Label: METOPROLOL SUCCINATE tablet, extended release
- NDC Code(s): 50742-615-01, 50742-615-05, 50742-615-10, 50742-615-90, view more
- Packager: Ingenus Pharmaceuticals, LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated August 9, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use METOPROLOL SUCCINATE EXTENDED-RELEASE TABLETS safely and effectively. See full prescribing information for METOPROLOL SUCCINATE ...
-
Table of ContentsTable of Contents
-
1 INDICATIONS AND USAGE1.1 Hypertension - Metoprolol succinate extended-release tablets are indicated for the treatment of hypertension, to lower blood pressure. Lowering blood pressure lowers the risk of fatal and ...
-
2 DOSAGE AND ADMINISTRATION2.1 Hypertension - Adults: The usual initial dosage is 25 to 100 mg daily in a single dose. Adjust dosage at weekly (or longer) intervals until optimum blood pressure reduction is achieved. In ...
-
3 DOSAGE FORMS AND STRENGTHS25 mg tablets white, oval, biconvex, scored on both sides, film-coated tablet engraved with "N / 25". 50 mg tablets: white, round, biconvex, scored on both sides, film-coated tablet engraved with ...
-
4 CONTRAINDICATIONSMetoprolol succinate extended-release tablets are contraindicated in severe bradycardia, second- or third-degree heart block, cardiogenic shock, decompensated heart failure, sick sinus syndrome ...
-
5 WARNINGS AND PRECAUTIONS5.1 Abrupt Cessation of Therapy - Following abrupt cessation of therapy with certain beta-blocking agents, exacerbations of angina pectoris and, in some cases, myocardial infarction have ...
-
6 ADVERSE REACTIONSThe following adverse reactions are described elsewhere in labeling: Worsening angina or myocardial infarction. [see Warnings and Precautions (5)] Worsening heart failure. [see Warnings and ...
-
7 DRUG INTERACTIONS7.1 Catecholamine Depleting Drugs - Catecholamine depleting drugs (e.g. reserpine, monoamine oxidase (MAO) inhibitors) may have an additive effect when given with beta-blocking agents. Observe ...
-
8 USE IN SPECIFIC POPULATIONS8.1 Pregnancy - Risk Summary - Untreated hypertension and heart failure during pregnancy can lead to adverse outcomes for the mother and the fetus (see Clinical Considerations). Available data ...
-
10 OVERDOSAGESigns and Symptoms - Overdosage of metoprolol succinate extended-release tablets may lead to severe bradycardia, hypotension, and cardiogenic shock. Clinical presentation can also include ...
-
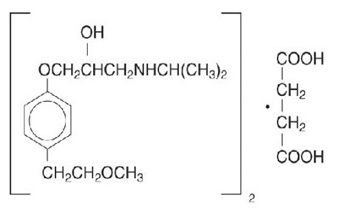
11 DESCRIPTIONMetoprolol succinate, is a beta1-selective (cardioselective) adrenoceptor blocking agent, for oral administration, available as extended-release tablets. Metoprolol succinate extended-release ...
-
12 CLINICAL PHARMACOLOGY12.1 Mechanism of Action - Metoprolol is a beta1-selective (cardioselective) adrenergic receptor blocking agent. This preferential effect is not absolute, however, and at higher plasma ...
-
13 NONCLINICAL TOXICOLOGY13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility - Long-term studies in animals have been conducted to evaluate the carcinogenic potential of metoprolol tartrate. In 2-year studies in ...
-
14 CLINICAL STUDIES14.1 Hypertension - In a double-blind study, 1092 patients with mild-to-moderate hypertension were randomized to once daily metoprolol succinate extended-release tablets (25, 100, or 400 mg) ...
-
16 HOW SUPPLIED/STORAGE AND HANDLING Tablets containing metoprolol succinate ,USP equivalent to the indicated weight of metoprolol tartrate, USP, are white, biconvex, film-coated, and scored on both sides. Tablet Shape ...
-
17 PATIENT COUNSELING INFORMATIONAdvise patients to take metoprolol succinate extended–release tablets regularly and continuously, as directed, preferably with or immediately following meals. If a dose is missed, the patient ...
-
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL – 25 mg NDC 50742-615-01 - 100 Tablets - Metoprolol succinate extended-release tablets, USP - 25 mg* Rx only - Ingenus
-
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL – 50 mg NDC 50742-616-01 - 100 Tablets - Metoprolol succinate extended-release tablets, USP - 50 mg* Rx only - Ingenus
-
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL – 100 mg NDC 50742-617-01 - 100 Tablets - Metoprolol succinate extended-release tablets, USP - 100 mg* Rx only - Ingenus
-
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL – 200 mg NDC 50742-618-01 - 100 Tablets - Metoprolol succinate extended-release tablets, USP - 200 mg* Rx only - Ingenus
-
INGREDIENTS AND APPEARANCEProduct Information