Label: T.R.U.E. TEST THIN-LAYER RAPID USE PATCH TEST- standardized allergenic kit
- NDC Code(s): 67334-0457-1, 67334-0457-5
- Packager: SmartPractice Denmark ApS
- Category: STANDARDIZED ALLERGENIC
- DEA Schedule: None
- Marketing Status: Biologic Licensing Application
Drug Label Information
Updated November 15, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use T.R.U.E. TEST safely and effectively. See full prescribing information for T.R.U.E. TEST. T.R.U.E. TEST - Thin-Layer ...These highlights do not include all the information needed to use T.R.U.E. TEST safely and effectively. See full prescribing information for T.R.U.E. TEST.
T.R.U.E. TEST
Thin-Layer Rapid Use Epicutaneous Patch Test
For Topical Use Only
Initial U.S. Approval: 1994RECENT MAJOR CHANGES
Indications and Usage ( 1) 08/2017 INDICATIONS AND USAGE
T.R.U.E. TEST is an epicutaneous patch test indicated for use as an aid in the diagnosis of allergic contact dermatitis in persons 6 years of age and older whose history suggests sensitivity to one or more of the 35 allergens and allergen mixes included on the T.R.U.E. TEST panels. ( 1)
DOSAGE AND ADMINISTRATION
- For topical use only.
Apply the three adhesive panels of allergens and allergen mixes on healthy skin of the back. Remove panels and evaluate the skin 48 hours after application. Re-evaluate the skin 72 to 96 hours after application. ( 2)
DOSAGE FORMS AND STRENGTHS
Three adhesive panels consisting of 35 allergens and allergen mix patches and a negative control. Panel 1.3 contains 11 allergens and allergen mixes and a negative control. Panel 2.3 contains 12 allergens and allergen mixes. Panel 3.3 contains 12 allergens and allergen mixes. ( 3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- Acute allergic reactions, including anaphylaxis, may occur. ( 5.1)
- Sensitization to one or more of the allergens may occur with initial or repeat testing. ( 5.2, 5.9)
- Extreme positive reactions, excited skin syndrome, tape reactions, irritant contact dermatitis, persistent reactions, and late reactions at the test site may occur. ( 5.3, 5.4, 5.5, 5.6, 5.7, 5.8)
ADVERSE REACTIONS
- In adults 18 years of age and older, the most common (occurring in >1% of the study population) adverse reactions were burning (25.4%), tape irritation (15.8%), persistent reactions (6.8%), erythema (5.7%), and hyper/hypopigmentation (4.9%).
- In children and adolescents 6 through 17 years of age, the most common (occurring in >1% of the study population) adverse reactions were itching (up to 61.2%), tape irritation (up to 50.0%), persistent reactions (4.6%), ectopic flare of pre-existing dermatitis (12.8%), burning (up to 10.5%), skin infections (1.8%), and skin reactions near a panel site (1.4%).
To report SUSPECTED ADVERSE REACTIONS, contact SmartPractice at 1-800-878-3837 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
Patients on systemic or topical immunosuppressant therapy may have a diminished reaction to T.R.U.E. TEST. ( 7)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 8/2017
Close -
Table of ContentsTable of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Dose
2.2 Administration
2.3 Timing of Test Readings
2.4 Interpretation Instructions
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Acute Allergic Reactions
5.2 Sensitization
5.3 Extreme Positive Reactions
5.4 Excited Skin Syndrome (Angry Back)
5.5 Tape Reactions
5.6 Irritant Contact Dermatitis
5.7 Persistent Reactions
5.8 Late Reactions
5.9 Repeat Testing
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Systemic Antihistamines
7.2 Systemic Cyclosporins
7.3 Systemic Glucocorticoids
7.4 Topical Immunosuppressants and Immunomodulators
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
11 DESCRIPTION
11.1 Allergens on Panel 1.3
11.2 Allergens on Panel 2.3
11.3 Allergens on Panel 3.3
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
14 CLINICAL STUDIES
14.1 Adults
14.2 Children and Adolescents 6 through 17 Years of Age
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
SPL UNCLASSIFIED SECTIONFor topical use only.
-
1 INDICATIONS AND USAGET.R.U.E. TEST® is an epicutaneous patch test indicated for use as an aid in the diagnosis of allergic contact dermatitis (ACD) in persons 6 years of age and older whose history suggests ...
T.R.U.E. TEST® is an epicutaneous patch test indicated for use as an aid in the diagnosis of allergic contact dermatitis (ACD) in persons 6 years of age and older whose history suggests sensitivity to one or more of the 35 allergens and allergen mixes included on the T.R.U.E. TEST panels.
Close -
2 DOSAGE AND ADMINISTRATION2.1 Dose - T.R.U.E. TEST contains three adhesive panels consisting of 35 allergen and allergen mix patches and a negative control. See - Description (11)for allergen types and amounts ...
2.1 Dose
T.R.U.E. TEST contains three adhesive panels consisting of 35 allergen and allergen mix patches and a negative control. See Description (11)for allergen types and amounts.
2.2 Administration
Application Instructions
T.R.U.E. TEST should only be applied to healthy skin. Test sites should be free of scars, acne, dermatitis, or other conditions that may interfere with test result interpretation. Avoid application of T.R.U.E. TEST panels to recently tanned or sun-exposed skin because this may increase the risk of false negatives. Avoid patch testing on patients for three (3) weeks after ultraviolet (UV) treatments, heavy sun, or tanning bed exposure. Avoid using alcohol or other irritating substances on the skin prior to testing. Avoid excessive sweating during the testing period to maintain sufficient adhesion to the skin. Avoid excessive physical activity to maintain sufficient adhesion and to prevent actual loss of patch test material. Avoid getting the panels and surrounding area wet. If excessive body hair exists at the test site, remove with an electric shaver (do not use razors). Very oily skin may be cleaned with mild soap and water prior to testing.
T.R.U.E. Test panels should be applied as follows:
Figure 1
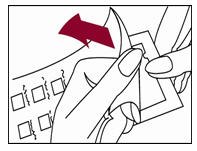
Figure 2
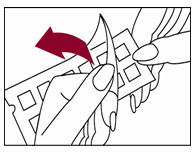
Figure 3
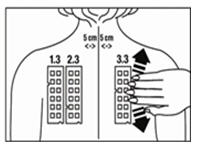
Figure 4
1. Peel open the package and remove the test panel (Figure 1).
2. Remove the protective plastic covering from the test surface of the panel (Figure 2). Be careful not to touch the test allergens or allergen mixes.
3. Position test Panel 1.3 on the patient's back as shown in Figure 3. Allergen number 1 should be in the upper left corner. Avoid applying the panel on the margin of the scapula or directly over the midline of the spine. Ensure that each patch of the allergen panel is in contact with the skin by smoothing the panel outward from the center to the edge (as illustrated for Panel 3.3 in Figure 3).
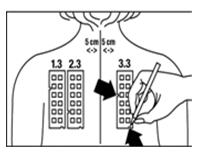
4. With a medical marking pen, indicate on the skin the location of the two notches on the panel (as illustrated for Panel 3.3 in Figure 4).
5. Repeat the process with test Panel 2.3. Position the test Panel 2.3 beside Panel 1.3, on the left side of the patient's back so that the number 13 allergen is in the upper left corner. Apply test Panel 2.3 five (5) cm from the midline of the spine (Figure 3).
6. Repeat the process with Panel 3.3 positioning the panel on the right side of the patient's back so that the number 25 allergen is in the upper left corner. Apply test Panel 3.3 five (5) cm from the midline of the spine. (Figure 3)
7. If needed, hypoallergenic surgical tape, appropriate for patch testing, may be used for increased adhesion around the outside edges of the panels.
2.3 Timing of Test Readings
Schedule patients to return approximately 48 hours after patch test application to have the panels removed. Prior to removal of the panels, use a medical marking pen to remark the notches found on the panels. The patch test reaction on the patient's skin may be evaluated at 48 hours, but an additional reading(s) at 72 and/or 96 hours is necessary. Late positive reactions may occur 7 to 21 days after application of the panels. [see Warnings and Precautions (5.2)].
Close2.4 Interpretation Instructions
An identification template is provided for each of the three (3) panels for quick identification of any allergen that causes a reaction. To assure correct positioning, marks on the skin made with the medical marking pen should correlate with the notches on the template. The interpretation method, similar to the one recommended by the International Contact Dermatitis Research Group, is as follows:
? Doubtful reaction: faint macular erythema only + Weak positive reaction: non-vesicular with erythema, infiltration, possibly papules ++ Strong positive reaction: vesicular, erythema, infiltration, papules +++ Extreme positive reaction: bullous or ulcerative reaction - Negative reaction IR Irritant reaction: Pustules as well as patchy follicular or homogeneous erythema without infiltrations are usually signs of irritation and do not indicate allergy. Itching is a subjective symptom that is expected to accompany a positive reaction.
False Negatives
False negative results may be due to insufficient patch contact with the skin and/or premature evaluation of the test. Repeat testing may be indicated. The effect of repetitive testing with T.R.U.E. TEST is unknown [see Warnings and Precautions (5.9)].
False Positives
A false positive result may occur when an irritant reaction cannot be differentiated from an allergic reaction. A positive test reaction should meet the criteria for an allergic reaction. If an irritant reaction cannot be distinguished from a true positive reaction or if a doubtful reaction is present, a retest may be considered. The effect of repetitive testing with T.R.U.E. TEST is unknown [see Warnings and Precautions (5.9)].
-
3 DOSAGE FORMS AND STRENGTHST.R.U.E. TEST contains three (3) adhesive panels consisting of 35 allergen and allergen mix patches and a negative control. Panel 1.3 contains 11 allergens and allergen mixes and a negative ...
T.R.U.E. TEST contains three (3) adhesive panels consisting of 35 allergen and allergen mix patches and a negative control. Panel 1.3 contains 11 allergens and allergen mixes and a negative control. Panel 2.3 contains 12 allergens and allergen mixes. Panel 3.3 contains 12 allergens and allergen mixes. See Description (11)for allergen types and amounts.
Close -
4 CONTRAINDICATIONSDo not apply to skin of patients with a history of severe allergic reaction (systemic and/or local) to any of the allergen components or inactive substances of T.R.U.E. TEST [see - Description ...
Do not apply to skin of patients with a history of severe allergic reaction (systemic and/or local) to any of the allergen components or inactive substances of T.R.U.E. TEST [see Description (11)].
Do not apply to skin that is injured or inflamed.
Close -
5 WARNINGS AND PRECAUTIONS5.1 Acute Allergic Reactions - Acute allergic reactions, including anaphylaxis, may occur following the application of T.R.U.E. TEST. If a severe allergic reaction occurs, remove the T.R.U.E ...
5.1 Acute Allergic Reactions
Acute allergic reactions, including anaphylaxis, may occur following the application of T.R.U.E. TEST. If a severe allergic reaction occurs, remove the T.R.U.E. TEST panel(s) and initiate appropriate medical treatment. Immediate contact urticaria may present within minutes to an hour after application in patients who are pre-sensitized to some allergens and may be local or generalized. Patients may be advised to remove the panels themselves if they experience systemic symptoms [see Patient Counseling Information (17)].
5.2 Sensitization
A negative patch test reaction, followed by a positive reaction 10 to 20 days after panel application, may indicate active sensitization. Active sensitization is confirmed upon retesting with a positive reaction occurring at the 72 and/or the 96 hour reading. If patients undergo a second series of patch tests immediately, select a new test site for T.R.U.E. TEST application. Alternatively, the same site may be retested after a 3-week clearing period, provided the site remains free of conditions that might affect test results [see Dosage and Administration (2.1)]. The safety and effectiveness of repetitive testing with T.R.U.E. TEST is unknown [see Warnings and Precautions (5.9)].
5.3 Extreme Positive Reactions
Extreme positive (+++) reactions that are bullous or ulcerative with pronounced erythema, infiltration, and coalescing vesicles may present in extremely sensitive patients [see Dosage and Administration (2.3)].
5.4 Excited Skin Syndrome (Angry Back)
Excited skin syndrome is a regional state of skin hyper-reactivity caused by the presence of a strong positive reaction which may result in other patch test sites to become reactive.
