Label: HYDRALAZINE HYDROCHLORIDE tablet
- NDC Code(s): 23155-832-01, 23155-832-10, 23155-833-01, 23155-833-10, view more
- Packager: Heritage Pharmaceuticals Inc. d/b/a Avet Pharmaceuticals Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated January 6, 2025
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
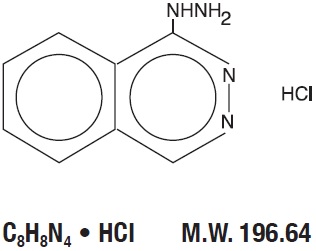
DESCRIPTIONHydrALAZINE hydrochloride, USP, is an antihypertensive, for oral administration. Its chemical name is 1-hydrazinophthalazine monohydrochloride, and its structural formula is: HydrALAZINE ...
-
CLINICAL PHARMACOLOGYAlthough the precise mechanism of action of hydrALAZINE is not fully understood, the major effects are on the cardiovascular system. HydrALAZINE apparently lowers blood pressure by exerting a ...
-
INDICATIONS AND USAGEEssential hypertension, alone or as an adjunct.
-
CONTRAINDICATIONSHypersensitivity to hydrALAZINE; coronary artery disease; mitral valvular rheumatic heart disease.
-
WARNINGSIn a few patients hydrALAZINE may produce a clinical picture simulating systemic lupus erythematosus including glomerulonephritis. In such patients hydrALAZINE should be discontinued unless the ...
-
PRECAUTIONSGeneral - Myocardial stimulation produced by hydrALAZINE can cause anginal attacks and ECG changes of myocardial ischemia. The drug has been implicated in the production of myocardial infarction ...
-
ADVERSE REACTIONSAdverse reactions with hydrALAZINE are usually reversible when dosage is reduced. However, in some cases it may be necessary to discontinue the drug. The following adverse reactions have been ...
-
OVERDOSAGEAcute Toxicity: No deaths due to acute poisoning have been reported. Highest known dose survived: adults, 10 g orally. Oral LD50 in rats: 173 and 187 mg/kg. Signs and Symptoms - Signs and ...
-
DOSAGE AND ADMINISTRATIONInitiate therapy in gradually increasing dosages; adjust according to individual response. Start with 10 mg four times daily for the first 2 to 4 days, increase to 25 mg four times daily for the ...
-
HOW SUPPLIEDHydrALAZINE Hydrochloride Tablets, USP are available as: 10 mg – Round, peach core tablet, debossed EP on one side and 101 on the reverse side. NDC 23155-832-01 Bottles of 100 - NDC 23155-832-10 ...
-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL - 10 mgNDC 23155-832-01 - HydrALAZINE Hydrochloride Tablets, USP - 10 mg - 100 Tablets - Rx only
-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL - 25 mgNDC 23155-833-01 - HydrALAZINE Hydrochloride Tablets, USP - 25 mg - 100 Tablets - Rx only -
-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL - 50 mgNDC 23155-834-01 - HydrALAZINE Hydrochloride Tablets, USP - 50 mg - 100 Tablets - Rx only
-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL - 100 mgNDC 23155-835-01 - HydrALAZINE Hydrochloride Tablets, USP - 100 mg - 100 Tablets - Rx only
-
INGREDIENTS AND APPEARANCEProduct Information