Label: IRBESARTAN tablet
- NDC Code(s): 43547-374-03, 43547-374-09, 43547-374-50, 43547-375-03, view more
- Packager: Solco Healthcare U.S., LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated July 14, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use IRBESARTAN TABLETS safely and effectively. See full prescribing information for IRBESARTAN TABLETS. IRBESARTAN tablets, for oral ...
-
Table of ContentsTable of Contents
-
BOXED WARNING
(What is this?)
WARNING: FETAL TOXICITY
- •
- When pregnancy is detected, discontinue irbesartan tablets as soon as possible[see Warnings and Precautions (5.1) and Use in Specific Populations (8.1)].
- •
- Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1)].
-
1 INDICATIONS AND USAGE1.1 Hypertension - Irbesartan tablets are indicated for the treatment of hypertension, to lower blood pressure. Lowering blood pressure lowers the risk of fatal and non-fatal cardiovascular (CV ...
-
2 DOSAGE AND ADMINISTRATION2.1 General Considerations - Irbesartan tablets may be administered with other antihypertensive agents and with or without food. 2.2 Hypertension - The recommended initial dose of irbesartan ...
-
3 DOSAGE FORMS AND STRENGTHSIrbesartan tablet USP, 75 mg is a white to off-white biconvex oval tablet debossed ‘HH’ on one side and ‘329’ on the other. Irbesartan tablet USP, 150 mg is a white to off-white biconvex oval ...
-
4 CONTRAINDICATIONSIrbesartan tablets are contraindicated in patients who are hypersensitive to any component of this product. Do not coadministrate aliskiren with irbesartan tablets in patients with diabetes.
-
5 WARNINGS AND PRECAUTIONS5.1 Fetal Toxicity - Irbesartan tablets can cause fetal harm when administered to a pregnant woman. Use of drugs that act on the renin-angiotensin system during the second and third trimesters of ...
-
6 ADVERSE REACTIONSThe following important adverse reactions are described elsewhere in the labeling: • Hypotension in Volume or Salt-Depleted Patients [see Warnings and Precautions (5.2)] • Impaired Renal ...
-
7 DRUG INTERACTIONS7.1 Agents Increasing Serum Potassium - Coadministration of irbesartan tablets with other drugs that raise serum potassium levels may result in hyperkalemia, sometimes severe. Monitor serum ...
-
8 USE IN SPECIFIC POPULATIONS8.1 Pregnancy - Risk Summary - Irbesartan tablets can cause fetal harm when administered to a pregnant woman. Use of drugs that act on the renin-angiotensin system during the second and third ...
-
10 OVERDOSAGENo data are available in regard to overdosage in humans. However, daily doses of 900 mg for 8 weeks were well-tolerated. The most likely manifestations of overdosage are expected to be hypotension ...
-
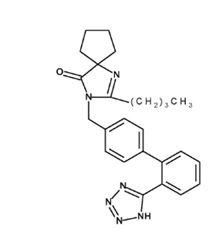
11 DESCRIPTIONIrbesartan is an angiotensin II receptor (AT1 subtype) antagonist. Irbesartan is a non-peptide compound, chemically described as a ...
-
12 CLINICAL PHARMACOLOGY12.1 Mechanism of Action - Angiotensin II is a potent vasoconstrictor formed from angiotensin I in a reaction catalyzed by angiotensin-converting enzyme (ACE, kininase II). Angiotensin II is the ...
-
13 NONCLINICAL TOXICOLOGY13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility - No evidence of carcinogenicity was observed when irbesartan was administered at dosages of up to 500/1000 mg/kg/day (males/females ...
-
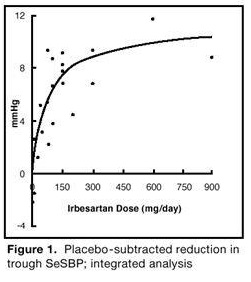
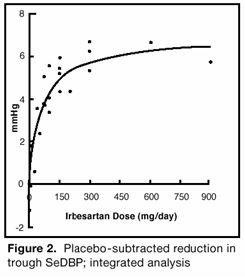
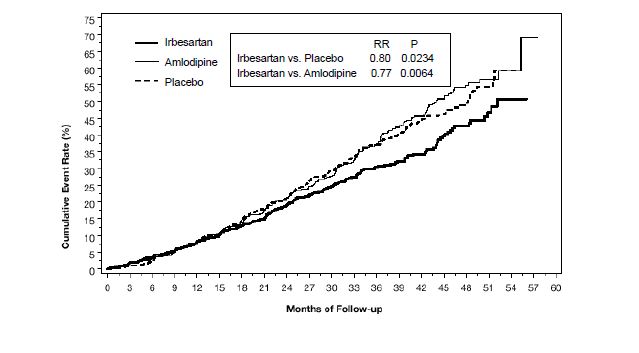
14 CLINICAL STUDIES14.1 Hypertension - The antihypertensive effects of irbesartan tablets were examined in 7 placebo-controlled 8- to 12-week trials in patients with baseline diastolic blood pressures of 95 to 110 ...
-
16 HOW SUPPLIED/STORAGE AND HANDLINGIrbesartan tablets are available as white to off-white biconvex oval tablets, debossed with “HH” on one side and a code on the other (see Table below). Unit-of-use bottles contain 30, 90 or 500 ...
-
17 PATIENT COUNSELING INFORMATIONPregnancy - Advise female patients of childbearing age about the consequences of exposure to irbesartan tablets during pregnancy. Discuss treatment options with women planning to become pregnant ...
-
SPL UNCLASSIFIED SECTIONDistributed by: Solco Healthcare US, LLC - Somerset, NJ 08873, USA - Manufactured by: Zhejiang Huahai Pharmaceutical Co., Ltd. Xunqiao, Linhai, Zhejiang 317024, China - Revised ...
-
PRINCIPAL DISPLAY PANELNDC 43547-374-03 Rx only - Irbesartan - Tablets, USP - 75 mg - 30 Tablets SolcoHealthcare U.S.
-
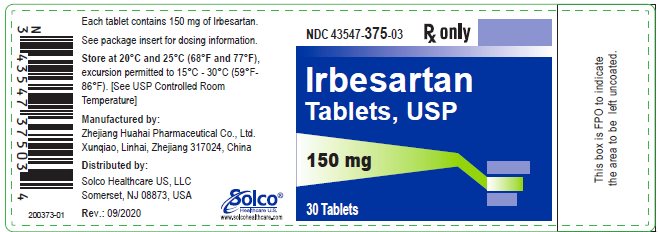
PRINCIPAL DISPLAY PANELNDC 43547-375-03 Rx only - Irbesartan - Tablets, USP - 150 mg - 30 Tablets SolcoHealthcare U.S.
-
PRINCIPAL DISPLAY PANELNDC 43547-376-03 Rx only - Irbesartan - Tablets, USP - 300 mg - 30 Tablets SolcoHealthcare U.S.
-
INGREDIENTS AND APPEARANCEProduct Information