5.5 Tape Reactions
Reactions to the T.R.U.E. TEST tape or adhesive may occur. T.R.U.E. TEST panel tape and the individual patches are composed of polyester. The adhesive used in the panels is acrylate-based and processed to remove free monomers that may be allergenic [see Description (11)].
5.6 Irritant Contact Dermatitis
Patients may experience irritant contact dermatitis upon exposure to any of the allergens contained within T.R.U.E. TEST that cause direct damage to the skin at the test site. Recurrence of an irritant response is not limited to exposure to the specific allergens or allergen mixes, but may follow exposure to any chemical irritants.
5.7 Persistent Reactions
Positive reactions may persist from 7 days to months after panel application.
Close5.9 Repeat Testing
The safety and efficacy of repetitive testing with T.R.U.E. TEST is unknown. Sensitization or increased reactivity to one or more of the allergens may occur [see Warnings and Precautions (5.2)]. If patients undergo a second series of patch tests immediately, select a new test site for T.R.U.E. TEST application. Alternatively, the same site may be retested after a 3-week clearing period, provided the site remains free of conditions that might affect test results. [see Dosage and Administration (2.1)].
-
6 ADVERSE REACTIONSIn adults 18 years of age and older, the most common (occurring in >1% of the study population) adverse reactions were burning (25.4%), tape irritation (15.8%), persistent reactions (6.8%) ...
In adults 18 years of age and older, the most common (occurring in >1% of the study population) adverse reactions were burning (25.4%), tape irritation (15.8%), persistent reactions (6.8%), erythema (5.7%), and hyper/hypopigmentation (4.9%). In children and adolescents 6 through 17 years of age, the most common (occurring in >1% of the study population) adverse reactions were itching (up to 61.2%), tape irritation (up to 50.0%), persistent reactions (4.6%), ectopic flare of pre-existing dermatitis (12.8%), burning (up to 10.5%), skin infections (1.8%), and skin reactions near a panel site (1.4%).
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug may not reflect the rates observed in clinical practice.
Adult Subjects 18 Years of Age and Older
Table 1 presents a summary of ten clinical trials conducted in adults in North America and Europe using T.R.U.E. TEST.
Table 1- Overview of Clinical Studies using T.R.U.E. TEST Among Adults 18 Years of Age and Older Clinical Study Overview Study 1 Study 2 Study 3 Study 4 Study 5 Study 6 Study 7 Study 8 Study 9 Study 10 Total N 127 121 119 50 130 128 200 235 49 9 1168 Age Range (years) 19-79 18-77 19-76 19-82 19-86 20-83 19-78 18-85 18-68 36-76 18-86 Sex (% female) 68% 68% 73% 72% 68% 63% 59% 71% 98% 78% 69% Ethnicity Caucasian 86% 88% 83% 92% 85% 88% 97% 91% 98% 100% 83% Black 9% 12% 11% 4% 13% 12% 0% 5% 0% 0% 9% Other 5% 1% 6% 4% 2% 0.8% 3% 4% 2% 0% 7% Allergens Nickel sulfate X X X X Wool alcohols X X X Neomycin sulfate X X X X Potassium dichromate X X X X Caine Mix X X X X Fragrance Mix X X X X Colophony X X X X Paraben Mix X X X Balsam of Peru X X X X Ethylenediamine dihydrochloride X X X X Cobalt dichloride X X X X p-tert-Butylphenol formaldehyde resin X X X Epoxy resin X X X X Carba mix X X X Black rubber mix X X X Cl+ Me- Isothiazolinone X X X Quaternium-15 X X X X Methyldibromo glutaronitrile X p-Phenylenediamine X X X X Formaldehyde X X Mercapto Mix X X X Thimerosal X X X Thiuram Mix X X X X Diazolidinyl urea X Quinoline Mix X X X Tixocortol-21-pivalate X X Gold Sodium Thiosulfate X Imidazolidinyl urea X Budesonide X X Hydrocortisone-17-butyrate X X Mercaptobenzothiazole X X X Bacitracin X Parthenolide X Disperse blue 106 X Bronopol X Table 2 summarizes the adverse reactions recorded in ten clinical studies. Subjects' adverse reactions were recorded on case report forms by study personnel. Adverse reactions were recorded during subject follow-up visits, which varied between 24 and/or 96 hours and/or Day 21 [see Clinical Studies (14)].
Table 2- Summary of Adverse Reactions Reported Among Adult Subjects 18 Years of Age and Older Study 1 Study 2 Study 3 Study 4 Study 5 Study 6 Study 7 Study 8 Study 9 Study 10 Total (%) ND=Not Done N 127 121 119 50 130 128 200 235 49 9 1168 Burning * 12 6 9 16 50 51 25 123 5 0 297 (25.4) Tape Irritation * 4 0 2 1 22 51 25 79 0 0 184 (15.8) Persistent Reactions † 11 0 0 8 2 16 1 41 0 0 79 (6.8) Erythema † 0 3 27 ‡ 2 0 3 0 32 0 0 67 (5.7) Hyper/Hypopigmentation † 11 2 8 ‡ 6 1 7 0 14 8 0 57 (4.9) Sensitization (possible) † 0 2 5 ‡ 1 0 0 0 ND ND ND 8 (0.9) § Late Reaction † 0 0 0 1 0 0 0 7 0 0 8 (0.7) Scarring † 0 0 2 ‡ ND ND ND ND ND ND ND 2 (0.5) § Ectopic Flare † ND ND ND ND 1 0 1 ND ND ND 2(0.4) § Panel Adhesion:Problems with panel adhesion were observed during some of the clinical studies. Poor panel adhesion was defined as any panel that fell off prior to the 48-hour removal time, any test panel that was not in good contact with the skin, or if one or more of the patch test allergens were not in good contact with the skin as evidenced at the time of panel removal, 48 hours. If the panel fell off the back prior to the 48-hour removal time frame, the subject was excluded from the efficacy calculations (sensitivity and specificity) but not from the safety analysis. Over all studies, poor panel adhesion occurred 49 times (4.2%) (Table 3). In study 2, the poor adhesion was attributed to the particular lot of adhesive used to manufacture the clinical test tape.
Table 3- Incidence and Proportion of Poor Panel Adhesion Among Subjects 18 Years of Age and Older Study 1 Study 2 Study 3 Study 4 Study 5 Study 6 Study 7 Study 8 Study 9 Study 10 Total N 127 121 119 50 130 128 200 235 49 9 1168 Poor Adhesion
(%)0 (0.0) 14 (11.6) 2 (1.7) 5 (10.0) 1 (0.8) 1 (0.8) 0 (0.0) 10 (4.3) 12 (23.5) 0 (0.0) 49 (4.2) Children and Adolescents 6 through 17 Years of Age
The safety of T.R.U.E. TEST in children and adolescents 6 through 17 years of age was evaluated in two open-label studies conducted in the US. In these studies, children and adolescents with suspected allergic contact dermatitis had three T.R.U.E. TEST panels applied to their backs and upper arms by investigators. Some of the panels were from previously approved versions of T.R.U.E. TEST. Subjects were instructed to keep the panels in place for 48 hours. Subjects were monitored for safety for 21 days after application of T.R.U.E. TEST panels. The safety monitoring plan included investigator's assessment of panel adhesion, tape irritation, and participant reporting of burning and itching (as a combined symptom) when panels were removed 2 days after application of T.R.U.E. TEST. Surveillance for late reactions, possible sensitization and persistent reactions occurred at Day 7 and 21. Unsolicited adverse events, serious adverse events and deaths were monitored for 21 days after patch application.
Pediatric Study 1 (NCT: 00795951)
In this prospective, single-center, open label study, 102 subjects were enrolled to evaluate the safety of T.R.U.E. TEST [see Clinical Studies (14.2)Table 7 for a list of the allergens and allergen mixes]. Of enrolled subjects, 52% were female, 39.2% were White, 31.4% were Hispanic, 6.9% were Black, 12.7% were Asian and 10.5% were of other racial/ethnic groups. The mean age of subjects was 11.6 years.
Pediatric Study 2 (NCT: 01797562)
In a prospective, multi-center, open-label study conducted in the US, up to 116 children and adolescents were enrolled to evaluate the safety of T.R.U.E. TEST [see Clinical Studies (14.2) Table 7for a list of the allergens and allergen mixes]. Of enrolled subjects, 69% were female, 37.9% were Hispanic or Latino, 28.5% were White, 11.2% were Asian, 6.0% were Black, and 16.4% were of other racial/ethnic groups. The mean age of subjects was 12.6 years. Table 4 summarizes adverse reactions occurring within 21 days after T.R.U.E. TEST application.
Table 4: Pediatric Study 2 *: Adverse Reactions Occurring 2 Days after T.R.U.E. TEST Application in Children and Adolescents 6 through 17 Years of Age Adverse Reactions Any
n (%)Severe
n (%)Panel 1.3
N=54 †Panel 2.3
N=114 ‡Panel 3.3
N=114Panel 1.3
N=54Panel 2.3
N=114Panel 3.3
N=114- *
- NCT: 01797562
- †
- Fifty five subjects received Panel 1.3 and 61 subjects received Panel 1.2, which included 4 allergens with outdated formulations. Fifty four of the 55 subjects presented on day 2 as scheduled.
- ‡
- Of the 116 subjects who received T.R.U.E. TEST, 114 presented to visit 2 and had itching, burning, and tape irritation data documented.
- §
- Itching and burning were graded as none, mild/weak (minimal discomfort), moderate (definite discomfort), or severe (significantly bothersome, possible interference with sleep or daily activity).
- ¶
- Tape irritation was graded at Day 2 by investigators using a 4-point scale, including none, weak (faint to definite pink erythema), moderate (moderate erythema, definite redness), or severe (severe erythema, very intense redness).
Itching § 31 (57.4) 62 (54.4) 72 (63.2) 1 (1.9) 7 (6.1) 11 (9.7) Burning § 3 (5.6) 7 (6.1) 12 (10.5) 0 (0.0) 0 (0.0) 1 (0.9) Tape irritation ¶ 27 (50.0) 56 (49.1) 53 (46.5) 0 (0.0) 1 (0.9) 0 (0.0) Across both pediatric studies (N=218), extreme positive reactions (+++, indicating a bullous or ulcerative reaction with pronounced erythema, infiltration, and coalescing vesicles) occurred in two subjects. Both extreme positive reactions occurred in response to metal allergens (nickel sulfate and gold sodium thiosulfate) by Day 3 and resolved by Day 21. Late positive reactions occurred in 2 subjects (0.9%) 21 days after T.R.U.E. TEST application to the following allergen: gold sodium thiosulfate (n=2). Persistent reactions occurred in 10 subjects (4.6%) 21 days after T.R.U.E. TEST application to the following allergens: bronopol (n=1), Cl+Me+isothiazolinone (n=1), diazolidinyl urea (n=1), gold sodium thiosulfate (n=6), nickel sulfate (n=2), and quaternium-15 (n=1). Ectopic flare of pre-existing dermatitis occurred in 28 (12.8%) of subjects. Of these cases, 1 (0.5%) was severe and 3 (1.4%) were complicated by skin infection. Skin reactions near a panel site were observed in 3 subjects (1.4%). No serious adverse events or deaths considered related to T.R.U.E. TEST occurred.
Close6.2 Postmarketing Experience
The following additional adverse reactions have been identified during post-approval use of T.R.U.E. TEST. Because these reactions are continuously reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to T.R.U.E. TEST exposure.
- Acute allergic reactions [see Warnings and Precautions (5.1)]
- Extreme positive reactions [see Warnings and Precautions (5.3)]
- Excited skin syndrome (Angry back) [see Warnings and Precautions (5.4)]
- Irritant contact dermatitis [see Warnings and Precautions (5.6)]
-
7 DRUG INTERACTIONS7.1 Systemic Antihistamines - The effect of concomitant systemic antihistamine administration on the performance of patch testing with T.R.U.E. TEST is unknown. 7.2 Systemic Cyclosporins - The ...
7.1 Systemic Antihistamines
The effect of concomitant systemic antihistamine administration on the performance of patch testing with T.R.U.E. TEST is unknown.
