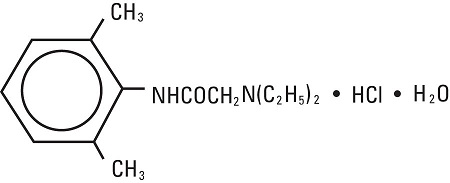Label: LIDOCAINE HYDROCHLORIDE AND EPINEPHRINE injection, solution
- NDC Code(s): 0409-0007-01, 0409-0007-10, 0409-0147-01, 0409-0147-10, view more
- Packager: Hospira, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated March 17, 2025
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use LIDOCAINE HYDROCHLORIDE AND EPINEPHRINE INJECTION safely and effectively. See full prescribing information for LIDOCAINE ...
-
Table of ContentsTable of Contents
-
1 INDICATIONS AND USAGE Lidocaine Hydrochloride and Epinephrine Injection is indicated in adult and pediatric patients for the production of local or regional anesthesia or analgesia for surgery, dental, and oral surgery ...
-
2 DOSAGE AND ADMINISTRATION 2.1 Important Dosage and Administration Information - • Lidocaine Hydrochloride and Epinephrine Injection is not recommended for intrathecal use. • Avoid use of Lidocaine Hydrochloride and ...
-
3 DOSAGE FORMS AND STRENGTHS For Epidural test dose, Lidocaine Hydrochloride and Epinephrine Injection, USP 1:200,000 is a clear, colorless to slightly yellow solution available as: • 1.5% (75 mg/5 mL) (15 mg/mL), 5 mL ...
-
4 CONTRAINDICATIONS Lidocaine Hydrochloride and Epinephrine Injections are contraindicated in patients with a known hypersensitivity to lidocaine or to any local anesthetics of the amide-type or to other components ...
-
5 WARNINGS AND PRECAUTIONS 5.1 Dose-Related Toxicity - The safety and effectiveness of Lidocaine Hydrochloride and Epinephrine Injection depend on proper dosage, correct technique, adequate precautions, and readiness for ...
-
6 ADVERSE REACTIONS The following clinically significant adverse reactions have been reported and described in the Warnings and Precautions section of the labeling: • Dose-Related Toxicity [see Warnings and ...
-
7 DRUG INTERACTIONS 7.1 Local Anesthetics - The toxic effects of local anesthetics are additive. If coadministration of other local anesthetics with Lidocaine Hydrochloride and Epinephrine Injection cannot be ...
-
8 USE IN SPECIFIC POPULATIONS 8.1 Pregnancy - Risk Summary - Available published data and decades of clinical use with lidocaine hydrochloride in pregnant women have not identified any drug-associated risk for major birth ...
-
10 OVERDOSAGE Clinical Presentation - Acute emergencies from local anesthetics are generally related to high plasma levels encountered during therapeutic use of local anesthetics or to unintended subarachnoid ...
-
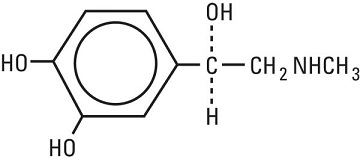
11 DESCRIPTION Lidocaine Hydrochloride and Epinephrine Injection, USP is a sterile, nonpyrogenic solution of lidocaine hydrochloride and epinephrine in water for injection for parenteral administration in ...
-
12 CLINICAL PHARMACOLOGY 12.1 Mechanism of Action - Lidocaine hydrochloride stabilizes the neuronal membrane by inhibiting the ionic fluxes required for the initiation and conduction of impulses thereby effecting local ...
-
13 NONCLINICAL TOXICOLOGY 13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility - Carcinogenesis - Studies of lidocaine hydrochloride in animals to evaluate the carcinogenic potential have not been ...
-
16 HOW SUPPLIED/STORAGE AND HANDLING Do not autoclave. Storage: All solutions should be stored at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Protect ...
-
17 PATIENT COUNSELING INFORMATION 17.1 Allergic-Type Reactions - Assess if the patient has had allergic-type reactions to amide-type local anesthetics or to other formulation ingredients, such as the antimicrobial preservative ...
-
PRINCIPAL DISPLAY PANEL - 5 mL Ampule Label5 mL Single-dose - NDC 0409-1209-10 - LIDOCAINE HCl 1.5% 75 mg/5 mL (15 mg/mL) and ...
-
PRINCIPAL DISPLAY PANEL - 5 mL Ampule Tray Label5 mL Single-dose - 10 Ampuls - Rx only - NDC 0409-1209-01 - Contains 10 of NDC 0409-1209-10 - Lidocaine HCl 1.5% 75 mg/5 mL (15 mg/mL ...
