. Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be ...
-
OVERDOSAGEOverdosage of estrogen plus progestin therapy may cause nausea and vomiting, breast tenderness, dizziness, abdominal pain, drowsiness/fatigue and withdrawal bleeding may occur in women. Treatment ...
-
DOSAGE AND ADMINISTRATIONSecondary Amenorrhea - MPA tablets may be given in dosages of 5 or 10 mg daily for 5 to 10 days. A dose for inducing an optimum secretory transformation of an endometrium that has been adequately ...
-
HOW SUPPLIEDMedroxyprogesterone acetate tablets are available in the following strengths and package sizes: 2.5 mg tablets - Bottles of 100: NDC 59762-0055-1 (scored, round, orange, imprinted PROVERA ...
-
SPL UNCLASSIFIED SECTIONRx only - LAB-0145-9.0 - Revised March 2024
-
PATIENT INFORMATIONMedroxyprogesterone Acetate Tablets, USP - Read this Patient Information before you start taking medroxyprogesterone acetate tablets and read what you get each time you refill your ...
-
PRINCIPAL DISPLAY PANEL - 2.5 mg Tablet Bottle Label - NDC 59762-0055-1NDC 59762-0055-1 - 100 Tablets - GREENSTONE® BRAND - medroxy- progesterone - acetate - tablets, USP - 2.5 mg - Rx only
-
PRINCIPAL DISPLAY PANEL - 5 mg Tablet Bottle Label - NDC 59762-0058-1NDC 59762-0058-1 - 100 Tablets - GREENSTONE® BRAND - medroxy- progesterone - acetate - tablets, USP - 5 mg - Rx only
-
PRINCIPAL DISPLAY PANEL - 10 mg Tablet Bottle Label - NDC 59762-0056-1NDC 59762-0056-1 - 100 Tablets - GREENSTONE® BRAND - medroxy- progesterone - acetate - tablets, USP - 10 mg - Rx only
-
INGREDIENTS AND APPEARANCEProduct Information