7.2 Systemic Cyclosporins
The effect of concomitant or prior systemic cyclosporin administration on the performance of patch testing with T.R.U.E. TEST is unknown.
7.3 Systemic Glucocorticoids
Oral steroids may cause false negative results of patch testing with T.R.U.E. TEST. The risk of discontinuing or decreasing the dose of oral corticosteroids in order to perform the patch test must be weighed against the benefits of patch testing.
Close7.4 Topical Immunosuppressants and Immunomodulators
Avoid using test sites to which topical glucocorticoids, antihistamines, immunosuppressants, or immunomodulators are applied. The use of topical steroids or immunosuppressants at or near potential test sites should be avoided from at least one week prior to patch testing through the conclusion of patch testing.
-
8 USE IN SPECIFIC POPULATIONS8.1 Pregnancy - Risk Summary - All pregnancies have a risk of birth defect, loss, or other adverse outcomes. In the US general population, the estimated background risk of major birth defects ...
8.1 Pregnancy
Risk Summary
All pregnancies have a risk of birth defect, loss, or other adverse outcomes. In the US general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively. There are no human or animal data to establish the presence or absence of T.R.U.E. TEST-associated risks during pregnancy.
8.2 Lactation
Risk Summary
It is not known whether the allergens in T.R.U.E. TEST are excreted in human milk. Data are not available to assess the effects of T.R.U.E. TEST on the breastfed child or on milk production/excretion. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for T.R.U.E. TEST and any potential adverse effects on the breastfed child from T.R.U.E. TEST or from the underlying maternal condition.
8.4 Pediatric Use
Safety and effectiveness of T.R.U.E. TEST have not been established in persons younger than 6 years of age.
Close8.5 Geriatric Use
Clinical studies of T.R.U.E. TEST did not include sufficient numbers of subjects aged 65 years and over to determine whether they respond differently from younger subjects.
-
11 DESCRIPTIONThin-layer Rapid Use Epicutaneous Patch Test, T.R.U.E. TEST - ,is a ready-to-use allergen patch test system consisting of 35 allergen and allergen mix patches, containing 58 allergenic ...
Thin-layer Rapid Use Epicutaneous Patch Test, T.R.U.E. TEST ,is a ready-to-use allergen patch test system consisting of 35 allergen and allergen mix patches, containing 58 allergenic substances, and a negative control.
Each test consists of the following:
Panel-The panel consists of three pieces of surgical tape (5.2 × 13.0 cm), each with 12 polyester patches of approximately 0.81 cm 2. Each patch is coated with a film containing a uniformly dispersed specific allergen or allergen mix. The negative control, located on Panel 1.3, is an uncoated polyester patch.
Tape-The panel tape is composed of polyester. The adhesive used in the panels is acrylate-based. There is no natural rubber latex, rubber components, balsams or rosins in the adhesive or tape. Acrylate adhesives are processed to remove free monomers that may be allergenic.
Foil Pouch-Each test panel is covered by a protective sheet and sealed in a pouch of laminated foil.
Inactive Ingredients-The allergens are homogenized in one or more of the following materials to produce the allergen films that coat the patches: hydroxypropyl cellulose (HPC), povidone (PVP), povidone with butylhydroxyanisole (BHA) and butylhydroxytoluene (BHT), povidone with sodium bicarbonate and sodium carbonate (PSBSC), and hydroxypropylcellulose with β-cyclodextrin (HPCβ).
The individual components of T.R.U.E. TEST Panels 1.3, 2.3, and 3.3 are listed below along with a quantitative description of each patch formulation. Panel 1.3 contains 11 allergens or allergen mixes and a negative control, Panel 2.3 contains 12 allergens or allergen mixes, and Panel 3.3 contains 12 allergens or allergen mixes.
11.1 Allergens on Panel 1.3
Nickel Sulfate (Position 1):
Nickel sulfate hexahydrate (purity ≥98.5%) is used to formulate this patch. The active allergenic component is nickel. The gel vehicle is hydroxypropyl cellulose. The product is formulated to contain 200 mcg/cm 2of nickel sulfate hexahydrate, which corresponds to 36 mcg of nickel per patch. Nickel is one of the most common metals in the environment and is found in most metal and metal-plated objects.
Wool Alcohols (Lanolin) (Position 2):
Wool alcohols, USP are a natural product obtained from the fleece of sheep. This allergen is a highly complex mixture of alcohols containing cholesterol, lanosterol, agnosterol, and their dihydro derivatives as well as straight and branched chain aliphatic alcohols. The active allergenic component has not been identified. The gel vehicle is povidone. The product is formulated to contain 1000 mcg/cm 2of wool alcohols, which corresponds to 810 mcg of wool alcohols per patch. Wool alcohols are a common constituent of many ointments, creams, lotions, and soaps.
Neomycin Sulfate (Position 3):
Neomycin sulfate, USP an antibiotic drug substance, is used to formulate this patch. The gel vehicle is povidone. The product is formulated to contain 600 mcg/cm 2of neomycin sulfate, which corresponds to 486 mcg of neomycin sulfate per patch. Neomycin is a common antibiotic found in topical antibiotic creams, lotions, ointments, eye drops, and ear drops.
Potassium Dichromate (Position 4):
Potassium dichromate (purity ≥98.5%) is used to formulate this patch. The active allergenic component is chromium. The gel vehicle is povidone. The product is formulated to contain 54 mcg/cm 2of potassium dichromate, which corresponds to 15.7 mcg of chromium per patch. Chromium is found in cement, as well as in many industrial chemicals.
Caine Mix (Position 5):
Caine mix is composed of three drug substances: benzocaine, USP (purity ≥98.0); tetracaine hydrochloride, USP (purity ≥98.5); and dibucaine hydrochloride, USP (purity ≥97.0). The gel vehicle is povidone. The product is formulated to contain 630 mcg/cm 2of caine mix, which corresponds to 510 mcg of caine mix per patch (378 mcg of benzocaine, 66 mcg of tetracaine, and 66 mcg of dibucaine). Benzocaine, tetracaine, and dibucaine are found in many topical anesthetic medications.
Fragrance Mix (Position 6):
Fragrance mix is composed of eight substances: geraniol (purity ≥95%, identity of impurities unknown); cinnamaldehyde (purity ≥95%, contains trace amounts of cinnamyl alcohol); hydroxycitronellal (purity ≥95%, identity of impurities unknown); cinnamyl alcohol (purity ≥95%, identity of impurities unknown); eugenol, USP (purity ≥95%, identity of impurities unknown); isoeugenol (purity ≥88%, identity of impurities unknown); α-amylcinnamaldehyde (purity ≥90%, identity of impurities unknown); and oak moss. Oak moss, a dark green sticky paste, is a solvent extract of the lichen Evernia prunastri. The chemical composition is very complex. The acid fraction (95% of the extracted material) is made up of depsides including atranorin, evernic acid, usnic acid, chloratranorin, and degradation products of these depsides. Atranorin is suspected as a prime allergenic component, and its peak (measured with gas chromatography) is used to determine the amount of oak moss in the fragrance mix patch. The gel vehicles used in this patch are povidone and β-cyclodextrin. The product is formulated to contain 500 mcg/cm 2of fragrance mix, which corresponds to 405 mcg of fragrance mix per patch (approximately 81 mcg of geraniol, approximately 41 mcg of cinnamaldehyde, approximately 63 mcg of hydroxycitronellal, approximately 63 mcg of cinnamyl alcohol, approximately 41 mcg of eugenol, approximately 17 mcg of isoeugenol, approximately 17 mcg of α-amylcinnamaldehyde, and approximately 81 mcg of oak moss). The components of fragrance mix are commonly used in toiletries, fragrances, and flavorings.
Colophony (Position 7):
Colophony is produced from the resin of the pine trees Pinus massoniana and Pinus tabuliformis. It is translucent, pale yellow or brownish yellow, brittle, and glassy in appearance. Colophony consists of 75% to 85% resin acids, 10% neutral fractions (i.e., terpenes), with the remaining part oxidation products. Oxidation products of abietic acid and other resin acids have been identified as the active allergenic components. The ultraviolet absorbance measurement of one of the primary components, abietic acid, is used to quantify colophony. The gel vehicle is povidone. BHA and BHT are added in equal amounts of 11 mcg per patch as antioxidants. The product is formulated to contain 1200 mcg/cm 2of colophony, which corresponds to 972 mcg of colophony per patch. Colophony is found in adhesives, sealants, and pine oil cleaners.
Paraben Mix (Position 8):
Paraben mix contains the five ester derivatives of parahydroxybenzoic acid: methyl, USP; ethyl, USP; propyl, USP; butyl, USP; and benzyl parahydroxybenzoate in equal parts (purity of each derivative ≥ 98.0%). The gel vehicle is povidone. The product is formulated to contain 1000 mcg/cm 2of paraben mix, which corresponds to 810 mcg of paraben mix per patch. The components of paraben mix can be found in cosmetics, dermatological creams, and paste bandages.
Balsam of Peru (Position 10):
Balsam of peru is a resin from a South American tree, Myroxylon balsamum pereirae. The resin consists of a mixture of fragrances and other substances that have not all been identified. Balsam of peru patch content is measured by gas chromatography of its two major constituents, benzyl cinnamate and benzyl benzoate. Several components of Balsam of peru have been identified as allergens, including cinnamic acid, benzyl alcohol, and vanillin. The gel vehicle is povidone. This patch is formulated to contain 800 mcg/cm 2of Balsam of peru resin, which corresponds to 648 mcg of Balsam of peru resin per patch. This resin is found in many cosmetics and perfumes and is also used as a flavoring agent in cough syrups, lozenges, chewing gum, and candies.
Ethylenediamine Dihydrochloride (Position 11):
Ethylenediamine dihydrochloride (purity ≥98.5%) is used to formulate this patch. The active allergenic component is ethylenediamine. The gel vehicle is povidone. The product is formulated to contain 50 mcg/cm 2of ethylenediamine dihydrochloride, which corresponds to 18 mcg of ethylenediamine per patch. Ethylenediamine is used as a stabilizer, emulsifier, and preservative in topical fungicides, antibiotic creams, eye drops, and nose drops.
Cobalt Dichloride (Position 12):
Cobalt dichloride hexahydrate (purity ≥98.5%) is used to formulate this patch. The active allergenic component is cobalt. The gel vehicle is hydroxypropyl cellulose. The product is formulated to contain 20 mcg/cm 2of cobalt dichloride hexahydrate, which corresponds to 4 mcg of cobalt per patch. Cobalt is found in metal-plated objects and costume jewelry.
11.2 Allergens on Panel 2.3
p-tert-Butylphenol Formaldehyde Resin (Position 13):
p-tert-Butylphenol formaldehyde resin (purity ≥95%) is used to formulate this patch. The active allergenic components have been identified as p-tert-butylphenol formaldehyde and numerous other compounds. The gel vehicle is hydroxypropyl cellulose. The product is formulated to contain 45 mcg/cm 2of p-tert-butylphenol formaldehyde resin, which corresponds to 36 mcg of p-tert-butylphenol formaldehyde resin per patch. This resin is found in many waterproof glues used in the leather goods, furniture, and shoe industries.
Epoxy Resin (Position 14):
Epoxy resin, a clear viscous liquid, is used to formulate this patch. It consists of 75% to 85% diglycidylether of bisphenol A, the active allergenic component, which is a monomer used for the preparation of polymer epoxy resins. The remaining part consists of the dimer and the trimer. The gel vehicle is hydroxypropyl cellulose. This patch is formulated to contain 50 mcg/cm 2of epoxy resin, which corresponds to 32 mcg of diglycidylether of bisphenol A per patch. This resin is found in adhesives, surface coatings, and paints.
Carba Mix (Position 15):
Carba mix contains three chemicals used to stabilize rubber products: diphenylguanidine (purity ≥96%), zincdibutyldithiocarbamate (purity ≥96%), and zincdiethyldithiocarbamate (purity ≥96%) in equal parts. The gel vehicle is hydroxypropyl cellulose. The product is formulated to contain 250 mcg/cm 2of carba mix, which corresponds to 203 mcg of carba mix per patch. These chemical stabilizers and accelerators are found in many rubber products, pesticides, and some glues.