-
PRINCIPAL DISPLAY PANEL - 5 mL Ampule Label - 12095 mL Single-dose - NDC 0409-1209-70 - LIDOCAINE HCl 1.5% 75 mg/5 mL (15 ...
-
PRINCIPAL DISPLAY PANEL - 50 mL Vial Label50 mL - MULTIPLE-DOSE - Lidocaine HCl 0.5% 250 mg/50 mL (5 mg/mL) and - Epinephrine 1:200,000 - Injection, USP - Rx only - For Infiltration and Nerve Block. NOT FOR EPIDURAL OR CAUDAL USE. Distributed ...
-
PRINCIPAL DISPLAY PANEL - 50 mL Vial Tray50 mL MULTIPLE-DOSE Fliptop Vial - Rx only - NDC 0409-3177-01 - Contains 25 of NDC 0409-3177-16 - Lidocaine HCl 0.5% 250 mg/50 mL (5 mg/mL) and Epinephrine 1:200,000 Injection, USP - Protect from light ...
-
PRINCIPAL DISPLAY PANEL - 20 mL Vial Label20 mL - MULTIPLE-DOSE - Lidocaine HCl 1% 200 mg/20 mL - (10 mg/mL) and - Epinephrine 1:100,000 - Injection, USP - For Infiltration and Nerve Block. Rx only - NOT FOR EPIDURAL OR CAUDAL USE. Distributed by ...
-
PRINCIPAL DISPLAY PANEL - 20 mL Vial Tray20 mL MULTIPLE-DOSE Fliptop Vial - Rx only - NDC 0409-3178-01 - Contains 25 of NDC 0409-3178-16 - Lidocaine HCl 1% 200 mg/20 mL (10 mg/mL) and Epinephrine 1:100,000 Injection, USP - Protect from light ...
-
PRINCIPAL DISPLAY PANEL - 30 mL Vial Label30 mL - MULTIPLE-DOSE - Lidocaine HCl 1% 300 mg/30 mL - (10 mg/mL) and - Epinephrine - 1:100,000 - Injection, USP - For Infiltration and Nerve Block. NOT FOR EPIDURAL OR CAUDAL USE.
-
PRINCIPAL DISPLAY PANEL - 30 mL Vial Tray30 mL MULTIPLE-DOSE Fliptop Vial - Rx only - NDC 0409-3178-02 - Contains 25 of NDC 0409-3178-17 - Lidocaine HCl 1% 300 mg/30 mL (10 mg/mL) and Epinephrine 1:100,000 Injection, USP - Protect from light ...
-
PRINCIPAL DISPLAY PANEL - 50 mL Vial Label - 317850 mL - MULTIPLE-DOSE - Lidocaine HCl 1% 500 mg/50 mL (10 mg/mL) and Epinephrine 1:100,000 - Injection, USP - For Infiltration and Nerve Block. Rx only - NOT FOR EPIDURAL OR CAUDAL USE. Distributed ...
-
PRINCIPAL DISPLAY PANEL - 50 mL Vial Tray - 317850 mL MULTIPLE-DOSE Fliptop Vial - Rx only - NDC 0409-3178-03 - Contains 25 of NDC 0409-3178-18 - Lidocaine HCl 1% 500 mg/50 mL (10 mg/mL) and Epinephrine 1:100,000 Injection, USP - Protect from light ...
