Label: HYDROCORTISONE- hydrocortisone acetate cream
- NDC Code(s): 50332-0042-1, 50332-0042-2, 50332-0042-4, 50332-0042-7
- Packager: HART Health
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated February 16, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- DO NOT USE
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- INACTIVE INGREDIENT
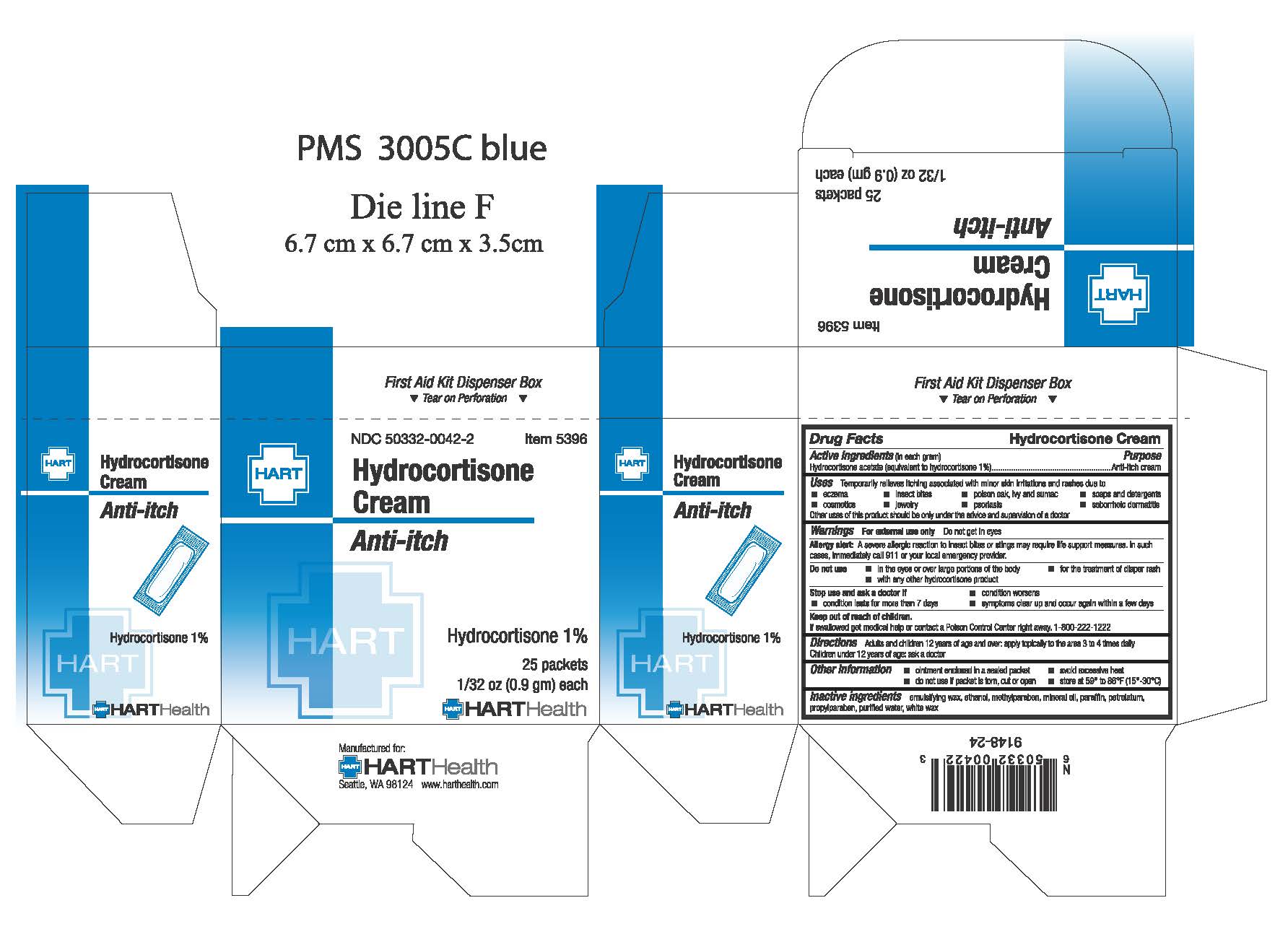
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
HYDROCORTISONE
hydrocortisone acetate creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:50332-0042 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYDROCORTISONE ACETATE (UNII: 3X7931PO74) (HYDROCORTISONE - UNII:WI4X0X7BPJ) HYDROCORTISONE ACETATE 10 mg in 1 g Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) METHYLPARABEN (UNII: A2I8C7HI9T) MINERAL OIL (UNII: T5L8T28FGP) PARAFFIN (UNII: I9O0E3H2ZE) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) WHITE WAX (UNII: 7G1J5DA97F) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50332-0042-1 10 in 1 BOX, UNIT-DOSE 01/01/1974 1 0.9 g in 1 PACKET; Type 0: Not a Combination Product 2 NDC:50332-0042-7 10 in 1 BAG 01/01/2000 2 0.9 g in 1 PACKET; Type 0: Not a Combination Product 3 NDC:50332-0042-2 25 in 1 BOX, UNIT-DOSE 01/01/1974 3 0.9 g in 1 PACKET; Type 0: Not a Combination Product 4 NDC:50332-0042-4 144 in 1 BOX, UNIT-DOSE 01/01/1974 4 0.9 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M 01/01/1974 Labeler - HART Health (069560969)