Black Rubber Mix (Position 16):
Black rubber mix contains the antioxidant and antiozonate chemicals N-isopropyl-N'-phenyl paraphenylenediamine (purity ≥95%), N-cyclohexyl-N'-phenyl paraphenylenediamine (purity ≥90%), and N, N'-diphenyl paraphenylenediamine (purity ≥90%) in the ratio 2:5:5. The gel vehicle is povidone. The product is formulated to contain 75 mcg/cm 2of black rubber mix, which corresponds to 61 mcg of black rubber mix per patch. The components of black rubber mix are found in almost all black rubber products, such as tires, handles, and hoses.
Cl+ Me- Isothiazolinone (MCI/MI) (Position 17):
Cl+ Me- Isothiazolinone is an antibacterial preservative that consists of two active ingredients, 5-chloro-2-methyl-4-isothiazolin-3-one (1.05% to 1.25% w/w) and 2-methyl-4-isothiazolin-3-one (0.25% to 0.40% w/w) in a 3:1 ratio at a concentration of 1.5% in aqueous magnesium salts. The gel vehicle is povidone. The product is formulated to contain 4 mcg/cm 2of Cl+ Me- iso-thiazolinone, which corresponds to 3 mcg of Cl+ Me- isothiazolinone per patch. This preservative is found in many shampoos, creams, lotions, and other skin care products.
Quaternium-15 (Q-15) (Position 18):
Quaternium-15, 1-(3-chloroallyl)-3,5,7,-triaza-1-azonium-adamantane chloride (purity ≥94%), is a preservative. The gel vehicle is hydroxypropyl cellulose. The product is formulated to contain 100 mcg/cm 2of Quaternium-15, which corresponds to 81 mcg of Quaternium-15 per patch. This preservative is found in creams, lotions, shampoos, soaps, and other cosmetics and skin care products.
Methyldibromo Glutaronitrile (MDBGN) (Position 19):
Methyldibromo Glutaronitrile, 1,2-Dibromo-2,4-dicyanobutane (purity ≥95%), is a component of the preservative Euxyl K400. The gel vehicle is povidone. The patch is formulated to contain 5 mcg/cm 2of methyldibromo glutaronitrile, which corresponds to 4 mcg of methyldibromo glutaronitrile per patch. Methyldibromo glutaronitrile is commonly used in cosmetic and personal care products such as body creams, facial and hand lotions, sun screens, baby lotions, shower gels, ultrasonic gel, toilet paper, shampoos, and massage oils. It is also found in cutting oils, drilling oils, glues, and coolants.
p-Phenylenediamine (Position 20):
p-Phenylenediamine (purity ≥97.5%), a blue-black aniline dye, is used to formulate this patch. The gel vehicle is povidone. The product is formulated to contain 80 mcg/cm 2of p-phenylenediamine, which corresponds to 65 mcg of p-phenylenediamine per patch. This dye is found most often in permanent and semipermanent hair dyes.
Formaldehyde (Position 21):
Formaldehyde is released from the proallergen N-hydroxymethyl succinimide, which is cleaved into succinimide and formaldehyde when it comes in contact with the transepidermal water on the surface of the skin. Formaldehyde is the active allergenic compound. The content of formaldehyde in the proallergen is 22.1% to 24.1%. The gel vehicle is povidone with sodium bicarbonate and sodium carbonate. The product is formulated to contain 180 mcg/cm 2of formaldehyde, which corresponds to 146 mcg of formaldehyde per patch. Formaldehyde is found in many building materials and plastic industries.
Mercapto Mix (Position 22):
Mercapto mix is composed of three chemical accelerators that are benzothiazole sulfenamide derivatives. N-cyclohexylbenzothiazyl-sulfenamide (purity ≥85%), dibenzothiazyl disulfide (purity ≥97%), and morpholinylmercaptobenzothiazole (purity ≥85%) are present in equal parts. The gel vehicle is povidone. The product is formulated to contain 75 mcg/cm 2of mercapto mix, which corresponds to 61 mcg of mercapto mix per patch. This group of chemicals is found in many rubber products, such as shoes, gloves, and elastics.
Thimerosal (Position 23):
Thimerosal, USP (purity ≥97%) is a preservative that contains mercury. The gel vehicle is povidone. The product is formulated to contain 7 mcg/cm 2of thimerosal, which corresponds to 6 mcg of thimerosal per patch. Thimerosal is found in some cosmetics, nose drops, eardrops, and vaccines.
Thiuram Mix (Position 24):
Thiuram mix is composed of four substances in equal parts: tetramethylthiuram monosulfide (purity ≥95%, contains small amounts of tetramethylthiuram disulfide); tetramethylthiuram disulfide (purity ≥95%, contains small amounts of tetramethylthiuram monosulfide); disulfiram, USP (tetraethylthiuram disulfide, purity ≥98.0%); and dipentamethylenethiuram disulfide (purity ≥95%, impurities unknown). The components of thiuram mix can chemically interact, resulting in the formation of mixed disulfides. Thiuram monosulfides and disulfides are the active allergens. The gel vehicle is povidone. The product is formulated to contain 27 mcg/cm 2of thiuram mix, which corresponds to 22 mcg of thiuram mix per patch (5.5 mcg of tetramethylthiuram monosulfide, 5.5 mcg of tetramethylthiuram disulfide, 5.5 mcg of disulfiram, and 5.5 mcg of dipentamethylenethiuram disulfide). These antimicrobial, accelerator, and antioxidant substances are found in many rubber products.
Close11.3 Allergens on Panel 3.3
Diazolidinyl Urea (DU) (Germall ®II) (Position 25):
Diazolidinyl urea is a complex mixture. The gel vehicle is povidone. The product is formulated to contain 550 mcg/cm 2of diazolidinyl urea, which corresponds to 446 mcg of diazolidinyl urea per patch. Diazolidinyl urea is a preservative found in cosmetics.
Quinoline Mix (Position 26):
Quinoline mix is composed of two chemical germicides. Clioquinol, USP (purity ≥93.0%) and clorquinaldol (purity ≥95%), which are present in equal parts. The product is formulated to contain 190 mcg/cm 2of quinoline mix, which corresponds to 154 mcg of quinoline mix per patch. The gel vehicle is povidone. Quinolines are found in paste bandages, medicated creams, and ointments.
Tixocortol-21-Pivalate (TIX) (Position 27):
Tixocortol-21-pivalate (purity ≥95%) is a corticosteroid. The gel vehicle is povidone. The product is formulated to contain 3 mcg/cm 2of tixocortol-21-pivalate, which corresponds to 2 mcg of tixocortol-21-pivalate per patch. Tixocortol-21-pivalate is found in some medical products. Patch testing with tixocortol-21-pivalate may be used to assist in the diagnosis of allergic contact dermatitis due to corticosteroids in Group A, based on the classification of topical corticosteroids by cross-reactivity.
Gold Sodium Thiosulfate (GST) (Position 28):
Gold sodium thiosulfate (purity ≥ 90%) is a fairly common sensitizer with elicitation of symptoms linked to gold in jewelry, occupational exposure to gold, previous rheumatoid arthritis treatment, dental restorations, and gold-plated intracoronary stents. The gel vehicle is hydroxypropyl cellulose and the product is formulated to contain 75 mcg/cm 2of gold sodium thiosulfate, which corresponds to 23 mcg of gold per patch.
Imidazolidinyl Urea (IMID) (Germall ®115) (Position 29):
Imidazolidinyl urea is a complex mixture. The gel vehicle is povidone. The product is formulated to contain 600 mcg/cm 2of imidazolidinyl urea, which corresponds to 486 mcg of imidazolidinyl urea per patch. Imidazolidinyl urea is a preservative found in cosmetics.
Budesonide (BUD) (Position 30):
Budesonide, USP (purity ≥98.0%) is a corticosteroid. The gel vehicle is povidone. The product is formulated to contain 1 mcg/cm 2of budesonide, which corresponds to 0.8 mcg of budesonide per patch. Budesonide is found in topical medicinal and anti-inflammatory products. Patch testing with budesonide may be used to assist in the diagnosis of allergic contact dermatitis due to corticosteroids in Group B and to certain esters in Group D, based on the classification of topical corticosteroids by cross-reactivity.
Hydrocortisone-17-Butyrate (H-17-B) (Position 31):
Hydrocortisone-17-butyrate, USP (purity ≥ 97.0%) is a mid-potent (Group D2) corticosteroid, most commonly used to treat inflammatory skin disease and psoriasis. The gel vehicle is povidone. The product is formulated to contain 20 mcg/cm 2of hydrocortisone-17-butyrate, which corresponds to 16 mcg of hydrocortisone-17-butyrate per patch. Hydrocortisone-17-butyrate is found in many topical over-the-counter (OTC) and prescription pharmaceuticals.
Mercaptobenzothiazole (Position 32):
Mercaptobenzothiazole (purity ≥98.5%) is a vulcanization accelerator used in rubber products. The gel vehicle is povidone. The product is formulated to contain 75 mcg/cm 2of mercaptobenzothiazole, which corresponds to 61 mcg of mercaptobenzothiazole per patch. This chemical is found in many rubber products, some adhesives, and is used as an industrial anticorrosive agent.
Bacitracin (Position 33):
Bacitracin, USP is an antibiotic used for postoperative and general wound care and is considered a frequent sensitizer. The gel vehicle is hydroxypropyl cellulose and the product is formulated to contain 600 mcg/cm 2of bacitracin which corresponds to 486 mcg of bacitracin per patch. Bacitracin is often a first-line topical remedy for cutaneous injuries and dermatoses as well as for many eye and ear disorders.
Parthenolide (Position 34):
Parthenolide (purity ≥ 95%) is a sesquiterpene lactone, which occurs naturally in thousands of plants including Daisies, Feverfew, and Magnolia. The gel vehicle is povidone and the product is formulated to contain 3 mcg/cm 2of parthenolide, which corresponds to 2 mcg of parthenolide per patch. Allergies to parthenolide occur frequently in gardeners and greenhouse employees, but non-occupational contact with plants and herbal teas containing sesquiterpenes also occurs.
Disperse Blue 106 (DB106) (Position 35):
Disperse blue 106 (purity ≥ 90%) is a commonly used thiazol-azoyl-p-phenylene diamine derivative dye used primarily in synthetic textiles and is a significant skin sensitization hazard. The product is formulated to contain 50 mcg/cm 2of disperse blue 106, which corresponds to 41 mcg of disperse blue 106 per patch. The gel vehicle is povidone. Together with Disperse blue 124, it has been determined to be the primary cause of textile dermatitis.
2-Bromo-2-nitropropane-1,3-diol (Bronopol) (Position 36):
2-Bromo-2-nitropropane-1,3-diol (purity ≥ 95%) is an antimicrobial agent commonly used as a preservative in many types of cosmetics, personal care products, and topical medications. The gel vehicle is povidone. The product is formulated to contain 250 mcg/cm 2of 2-Bromo-2-nitropropane-1,3-diol, which corresponds to 203 mcg of 2-Bromo-2-nitropropane-1,3-diol per patch.
-
12 CLINICAL PHARMACOLOGY12.1 Mechanism of Action - A positive response to the patch test is a classic delayed cell-mediated hypersensitivity reaction (type IV), which normally appears within 9 to 96 hours after ...Close
12.1 Mechanism of Action
A positive response to the patch test is a classic delayed cell-mediated hypersensitivity reaction (type IV), which normally appears within 9 to 96 hours after exposure. Following primary contact, an allergen penetrates the skin and binds covalently or noncovalently to epidermal Langerhans cells. The processed allergen is presented to helper T-lymphocytes, resulting in inflammation that produces a papular, vesicular, or bullous response with erythema and itching at the site of application.
-
14 CLINICAL STUDIESA basic description of the interpretation method used by study personnel to evaluate the patch reactions obtained during the clinical studies is as follows [see - Dosage and Administration ...