-
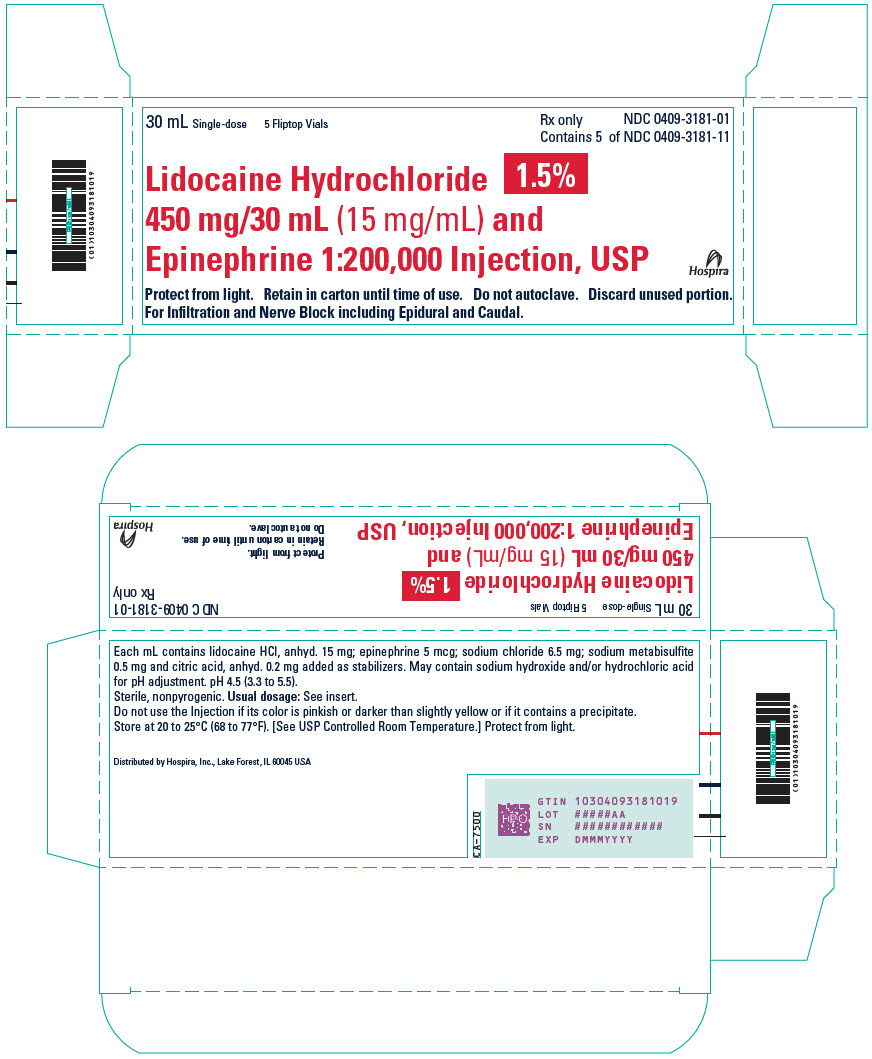
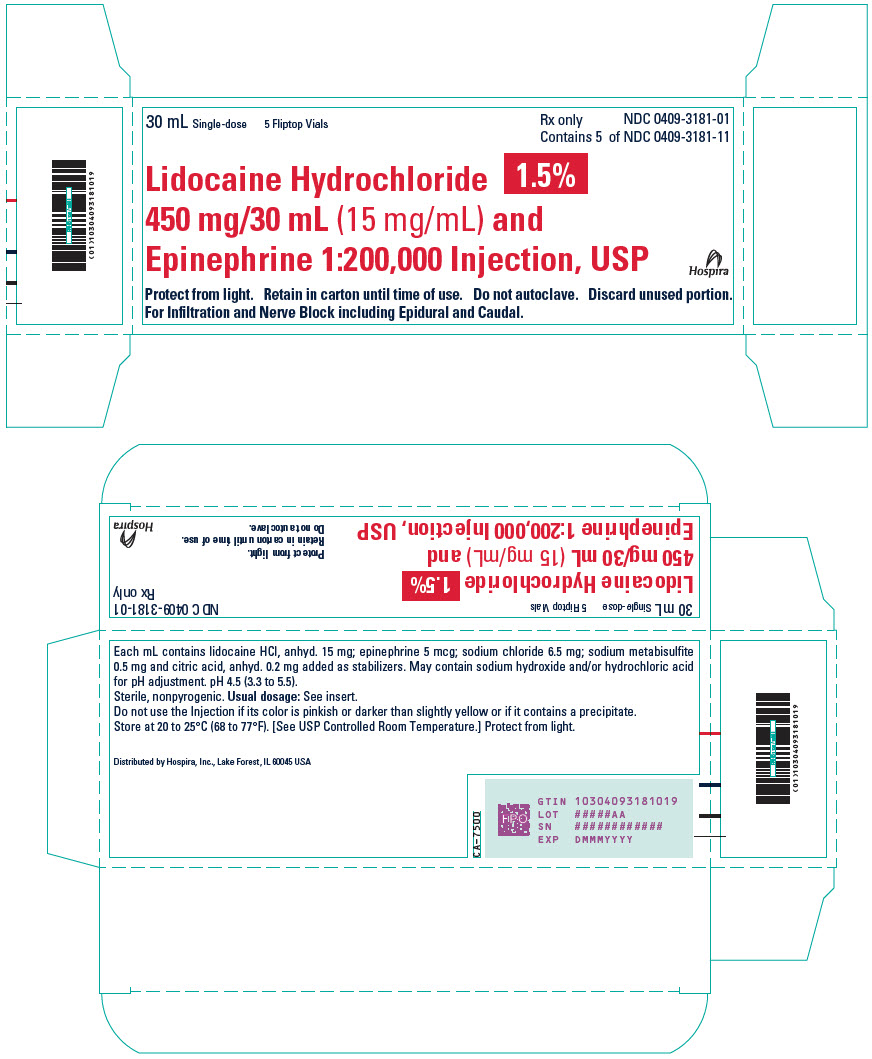
PRINCIPAL DISPLAY PANEL - 30 mL Vial Label - 318130 mL - Single-dose - LIDOCAINE HCl 1.5% and - 450 mg/30 mL (15 mg/mL) and Epinephrine 1:200,000 - Injection, USP - Rx only - For Infiltration and Nerve Block - including Epidural and Caudal. Discard unused ...
