Label: ALBUTEROL SULFATE aerosol, metered
- NDC Code(s): 0054-0742-87
- Packager: Hikma Pharmaceuticals USA Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated January 1, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
SPL UNCLASSIFIED SECTIONFOR ORAL INHALATION ONLY - Prescribing Information
-
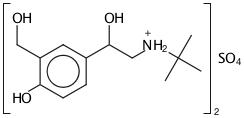
DESCRIPTION The active component of albuterol sulfate inhalation aerosol is albuterol sulfate, USP racemic α1 [(tert-Butylamino)methyl]-4-hydroxy-m-xylene-α,α'-diol sulfate (2:1)(salt), a relatively selective ...
-
CLINICAL PHARMACOLOGY Mechanism of Action - In vitro studies and in vivo pharmacologic studies have demonstrated that albuterol has a preferential effect on beta2-adrenergic receptors compared with isoproterenol ...
-
INDICATIONS AND USAGE Albuterol sulfate inhalation aerosol is indicated in adults and children 4 years of age and older for the treatment or prevention of bronchospasm with reversible obstructive airway disease and for ...
-
CONTRAINDICATIONS Albuterol sulfate inhalation aerosol is contraindicated in patients with a history of hypersensitivity to albuterol or any other albuterol sulfate inhalation aerosol component.
-
WARNINGS 1. Paradoxical Bronchospasm - Inhaled albuterol sulfate can produce paradoxical bronchospasm that may be life threatening. If paradoxical bronchospasm occurs, albuterol sulfate inhalation ...
-
PRECAUTIONS General Albuterol sulfate, as with all sympathomimetic amines, should be used with caution in patients with cardiovascular disorders, especially coronary insufficiency, cardiac arrhythmias ...
-
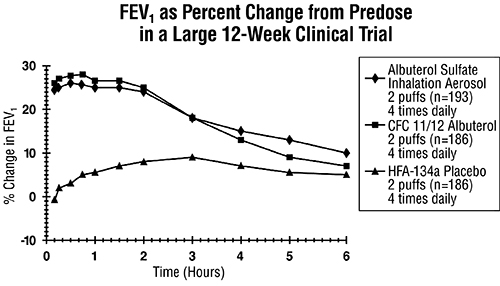
ADVERSE REACTIONS Adverse reaction information concerning albuterol sulfate inhalation aerosol is derived from a 12-week, double-blind, double-dummy study which compared albuterol sulfate inhalation aerosol, a CFC ...
-
OVERDOSAGE The expected symptoms with overdosage are those of excessive beta-adrenergic stimulation and/or occurrence or exaggeration of any of the symptoms listed under ADVERSE REACTIONS, e.g., seizures ...
-
DOSAGE AND ADMINISTRATION For treatment of acute episodes of bronchospasm or prevention of asthmatic symptoms, the usual dosage for adults and children 4 years of age and older is two inhalations repeated every 4 to 6 ...
-
HOW SUPPLIED Albuterol sulfate inhalation aerosol is supplied as a pressurized aluminum canister with an attached dose indicator, a light blue plastic actuator and dark blue dust cap each in boxes of one. Each ...
-
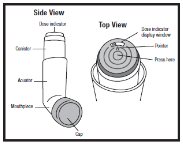
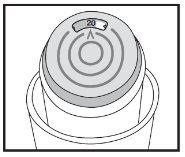
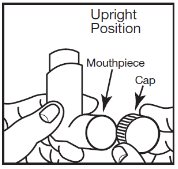
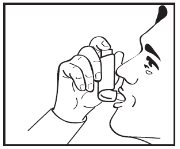
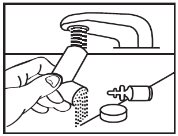
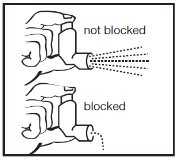
INSTRUCTIONS FOR USEAlbuterol Sulfate Inhalation AerosolWith Dose Indicator(al bue’ter ol sul’fate) Read this Instructions for Use before you start using albuterol sulfate inhalation aerosol and each time you get a refill. There may be new information. This information does not take the place of ...
-
Principal Display Panel NDC 0054-0742-87 - Albuterol Sulfate - Inhalation Aerosol - 90 mcg* With Dose Indicator - FOR ORAL INHALATION WITH - ENCLOSED ACTUATOR ONLY - 200 Metered Inhalations - Contains nochlorofluorocarbons ...
-
INGREDIENTS AND APPEARANCEProduct Information