A basic description of the interpretation method used by study personnel to evaluate the patch reactions obtained during the clinical studies is as follows [see Dosage and Administration (2)]:
? Doubtful reaction + Weak positive reaction ++ Strong positive reaction +++ Extreme positive reaction - Negative reaction IR Irritant reaction 14.1 Adults
Ten (10) studies were conducted in North America and Europe to evaluate the effectiveness, frequency of patch reactions, and/or sensitivity and specificity, and/or agreement with a reference allergen (when used) of T.R.U.E. TEST used to diagnosis allergic reactions to one or more allergens and or allergen mixes in the panels in adults. Subjects ranged in age from 18 through 86 years. Subjects with suspected allergic contact dermatitis, based on history or clinical signs, were tested in all studies. The allergens tested in each of the studies are noted in Table 1 [see Adverse Reactions (6.1)].
Study No. 1
This study evaluated the efficacy of T.R.U.E. TEST Panel 1.1. A total of 127 subjects with suspected contact dermatitis were recruited. T.R.U.E. TEST Panel 1.1, containing 12 allergens (no negative control was on the original Panel 1) was applied to the subject's back and remained there for 48 hours. The results were evaluated after 48 and 72 to 96 hours. Forty-five (45) subjects showed a total of 65 reactions to 11 of the 12 allergens in Panel 1.1. There were positive test reactions to all allergens except potassium dichromate. See Table 4.
Study No. 2
This study evaluated the efficacy of T.R.U.E. TEST Panel 2.1. A total of 121 subjects with suspected contact dermatitis were recruited. T.R.U.E. TEST Panel 2.1, containing 11 allergens and a negative control, was applied to the subject's back and remained there for 48 hours. The results were evaluated after 72 to 96 hours. Thirty-two (32) subjects showed a total of 46 positive test reactions. There were positive responses to all of the allergens except quinoline mix and paraben mix. See Table 4.
Study No. 3
This study evaluated the efficacy of T.R.U.E. TEST Panels 1.1 and 2.1 in a North American patient population referred for patch testing. One hundred nineteen (119) subjects were enrolled. T.R.U.E. TEST Panels 1.1 and 2.1, containing 23 allergens and a negative control, were applied to the subject's back and remained there for 48 hours. The results were evaluated at 72 to 96 hours after application. Results show that 66 subjects had a total of 123 positive test reactions. There were positive test responses to all of the allergens. See Table 4.
Study No. 4
This study was an open, multicenter, study that evaluated the efficacy of T.R.U.E. TEST and obtained information on late reactions and persistent local responses at a Day 21 safety visit (see Table 2). A total number of 50 prospectively identified subjects with suspected contact dermatitis were recruited. The most common dermatitis site was the hand, and the most common dermatitis type was allergic. T.R.U.E. TEST Panels 1.1 and 2.1 (24 allergens or allergen mixes, no negative control) were applied to the subject's back and remained there for 48 hours. The results were evaluated after 72 to 96, 120, or 168 hours. Thirty-two (32) subjects showed a total of 66 reactions to 21 of the 24 allergens included in T.R.U.E. TEST. The following allergens gave no reactions: caine mix, epoxy resin, quinoline mix, and black rubber mix. See Table 4.
Study No. 5
This single-site study evaluated the sensitivity and specificity of T.R.U.E. TEST Panel 3 allergens diazolidinyl urea (DU) (Germall® II) and imidazolidinyl urea (IMID) (Germall® 115) for diagnosing allergic contact dermatitis in a North American patient population. Comparison of allergen reactivity between allergens in T.R.U.E. TEST and allergens in petrolatum were made. One hundred thirty (130) subjects were enrolled and included 100 consecutive subjects (subjects with a clinical history consistent with allergic contact dermatitis without a previous positive patch test reaction) and sensitive subjects with a previous positive patch test reaction to petrolatum-based DU (15 subjects) and IMID (15 subjects) allergens in the past 5 years and a clinical history of allergic contact dermatitis. T.R.U.E. TEST Panel 3 allergens DU and IMID were applied to the subject's back and remained there for 48 hours. Patch test reactions were evaluated at 72 to 96 hours and again 7 days after application using the study endpoints, including measurements of positive reaction frequency, specificity, sensitivity, and agreement estimate as shown in Table 4 for each allergen.
Study No. 6
This study evaluated the sensitivity and specificity of T.R.U.E. TEST Panel 3 allergens tixocortol-21-pivalate (TIX) and budesonide (BUD) for diagnosing allergic contact dermatitis in a North American patient population. Comparison of allergen reactivity between allergens in T.R.U.E. TEST and allergens in petrolatum were made. One hundred twenty-eight (128) subjects were enrolled and included 100 consecutive subjects (subjects with a clinical history consistent with allergic contact dermatitis without a previous positive patch test reaction) and sensitive subjects with a previous positive patch test reaction to petrolatum based TIX (9 subjects) and BUD (19 subjects) allergens in the past 5 years and clinical history of allergic contact dermatitis. T.R.U.E. TEST Panel 3 allergens TIX and BUD were applied to the subject's back and remained there for 48 hours. Patch test reactions were evaluated 72 to 96 hours and again 7 days after application for 94 subjects. Six (6) subjects were withdrawn at Visit 2 due to poor tape adhesion prior to Visit 2. Patch test reactions were evaluated using the study endpoints, including measurements of positive reaction frequency, specificity, sensitivity, and agreement estimate as shown in Table 4.
Study No. 7
This study evaluated the sensitivity and specificity of T.R.U.E. TEST Panel 3.1 allergens tixocortol-21-pivalate (TIX), Hydrocortisone-17-butyrate (H-17-B), and budesonide (BUD) for diagnosing allergic contact dermatitis in a European patient population. Comparison of allergen reactivity between allergens in T.R.U.E. TEST and allergens in petrolatum were made. The enrolled study population included 200 consecutive subjects (subjects with a clinical history consistent with allergic contact dermatitis without a previous positive patch test reaction). T.R.U.E. TEST allergens TIX, H-17-B, BUD, and the corresponding petrolatum reference allergens were applied to the subject's back and remained there for 48 hours. Patch test reactions were evaluated at 72 to 96 hours and again 7 days after application. Of the 200 consecutive subjects evaluated, 1 subject was withdrawn due to poor tape adhesion prior to Visit 2 and 1 subject was excluded due to no follow-up visits. Therefore, 198 subjects were included in the evaluation of TIX and BUD. In addition, 3 subjects were withdrawn due to the H-17-B reference allergen patch not being applied at the initial visit. Therefore, 195 subjects were included in the evaluation for H-17-B. Patch test reactions were evaluated using the study endpoints, including measurements of positive reaction frequency, specificity, sensitivity, and agreement estimate as shown in Table 4.
Study No. 8
This was an open label, prospective, multi-center (5 site) study that evaluated the sensitivity and specificity of gold sodium thiosulphate (GST), Hydrocortisone-17-butyrate (H-17-B), bacitracin, parthenolide, methyldibromo glutaronitrile (MDBGN), disperse blue 106 (DB106), and 2-bromo-2-nitropropane-1,3-diol (bronopol) in adult subjects with suspected contact dermatitis and in adult subjects with a known or suspected sensitization to at least 1 of the 7 allergens. Of the 235 enrolled subjects, 110 were consecutive subjects (subjects with a clinical history consistent with allergic contact dermatitis without a previous positive patch test reaction) and 125 were sensitive subjects (subjects with a previous positive patch test reaction to at least 1 of the 7 allergens). The frequencies of all patch test reactions for each allergen were tabulated at 72 to 96 hours.
The agreement between the T.R.U.E. TEST allergens and their corresponding reference allergens was generally high among subjects who were sensitive to each allergen as seen in Table 4. With the exception of MDBGN, subjects who had sensitivities to each individual allergen had similar reactions to both the T.R.U.E. TEST allergens and the corresponding reference allergens with percent agreements ranging from 75.0% (for bacitracin) to 94.4% (for parthenolide). The results for MDBGN in this study may be unreliable due to the presence of phenoxyethanol (PE) in the reference allergen. Specifically, PE is a recognized irritant. Results of frequency, specificity, sensitivity, and agreement estimate are shown in Table 4.
Study No. 9
This in-use study evaluated the relationship between reactions caused by a natural sensitizer, such as nickel containing costume jewelry, and T.R.U.E. TEST. Forty nine (49) subjects with a history of cutaneous reactions to jewelry were tested with T.R.U.E. TEST Panel 1.1. A medallion containing approximately 20% nickel served as a positive control. Reactions were evaluated 72 to 96 hours after application. In comparing the in-use test results, 35% of the T.R.U.E. TEST nickel patch positive results would have been considered false positives and 5.3% would have been considered false negatives. However, the results from this study may be unreliable. The metal composition of jewelry can vary greatly from manufacturer to manufacturer and thereby alter the bioavailability of the nickel ions. A different medallion could have produced either a greater or lesser correlation with T.R.U.E. TEST nickel patch. The comparatively large number of additional nickel positive results obtained with T.R.U.E. TEST may be true positives unresponsive to the particular medallion used in this study, although false positive reactions cannot be ruled out.
Study No. 10
The study was a Phase 4 postmarketing open label, non-randomized, non-blinded prospective study including nine subjects. All subjects had previous positive patch test results for Quaternium-15 (Q-15). The subjects were exposed to the T.R.U.E. TEST Q-15 patch at a concentration of 100 mcg/cm 2and daily applications of a topical product containing Q-15 after the completion of the patch test. Reactions were evaluated 72 to 96 hours after application. T.R.U.E. TEST detected Q-15 sensitivity in 87.50% (7/8) of the Q-15 allergic subjects while the topically applied lotion elicited a positive response in 50% (4/8) of the study population. One subject tested negative to both methods of Q-15 and was removed from the endpoint analysis.
Study Data
Table 4 presents the frequency of positive reactions to T.R.U.E. TEST in consecutive subjects (subjects with a clinical history consistent with allergic contact dermatitis without a previous positive patch test reaction) for each allergen. The frequency of positive reactions to T.R.U.E. TEST in consecutive subjects and sensitive subjects (subjects with a previous positive patch test reaction to a specific allergen or allergens) for Quaternium-15 are presented in Table 4. The sensitivity, specificity, and agreement estimate between each T.R.U.E. TEST allergen and the corresponding reference allergen (when used) are presented for consecutive subjects and sensitive subjects (when done) in Table 4.
Sensitivity was determined using the formula [TP / (TP + FN)] × 100, where TP (true positive) represented positive results for both the T.R.U.E. TEST allergen and the corresponding reference allergen, and FN (false negative) represented negative results for the T.R.U.E. TEST allergen and positive results for the corresponding reference allergen. Specificity was determined using the formula [TN / (TN + FP)] × 100, where TN (true negative) represented negative results for both the T.R.U.E. TEST allergen and the corresponding reference allergen, and FP (false positive) represented positive results for the T.R.U.E. TEST allergen and negative results for the corresponding reference allergen. The agreement estimate (with 95% confidence intervals), as well as overall percent agreement and Cohen's kappa statistic between the test site skin reactions obtained for each of the seven T.R.U.E. TEST Panel 3.2 allergens and the test site skin reactions obtained for their associated reference allergens in petrolatum or ethanol were calculated for all subjects combined, as well as for the populations of sensitive and consecutive subjects.