-
PRINCIPAL DISPLAY PANEL - 30 mL Vial Carton30 mL Single-dose - 5 Fliptop Vials - Rx only - NDC 0409-3181-01 - Contains 5 of NDC 0409-3181-11 - Lidocaine Hydrochloride 1.5% 450 mg/30 mL (15 mg/mL) and - Epinephrine 1:200,000 Injection ...
-
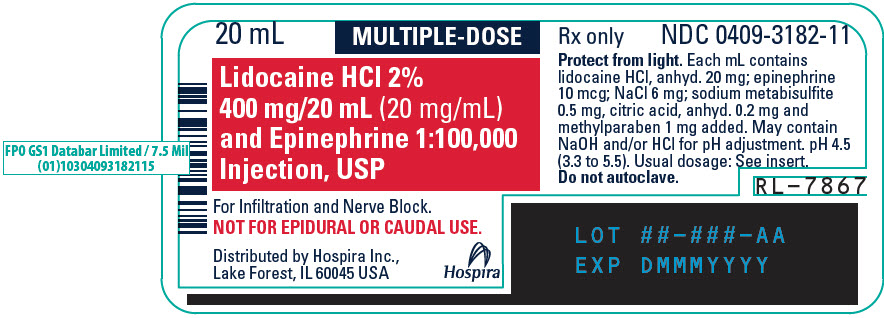
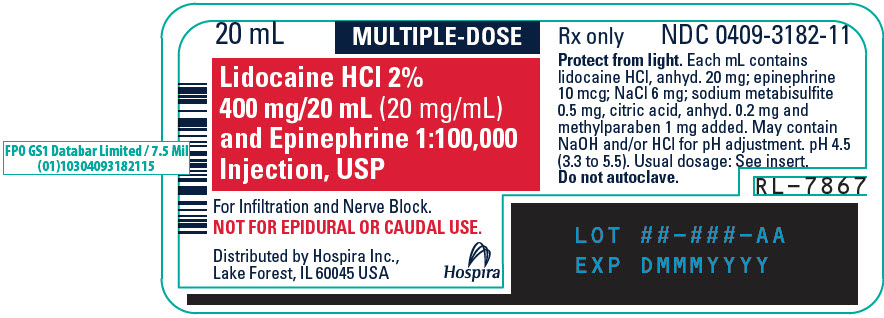
PRINCIPAL DISPLAY PANEL - 20 mL Vial Label - 318220 mL - MULTIPLE-DOSE - Lidocaine HCl 2% 400 mg/20 mL (20 mg/mL) and Epinephrine 1:100,000 - Injection, USP - For Infiltration and Nerve Block. NOT FOR EPIDURAL OR CAUDAL USE. Distributed by Hospira ...
-
PRINCIPAL DISPLAY PANEL - 20 mL Vial Tray - 318220 mL MULTIPLE-DOSE Fliptop Vial - Rx only - NDC 0409-3182-01 - Contains 25 of NDC 0409-3182-11 - Lidocaine HCl 2% 400 mg/20 mL (20 mg/mL) and Epinephrine 1:100,000 Injection, USP - Protect from light ...
-
PRINCIPAL DISPLAY PANEL - 30 mL Vial Label - 318230 mL - MULTIPLE-DOSE - Lidocaine HCl 2% 600 mg/30 mL (20 mg/mL) and Epinephrine 1:100,000 - Injection, USP - For Infiltration and Nerve Block. NOT FOR EPIDURAL OR CAUDAL USE. Rx only - Distributed ...
-
PRINCIPAL DISPLAY PANEL - 30 mL Vial Tray - 318230 mL MULTIPLE-DOSE Fliptop Vial - Rx only - NDC 0409-3182-02 - Contains 25 of NDC 0409-3182-21 - Lidocaine HCl 2% 600 mg/30 mL (20 mg/mL) and Epinephrine 1:100,000 Injection, USP - Protect from light ...