Table 6- Available Frequency, Sensitivity, Specificity, and Agreement Estimate of T.R.U.E. TEST Allergens ** Dose and/or excipients may have changed from clinical study product reported to finished product. Panel 1.3 Nickel sulfate *Position 1 (Studies 1, 3, 4 and 9) Frequency from consecutive subjects only (%) 90/345 (26.1) Wool alcohols (Lanolin) *Position 2 (Studies 2, 3, and 4) Frequency from consecutive subjects only (%) 4/290 (1.4) Neomycin sulfate *Position 3 (Studies 1, 3, 4, and 9) Frequency from consecutive subjects only (%) 16/345 (4.6) Potassium dichromate *Position 4 (Studies 1, 3, 4 and 9) Frequency from consecutive subjects only (%) 5/345 (1.4) Caine mix *Position 5 (Studies 1, 3, 4, and 9) Frequency from consecutive subjects only (%) 7/345 (2.0) Fragrance mix *Position 6 (Studies 1, 3, 4, and 9) Frequency from consecutive subjects only (%) 23/345 (6.7) Colophony *Position 7 (Studies 1, 3, 4, and 9) Frequency from consecutive subjects only (%) 11/345 (3.2) Paraben mix *Position 8 (Studies 1, 3, 4, and 9) Frequency from consecutive subjects only (%) 5/290 (1.7) Balsam of peru *Position 10 (Studies 1, 3, 4, and 9) Frequency from consecutive subjects only (%) 17/345 (4.9) Ethylenediamine dihydrochloride *Position 11 (Studies 1, 3, 4 and 9) Frequency from consecutive subjects only (%) 7/345 (2.0) Cobalt dichloride *Position 12 (Studies 1, 3, 4, and 9) Frequency from consecutive subjects only (%) 29/345 (8.4) Panel 2.3 p-tert-Butylphenol formaldehyde resin *Position 13 (Studies 2, 3, and 4) Frequency from consecutive subjects only (%) 9/290 (3.0) Epoxy resin *Position 14 (Studies 1, 3, 4, and 9) Frequency from consecutive subjects only (%) 5/345 (1.4) Carba mix *Position 15 (Studies 2, 3, and 4) Frequency from consecutive subjects only (%) 6/290 (2.1) Black rubber mix *Position 16 (Studies 2, 3, and 4) Frequency from consecutive subjects only (%) 4/290 (1.4) Cl+ Me- isothiazolinone (MCI/MI) *Position 17 (Studies 2, 3, and 4) Frequency from consecutive subjects only (%) 8/290 (2.8) Quaternium-15 (Q-15) *Position 18 Test on sensitive subjects (Study 10) Frequency from consecutive subjects only (%) 7/9 (77.8) Test on consecutive subjects (Studies 2, 3, and 4) Frequency from consecutive subjects only (%) 21/290 (7.2) Methyldibromo glutaronitrile (MDBGN) Position 19 (Study 8) Test on sensitive subjects Petrolatum-based MDBGN/PE † Pos Neg Total T.R.U.E. TEST Panel Allergen Pos 4 1 5 Neg 10 14 24 Total 14 15 29 Sensitivity (%) (95% CI) 28.6 (8.4, 58.1) Specificity (%) (95% CI) 93.3 (68.1, 99.8) Agreement (%) (95% CI) 62.1 (42.3, 79.3) Test on consecutive subjects Petrolatum-based MDBGN/PE † Pos Neg Total T.R.U.E. TEST Panel Allergen Pos 1 0 1 Neg 5 104 109 Total 6 104 110 Frequency from consecutive subjects only (%) 1/110 (0.9) Sensitivity (%)(95% CI) 16.7 (0.4, 64.1) Specificity (%) (95% CI) 100 (96.5, 100.0) Agreement (%) (95% CI) 95.5 (89.7, 98.5) Phenylenediamine *Position 20 (Studies 1, 3, 4, and 9) Frequency from consecutive subjects only (%) 13/345 (3.8) Formaldehyde *Position 21 (Studies 3 and 4) Frequency from consecutive subjects only (%) 10/169 (5.9) Mercapto mix *Position 22 (Studies 2, 3, and 4) Frequency from consecutive subjects only (%) 9/290 (3.1) Thimerosal *Position 23 (Studies 2, 3, and 4) Frequency from consecutive subjects only (%) 30/290 (10.3) Thiuram mix *Position 24 (Studies 1, 3, 4, and 9) Frequency from consecutive subjects only (%) 14/345 (4.1) Panel 3.3 Diazolidinyl urea (DU) (Germall® II) Position 25 (Study 5) Test on sensitive subjects * Petrolatum-based DU Pos Neg Total T.R.U.E. TEST Allergen Pos 4 2 6 Neg 2 7 9 Total 6 9 15 Sensitivity (%)(95% CI) 66.7 (22.3, 95.7) Specificity (%) (95% CI) 77.8 (40.0, 97.2) Agreement (%) (95% CI) 73.3 (44.9, 92.2) Test on consecutive subjects Petrolatum-based DU Pos Neg Total T.R.U.E. TEST Allergen Pos 3 1 4 Neg 3 91 94 Total 6 92 98 Frequency from consecutive subjects only (%) 4 /98 (4.1) Sensitivity (%)(95% CI) 50 (11.8, 88.2) Specificity (%) (95% CI) 98.9 (94.1, 100.0) Agreement (%) (95% CI) 95.9 (89.9, 98.9) Quinoline mix *Position 26 (Studies 2, 3, and 4) Frequency from consecutive subjects only (%) 2/290 (0.7) Tixocortol-21-pivalate (TIX) Position 27 (Studies 6 and 7 ‡) Test on sensitive subjects Petrolatum-based TIX Pos Neg Total T.R.U.E. TEST Allergen Pos 7 1 8 Neg 0 1 1 Total 7 2 9 Sensitivity (%)(95% CI) 100 (59.0, 100.0) Specificity (%) (95% CI) 50 (1.3, 98.7) Agreement (%) (95% CI) 88.9 (51.8, 99.7) Test on consecutive subjects Petrolatum-based TIX Pos Neg Total T.R.U.E. TEST Allergen Pos 5 4 9 Neg 1 282 283 Total 6 286 292 Frequency from consecutive subjects only (%) 9/292 (3.1) Sensitivity (%)(95% CI) 83.3 (35.9, 99.6) Specificity (%) (95% CI) 98.6 (96.5, 99.6) Agreement (%) (95% CI) 98.3 (96.0, 99.4) Gold sodium thiosulfate (GST) Position 28 (Study 8) Test on sensitive subjects Petrolatum-based GST Pos Neg Total T.R.U.E. TEST Allergen Pos 12 3 15 Neg 0 4 4 Total 12 7 19 Sensitivity (%)(95% CI) 100 (73.5, 100.0) Specificity (%) (95% CI) 57.1 (18.4, 90.1) Agreement (%) (95% CI) 84.2 (60.4, 96.6) Test on consecutive subjects Petrolatum-based GST Pos Neg Total T.R.U.E. TEST Allergen Pos 11 17 28 Neg 0 82 82 Total 11 99 110 Frequency from consecutive subjects only (%) 28/110 (25.5) Sensitivity (%)(95% CI) 100 (71.5, 100.0) Specificity (%) (95% CI) 82.8 (73.9, 89.7) Agreement (%) (95% CI) 84.5 (76.4, 90.7) Imidazolidinyl urea (IMID) (Germall 115®) Position 29 (Study 5) Test on sensitive subjects Petrolatum-based IMID Pos Neg Total T.R.U.E. TEST Allergen Pos 3 1 4 Neg 2 9 11 Total 5 10 15 Sensitivity (%)(95% CI) 60 (14.7, 94.7) Specificity (%) (95% CI) 90 (55.5, 99.7) Agreement (%) (95% CI) 80.0 (51.9, 95.7) Test on consecutive subjects Petrolatum-based IMID Pos Neg Total T.R.U.E. TEST Allergen Pos 3 0 3 Neg 3 92 95 Total 6 92 98 Frequency from consecutive subjects only (%) 3/98 (3.1) Sensitivity (%)(95% CI) 50 (11.8, 88.2) Specificity (%) (95% CI) 100 (96.1, 100.0) Agreement (%) (95% CI) 96.9 (91.3, 99.4) Budesonide (BUD) Position 30 (Study 6 and 7 ‡) Test on sensitive subjects Petrolatum-based budesonide Pos Neg Total T.R.U.E. TEST Allergen Pos 11 1 12 Neg 1 6 7 Total 12 7 19 Sensitivity (%)(95% CI) 91.7 (61.5, 99.8) Specificity (%) (95% CI) 85.7 (42.1, 99.6) Agreement (%) (95% CI) 89.5 (66.9, 98.7) Test on consecutive subjects Petrolatum-based budesonide Pos Neg Total T.R.U.E. TEST Allergen Pos 2 1 3 Neg 0 289 289 Total 2 290 292 Frequency from consecutive subjects only (%) 3/292 (1.0) Sensitivity (%)(95% CI) 100 (15.8, 100.0) Specificity (%) (95% CI) 99.7 (98.1, 100.0) Agreement (%) (95% CI) 99.7 (98.1, 100.0) Hydrocortisone-17-butyrate (H-17-B) Position 31 (Studies 7 ‡and 8) Test on sensitive subjects Ethanol-based H-17-B Pos Neg Total T.R.U.E. TEST Allergen Pos 12 1 13 Neg 1 6 7 Total 13 7 20 Sensitivity (%)(95% CI) 92.3 (64.0, 99.8) Specificity (%) (95% CI) 85.7 (42.1, 99.6) Agreement (%) (95% CI) 90.0 (68.3, 98.8) Test on consecutive subjects Ethanol-based H-17-B Pos Neg Total T.R.U.E. TEST Allergen Pos 0 0 0 Neg 1 304 305 Total 1 304 305 Frequency from consecutive subjects only (%) 0/305 (0.0) Sensitivity (%)(95% CI) 0 (0.0, 97.5) Specificity (%) (95% CI) 100 (98.8, 100.0) Agreement (%) (95% CI) 99.7 (98.2, 100.0) Mercaptobenzothiazole *Position 32 (Studies 2, 3, and 4) Frequency from consecutive subjects only (%) 8/290 (2.8) Bacitracin Position 33 (Study 8) Test on sensitive subjects Petrolatum-based bacitracin Pos Neg Total T.R.U.E. TEST Allergen Pos 13 5 18 Neg 1 5 6 Total 14 10 24 Sensitivity (%)(95% CI) 92.9 (66.1, 99.8) Specificity (%) (95% CI) 50 (18.7, 81.3) Agreement (%) (95% CI) 75.0 (53.3, 90.2 ) Test on consecutive subjects Petrolatum-based bacitracin Pos Neg Total T.R.U.E. TEST Panel Allergen Pos 5 0 5 Neg 1 104 105 Total 6 104 110 Frequency from consecutive subjects only (%) 5/110 (4.5) Sensitivity (%)(95% CI) 83.3 (35.9, 99.6) Specificity (%) (95% CI) 100 (96.5, 100.0) Agreement (%) (95% CI) 99.1 (95.0, 100.0) Parthenolide Position 34 (Study 8) Test on sensitive subjects Petrolatum-based parthenolide Pos Neg Total T.R.U.E. TEST Allergen Pos 13 0 13 Neg 1 4 5 Total 14 4 18 Sensitivity (%)(95% CI) 92.9 (66.1, 99.8) Specificity (%) (95% CI) 100 (39.8, 100.0) Agreement (%) (95% CI) 94.4 (72.7, 99.9) Test on consecutive subjects Petrolatum-based parthenolide Pos Neg Total T.R.U.E. TEST Panel Allergen Pos 1 0 1 Neg 0 109 109 Total 1 109 110 Frequency from consecutive subjects only (%) 1/110 (0.9) Sensitivity (%)(95% CI) 100 (2.5, 100.0) Specificity (%) (95% CI) 100 (96.7, 100.0) Agreement (%) (95% CI) 100.0 (96.7, 100.0) Disperse blue 106 (DB106) Position 35 (Study 8) Test on sensitive subjects Petrolatum-based DB106 Pos Neg Total T.R.U.E. TEST Allergen Pos 8 1 9 Neg 1 7 8 Total 9 8 17 Sensitivity (%)(95% CI) 88.9 (51.8, 99.7) Specificity (%) (95% CI) 87.5 (47.3, 99.7) Agreement (%) (95% CI) 88.2 (63.6, 98.5) Test on consecutive subjects Petrolatum-based DB106 Pos Neg Total T.R.U.E. TEST Panel Allergen Pos 1 0 1 Neg 0 109 109 Total 1 109 110 Frequency from consecutive subjects only (%) 1/110 (0.9) Sensitivity (%)(95% CI) 100 (2.5, 100.0) Specificity (%) (95% CI) 100 (96.7, 100.0) Agreement (%) (95% CI) 100.0 (96.7, 100.0) 2-Bromo-2-nitropropane-1,3-diol (Bronopol) Position 36 (Study 8) Test on sensitive subjects Petrolatum-based Bronopol Pos Neg Total T.R.U.E. TEST Allergen Pos 4 4 8 Neg 0 15 15 Total 4 19 23 Sensitivity (%)(95% CI) 100 (39.8, 100.0) Specificity (%) (95% CI) 78.9 (54.4, 93.9) Agreement (%) (95% CI) 82.6 (61.2, 95.0) Test on consecutive subjects Petrolatum-based Bronopol Pos Neg Total T.R.U.E. TEST Panel Allergen Pos 1 2 3 Neg 0 107 107 Total 1 109 110 Frequency from consecutive subjects only (%) 3/110 (2.7) Sensitivity (%)(95% CI) 100 (2.5, 100.0) Specificity (%) (95% CI) 98.2 (93.5, 99.8) Agreement (95% CI) 98.2 (93.6, 99.8) Close14.2 Children and Adolescents 6 through 17 Years of Age
Two studies were conducted in the US to evaluate the diagnostic performance of T.R.U.E. TEST in children and adolescents 6 through 17 years of age. Subjects had three T.R.U.E. TEST panels applied to their back or upper arm for 48 hours. Reactions at the patch test sites were evaluated at days 3 and/or 4, 7 and 21 after patch test application [see Interpretation Instructions (2.4)].
Pediatric Study 1
In an open-label, prospective, single-center study conducted in the US, 102 children and adolescents 6 through 17 years of age with suspected allergic contact dermatitis were enrolled to evaluate the diagnostic performance of a previously approved version of T.R.U.E. TEST (Panels 1.1, 2.1, 3.1). This version included a negative control and 28 allergens and allergen mixes, 4 (on Panel 1.1) of which were reformulated and are not included on Panel 1.3 .The per-protocol analysis set included 100 subjects. The frequencies of positive reactions to the 24 allergens contained in T.R.U.E. TEST and to the negative controls are shown in Table 7.
Pediatric Study 2
In an open-label, prospective, multi-center study conducted in the US, 116 children and adolescents 6 through 17 years of age with suspected allergic contact dermatitis were enrolled. Although the three T.R.U.E. TEST panels administered to subjects in this study included all 35 allergens and allergen mixes, the primary analysis of diagnostic performance was limited to the 4 reformulations and the 7 new allergens and allergen mixes. The frequencies of positive reactions to 11 of the allergens contained in T.R.U.E. TEST and to the negative control were analyzed and are shown in Table 7.
Table 7- Pediatric Studies 1 *and 2 †: Frequencies of Positive Reactions ‡(on Days 3, 4, and 7 After T.R.U.E. TEST Application) to T.R.U.E. TEST Allergens Among Children and Adolescents 6 through 17 Years of Age with Suspected Allergic Contact Dermatitis: Per Protocol Analysis Sets Pediatric Study 1 * Pediatric Study 2 † - *
- NCT: 00795951
- †
- NCT: 01797562
- ‡
- In Pediatric Studies 1 and 2, positive patch reactions were read on days 3, 4, and 7. Frequencies of positive reactions reported in this table include those characterized as weak (+), strong (++), and extreme positive (+++) based on presence and degree of erythema, infiltration, papules, vesicles, and bullae.
- §
- Only 53 subjects in Pediatric Study 2 in the per-protocol analysis set received Panel 1.3.
- ¶
- NA= Not Applicable/Not Available.The results are not applicable because the allergens and allergen mixes were either from previously approved formulations, not evaluated or not pre-specified as the primary endpoint of the study.
N=100 N=53 § Panel Allergen n (%) n (%) Panel 1.3 1. Nickel Sulfate 30 (30.0) NA ¶ 2. Wool Alcohols 16 (16.0) NA 3. Neomycin Sulfate NA 2 (3.8) 4. Potassium Dichromate NA 2 (3.8) 5. Caine Mix 0 (0.0) NA 6. Fragrance Mix NA 2 (3.8) 7. Colophony 9 (9.0) NA 8. Paraben Mix 2 (2.0) NA 9. Negative Control 0 (0.0) NA 10. Balsam of Peru 10 (10.0) NA 11. Ethylenediamine Dihydrochloride NA 0 (0.0) 12. Cobalt Dichloride 13 (13.0) NA N=100 N=111 Panel 2.3 13. p-tert-Butylphenol Formaldehyde Resin 17 (17.0) NA 14. Epoxy Resin 4 (4.0) NA 15. Carba Mix 7 (7.0) NA 16. Black Rubber Mix 2 (2.0) NA 17. Cl+Me-Isothiazolinone (MCI/MI) 4 (4.0) NA 18. Quaternium-15 4 (4.0) NA 19. Methyldibromo Glutaronitrile (MDBGN) NA 1 (0.9) 20. p-Phenylenediamine 2 (2.0) NA 21. Formaldehyde 7 (7.0) NA 22. Mercapto Mix 2 (2.0) NA 23. Thimerosal 4 (4.0) NA 24. Thiuram Mix 7 (7.0) NA Panel 3.3
.25. Diazolidinyl urea 5 (5.0) NA 26. Imidazolidinyl urea 2 (2.0) NA 27. Budesonide 1 (1.0) NA 29. Quinoline Mix 1 (1.0) NA 28. Tixocortol-21-pivalate 8 (8.0) NA 28. Gold Sodium Thiosulfate (GST) NA 30 (27.0) 31. Hydrocortisone-17-Butyrate (H-17-B) NA 2 (1.8) 19. & 32. Mercaptobenzothiazole 2 (2.0) NA 33. Bacitracin NA 14 (12.6) 34. Parthenolide NA 8 (7.2) 35. Disperse Blue 106 NA 4 (3.6) 36. 2-Bromo-2-nitropane-1,3-diol (Bronopol) NA 19 (17.1) -
16 HOW SUPPLIED/STORAGE AND HANDLINGA multipack carton contains five units. Each unit consists of three adhesive panels, each containing 12 patches - NDC 67334-0457-1. Store T.R.U.E. TEST between 2° and 8°C (36° and 46°F) ...
A multipack carton contains five units. Each unit consists of three adhesive panels, each containing 12 patches - NDC 67334-0457-1.
CloseStore T.R.U.E. TEST between 2° and 8°C (36° and 46°F). Refrigeration required. Do not freeze. Failure to store T.R.U.E. TEST as recommended may result in loss of potency and inaccurate test results.
-
17 PATIENT COUNSELING INFORMATIONInform the patient of the following: Patients should seek immediate medical attention and contact their healthcare provider if they experience symptoms of a severe allergic reaction such as ...
Inform the patient of the following:
- Patients should seek immediate medical attention and contact their healthcare provider if they experience symptoms of a severe allergic reaction such as trouble breathing or wheezing; a swollen tongue or throat; a drop in blood pressure resulting in dizziness or fainting; a weak and rapid pulse; hives or widespread itching [see Warnings and Precautions (5.1)].
- Patients may remove the panels themselves if advised by their healthcare provider to do so or if they are experiencing systemic symptom [see Warnings and Precautions (5.1)].
- Itching and burning sensations are common with patch testing and may be severe in extremely sensitive patients.
- Avoid UV exposure and tanning beds [see Dosage and Administration (2.1)].
- Patients should report to their physician any reactions at the patch test site occurring seven or more days after panel removal to identify potential late or persistent reactions or possible sensitizations [see Warnings and Precautions (5.2, 5.7, 5.8) and Dosage and Administration (2.2)] .
- p-Phenylenediamine and disperse blue 106 (DB106) are dyes and may leave a dark spot on the skin at the allergen location. This is not an allergic reaction. This discoloration may remain for approximately two weeks.
- Avoid getting the panels and surrounding area wet [see Dosage and Administration (2.1)].
- Patients should avoid physical activity that may result in reduced adhesion or actual loss of the test panels [see Dosage and Administration (2.1)].
- Patients should avoid excessive sweating and keep the test panels and surrounding area dry [see Dosage and Administration (2.1)].
-
SPL UNCLASSIFIED SECTIONManufactured by SmartPractice Denmark ApS - Hillerød, Denmark - U.S. License No. 1888 - Distributed by SmartPractice - Phoenix, AZ 85008 - US Patents 4,836,217 and 5,182,081 ...
Manufactured by SmartPractice Denmark ApS
Close
Hillerød, Denmark
U.S. License No. 1888
Distributed by SmartPractice
Phoenix, AZ 85008
US Patents 4,836,217 and 5,182,081, 5,614,167 -
INSTRUCTIONS FOR USETHIN-LAYER RAPID USE - EPICUTANEOUS PATCH TEST - T.R.U.E. TEST - ® NDC: 67334-0457-1 - For further details of allergen constituents and dose, see outer box. Rx only - For ...
THIN-LAYER RAPID USE
EPICUTANEOUS PATCH TEST
T.R.U.E. TEST ®
NDC: 67334-0457-1
For further details of allergen constituents and dose, see outer box.
Rx only
For Topical Use OnlyStore between 2° and 8°C (36° and 46°F).
PANEL 1.3
Application Instructions
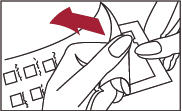
Peel open the package and remove the test panel.
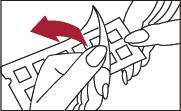
Remove the protective plastic covering from the test surface of the panel. Be careful not to touch the test substances.
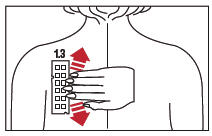
Position test Panel 1.3 on the patient's back. Allergen number 1 should be in the upper left corner. Avoid applying the panel on the margin of the scapula or directly over the midline of the spine. Ensure that each patch of the allergen panel is in contact with the skin by smoothing the panel outward from the center to the edge.
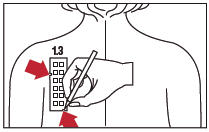
With a medical marking pen, indicate on the skin the location of the two notches on the panel.
Store between 2° and 8°C (36° and 46°F).
Refrigeration required. Do not freeze.Manufactured by SmartPractice Denmark ApS
Hillerød, Denmark
US License No. 1888Distributed by SmartPractice ®
Phoenix, AZ 85008
Made in Denmark51-2009-00/1
Close -
INSTRUCTIONS FOR USETHIN-LAYER RAPID USE - EPICUTANEOUS PATCH TEST - T.R.U.E. TEST - ® NDC: 67334-0457-1 - For further details of allergen constituents and dose, see outer box. Rx only - For Topical ...
THIN-LAYER RAPID USE
EPICUTANEOUS PATCH TEST
T.R.U.E. TEST ®
NDC: 67334-0457-1
For further details of allergen constituents and dose, see outer box.
Rx only
For Topical Use Only
Store between 2° and 8°C (36° and 46°F).
PANEL 2.3
Application Instructions
A desiccant is included in this package for stability purposes.
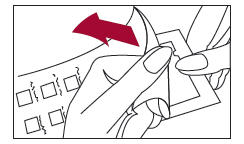
Peel open the package and remove the test panel.
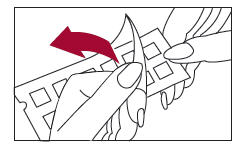
Remove the protective plastic covering from the test surface of the panel. Be careful not to touch the test substances.
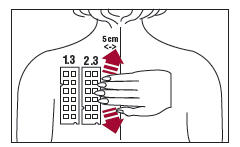
Position test Panel 2.3 on the patient's back. Allergen number 13 should be in the upper left corner. Avoid applying the panel on the margin of the scapula or directly over the midline of the spine. Ensure that each patch of the allergen panel is in contact with the skin by smoothing the panel outward from the center to the edge.
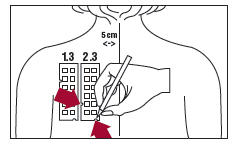
With a medical marking pen, indicate on the skin the location of the two notches on the panel.
Store between 2° and 8°C (36° and 46°F).
Refrigeration required. Do not freeze.Manufactured by SmartPractice Denmark ApS
Hillerød, Denmark
US License No. 1888Distributed by SmartPractice ®
Phoenix, AZ 85008
Made in Denmark51-2012-00/2
Close -
INSTRUCTIONS FOR USETHIN-LAYER RAPID USE - EPICUTANEOUS PATCH TEST - T.R.U.E. TEST - ® NDC: 67334-0457-1 - For further details of allergen constituents and dose, see outer box. Rx only - For ...