-
PRINCIPAL DISPLAY PANEL - 50 mL Vial Label - 318250 mL - MULTIPLE-DOSE - Lidocaine HCl 2% 1000 mg/50 mL (20 mg/mL) and Epinephrine 1:100,000 - Injection, USP - For Infiltration and Nerve Block. Rx only - NOT FOR EPIDURAL OR CAUDAL USE. Distributed ...
-
PRINCIPAL DISPLAY PANEL - 50 mL Vial Tray - 318250 mL MULTIPLE-DOSE Fliptop Vial - Rx only - NDC 0409-3182-03 - Contains 25 of NDC 0409-3182-31 - Lidocaine HCl 2% 1000 mg/50 mL (20 mg/mL) and Epinephrine 1:100,000 Injection, USP - Protect from light ...
-
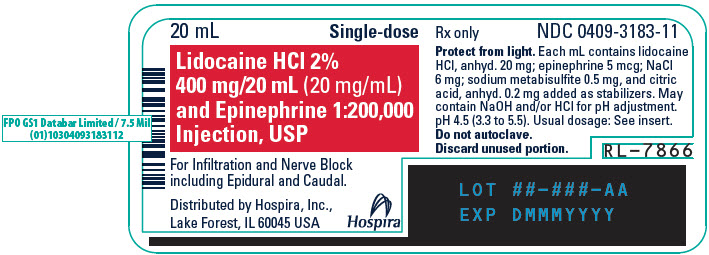
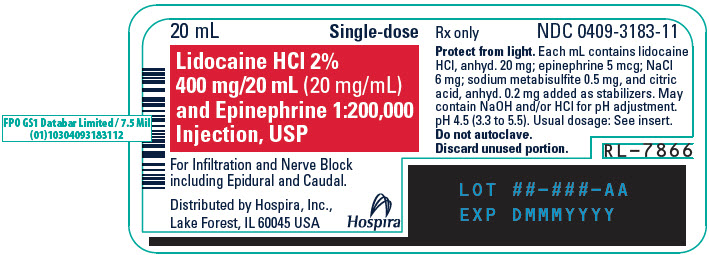
PRINCIPAL DISPLAY PANEL - 20 mL Vial Label - 318320 mL - Single-dose - Lidocaine HCl 2% 400 mg/20 mL (20 mg/mL) and Epinephrine 1:200,000 - Injection, USP - For Infiltration and Nerve Block - including Epidural and Caudal. Rx only - Distributed by Hospira ...
-
PRINCIPAL DISPLAY PANEL - 20 mL Vial Carton20 mL Single-dose - 5 Fliptop Vials - Rx only - NDC 0409-3183-01 - Contains 5 of NDC 0409-3183-11 - Lidocaine HCl 2% 400 mg/20 mL (20 mg/mL) and Epinephrine 1:200,000 Injection, USP - Protect from ...
-
PRINCIPAL DISPLAY PANEL – 20 mL vial Label - 0007 20 mL MULTIPLE-DOSE - Lidocaine HCL 1% 200 mg/20 mL - (10 mg/mL) and - Epinephrine 1:100,000 - Injection, USP - For Infiltration and Nerve Block. Rx only - NOT FOR EPIDURAL OR CAUDAL USE.
-
PRINCIPAL DISPLAY PANEL - 20 mL Vial Carton -0007 20 mL MULTIPLE-DOSE Fliptop Vial - Rx only - NDC 0409-0007-10 - Contains 10 of NDC 0409-0007-01 - Lidocaine HCl 1% 200 mg/20 mL (10 mg/mL) and Epinephrine 1:100,000 Injection, USP - Protect from light ...
-
PRINCIPAL DISPLAY PANEL – 20 mL vial Label - 0147 20 mL MULTIPLE-DOSE - Lidocaine HCl 2% 400 mg/20 mL (20 mg/mL) and Epinephrine 1:100,000 - Injection, USP - For Infiltration and Nerve Block - NOT FOR EPIDURAL OR CAUDAL USE.
-
PRINCIPAL DISPLAY PANEL – 20 mL vial Carton - 0147 20 mL MULTIPLE-DOSE Fliptop Vial - Rx only - NDC 0409-0147-10 - Contains 10 of NDC 0409-0147-01 - Lidocaine HCl 2% 400 mg/20 mL (20 mg/mL) and Epinephrine 1:100,000 Injection, USP - Protect from ...
-
INGREDIENTS AND APPEARANCEProduct Information