THIN-LAYER RAPID USE
EPICUTANEOUS PATCH TEST
T.R.U.E. TEST ®
NDC: 67334-0457-1
For further details of allergen constituents and dose, see outer box.
Rx only
For Topical Use OnlyStore between 2° and 8°C (36° and 46°F).
PANEL 3.3
Application Instructions
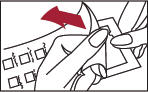
Peel open the package and remove the test panel.
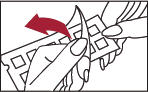
Remove the protective plastic covering from the test surface of the panel. Be careful not to touch the test substances.
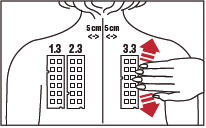
Position test panel 3.3 on the patient's back. Allergen number 25 should be in the upper left corner. Avoid applying the panel on the margin of the scapula or directly over the midline of the spine. Ensure that each patch of the allergen panel is in contact with the skin by smoothing the panel outward from the center to the edge.
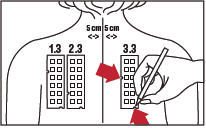
With a medical marking pen, indicate on the skin the location of the two notches on the panel.
Store between 2° and 8°C (36° and 46°F).
Refrigeration required. Do not freeze.Manufactured by SmartPractice Denmark ApS
Hillerød, Denmark
US License No. 1888Distributed by SmartPractice ®
Phoenix, AZ 85008
Made in Denmark51-2015-00/1
Close -
INSTRUCTIONS FOR USEPanel 1.3 - THIN-LAYER RAPID USE - EPICUTANEOUS PATCH TEST - T.R.U.E. TEST - ® Manufactured by SmartPractice Denmark ApS - Distributed by SmartPractice - ® Made in ...
Panel 1.3

THIN-LAYER RAPID USE
EPICUTANEOUS PATCH TEST
T.R.U.E. TEST ®Manufactured by SmartPractice Denmark ApS
Distributed by SmartPractice ®
Made in Denmark52-5115-00/1
Close -
INSTRUCTIONS FOR USEPanel 2.3 - THIN-LAYER RAPID USE - EPICUTANEOUS PATCH TEST - T.R.U.E. TEST - ® Manufactured by SmartPractice Denmark ApS - Distributed by SmartPractice - ® Made in ...
Panel 2.3

THIN-LAYER RAPID USE
EPICUTANEOUS PATCH TEST
T.R.U.E. TEST ®Manufactured by SmartPractice Denmark ApS
Distributed by SmartPractice ®
Made in Denmark52-5115-00/1
Close -
INSTRUCTIONS FOR USEPanel 3.3 - THIN-LAYER RAPID USE - EPICUTANEOUS PATCH TEST - T.R.U.E. TEST - ® Manufactured by SmartPractice Denmark ApS - Distributed by SmartPractice - ® Made in ...
Panel 3.3

THIN-LAYER RAPID USE
EPICUTANEOUS PATCH TEST
T.R.U.E. TEST ®Manufactured by SmartPractice Denmark ApS
Distributed by SmartPractice ®
Made in Denmark52-5125-00/1
Close -
PRINCIPAL DISPLAY PANEL - Kit CartonTHIN-LAYER RAPID USE - EPICUTANEOUS PATCH TEST - T.R.U.E. TEST - ® NDC No: 67334-0457-5Mcg/cm - 2 - PANEL - 1.3 - 1Nickel Sulfate200 - 2Wool Alcohols1000 - 3Neomycin ...
THIN-LAYER RAPID USE
EPICUTANEOUS PATCH TESTT.R.U.E. TEST ®
NDC No: 67334-0457-5 Mcg/cm 2 PANEL
1.31 Nickel Sulfate 200 2 Wool Alcohols 1000 3 Neomycin Sulfate 600 4 Potassium Dichromate 54 5 Caine Mix a 630 6 Fragrance Mix b 500 7 Colophony 1200 8 Paraben Mix c 1000 9 Negative Control — 10 Balsam of Peru 800 11 Ethylenediamine Dihydrochloride 50 12 Cobalt Dichloride 20 13 p-tert-Butylphenol Formaldehyde Resin 45 PANEL
2.314 Epoxy Resin 50 15 Carba Mix d 250 16 Black Rubber Mix e 75 17 Cl +Me -Isothiazolinone 4 18 Quaternium-15 100 19 Methyldibromo Glutaronitrile 5 20 p-Phenylenediamine 80 21 Formaldehyde f 180 22 Mercapto Mix g 75 23 Thimerosal 7 24 Thiuram Mix h 27 25 Diazolidinyl Urea 550 26 Quinoline Mix i 190 27 Tixocortol-21-Pivalate 3 PANEL
3.328 Gold Sodium Thiosulfate 75 29 Imidazolidinyl Urea 600 30 Budesonide 1 31 Hydrocortisone-17-Butyrate 20 32 Mercaptobenzothiazole 75 33 Bacitracin 600 34 Parthenolide 3 35 Disperse blue 106 50 36 2-Bromo-2-Nitropropane-1,3-diol 250 Each unit contains panel 1.3, panel 2.3 and panel 3.3.
A desiccant is included in panel 2.3 package for stability purposes.
Concentrations expressed in micrograms/centimeter squared.
Notations a-i are located on the back panel.Rx only
For Topical Use OnlyStore between 2° and 8°C
(36° and 46°F).
Refrigeration required.
Do not freeze.5
UnitsPUSH
TO OPENClose
-
INGREDIENTS AND APPEARANCEProduct Information
T.R.U.E. TEST THIN-LAYER RAPID USE PATCH TEST standardized allergenic kit Product Information Product Type STANDARDIZED ALLERGENIC Item Code (Source) NDC:67334-0457 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:67334-0457-5 5 in 1 BOX 1 NDC:67334-0457-1 1 in 1 BAG Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 PATCH 48 h Part 2 1 PATCH 48 h Part 3 1 PATCH 48 h Part 1 of 3 T.R.U.E. TEST PANEL 1.3 standardized allergenic patch Product Information Route of Administration CUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NICKEL SULFATE HEXAHYDRATE (UNII: JC9WZ4FK68) (NICKEL CATION - UNII:OIS2CXW7AM) NICKEL SULFATE HEXAHYDRATE 36 ug in 48 h LANOLIN ALCOHOLS (UNII: 884C3FA9HE) (LANOLIN ALCOHOLS - UNII:884C3FA9HE) LANOLIN ALCOHOLS 810 ug in 48 h NEOMYCIN SULFATE (UNII: 057Y626693) (NEOMYCIN - UNII:I16QD7X297) NEOMYCIN 486 ug in 48 h POTASSIUM DICHROMATE (UNII: T4423S18FM) (DICHROMATE ION - UNII:9LKY4BFN2V) POTASSIUM DICHROMATE 15.7 ug in 48 h BENZOCAINE (UNII: U3RSY48JW5) (BENZOCAINE - UNII:U3RSY48JW5) BENZOCAINE 378 ug in 48 h TETRACAINE HYDROCHLORIDE (UNII: 5NF5D4OPCI) (TETRACAINE - UNII:0619F35CGV) TETRACAINE HYDROCHLORIDE 66 ug in 48 h DIBUCAINE HYDROCHLORIDE (UNII: Z97702A5DG) (DIBUCAINE - UNII:L6JW2TJG99) DIBUCAINE HYDROCHLORIDE 66 ug in 48 h GERANIOL (UNII: L837108USY) (GERANIOL - UNII:L837108USY) GERANIOL 81 ug in 48 h CINNAMALDEHYDE (UNII: SR60A3XG0F) (CINNAMALDEHYDE - UNII:SR60A3XG0F) CINNAMALDEHYDE 41 ug in 48 h CINNAMYL ALCOHOL (UNII: SS8YOP444F) (CINNAMYL ALCOHOL - UNII:SS8YOP444F) CINNAMYL ALCOHOL 63 ug in 48 h HYDROXYCITRONELLAL (UNII: 8SQ0VA4YUR) (HYDROXYCITRONELLAL - UNII:8SQ0VA4YUR) HYDROXYCITRONELLAL 63 ug in 48 h EUGENOL (UNII: 3T8H1794QW) (EUGENOL - UNII:3T8H1794QW) EUGENOL 41 ug in 48 h ISOEUGENOL (UNII: 5M0MWY797U) (ISOEUGENOL - UNII:5M0MWY797U) ISOEUGENOL 17 ug in 48 h .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) (.ALPHA.-AMYLCINNAMALDEHYDE - UNII:WC51CA3418) .ALPHA.-AMYLCINNAMALDEHYDE 17 ug in 48 h EVERNIA PRUNASTRI (UNII: O3034Q5AHK) (EVERNIA PRUNASTRI - UNII:O3034Q5AHK) EVERNIA PRUNASTRI 81 ug in 48 h ROSIN (UNII: 88S87KL877) (ROSIN - UNII:88S87KL877) ROSIN 972 ug in 48 h PROPYLPARABEN (UNII: Z8IX2SC1OH) (PROPYLPARABEN - UNII:Z8IX2SC1OH) PROPYLPARABEN 162 ug in 48 h METHYLPARABEN (UNII: A2I8C7HI9T) (METHYLPARABEN - UNII:A2I8C7HI9T) METHYLPARABEN 162 ug in 48 h ETHYLPARABEN (UNII: 14255EXE39) (ETHYLPARABEN - UNII:14255EXE39) ETHYLPARABEN 162 ug in 48 h BUTYLPARABEN (UNII: 3QPI1U3FV8) (BUTYLPARABEN - UNII:3QPI1U3FV8) BUTYLPARABEN 162 ug in 48 h BENZYLPARABEN (UNII: 8Y41DYV4VG) (BENZYLPARABEN - UNII:8Y41DYV4VG) BENZYLPARABEN 162 ug in 48 h BALSAM PERU (UNII: 8P5F881OCY) (BALSAM PERU - UNII:8P5F881OCY) BALSAM PERU 648 ug in 48 h ETHYLENEDIAMINE (UNII: 60V9STC53F) (ETHYLENEDIAMINE - UNII:60V9STC53F) ETHYLENEDIAMINE 18 ug in 48 h COBALTOUS CHLORIDE HEXAHYDRATE (UNII: 17AVG63ZBC) (COBALTOUS CATION - UNII:AI1MR454XG) COBALTOUS CHLORIDE HEXAHYDRATE 4 ug in 48 h Inactive Ingredients Ingredient Name Strength HYDROXYPROPYL CELLULOSE (1600000 WAMW) (UNII: RFW2ET671P) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) BETADEX (UNII: JV039JZZ3A) BUTYLATED HYDROXYANISOLE (UNII: REK4960K2U) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 1 in 1 POUCH 1 48 h in 1 PATCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA103738 06/01/2013 Part 2 of 3 T.R.U.E. TEST PANEL 2.3 standardized allergenic patch Product Information Route of Administration CUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIPENTAMETHYLENETHIURAM DISULFIDE (UNII: CR113982E5) (DIPENTAMETHYLENETHIURAM DISULFIDE - UNII:CR113982E5) DIPENTAMETHYLENETHIURAM DISULFIDE 5.5 ug in 48 h P-TERT-BUTYLPHENOL-FORMALDEHYDE RESIN (LOW MOLECULAR WEIGHT) (UNII: Q51LPW21CH) (P-TERT-BUTYLPHENOL-FORMALDEHYDE RESIN (LOW MOLECULAR WEIGHT) - UNII:Q51LPW21CH) P-TERT-BUTYLPHENOL-FORMALDEHYDE RESIN (LOW MOLECULAR WEIGHT) 36 ug in 48 h BISPHENOL A DIGLYCIDYL ETHER (UNII: F3XRM1NX4H) (BISPHENOL A DIGLYCIDYL ETHER - UNII:F3XRM1NX4H) BISPHENOL A DIGLYCIDYL ETHER 32 ug in 48 h DIPHENYLGUANIDINE (UNII: 6MRZ85RNHQ) (DIPHENYLGUANIDINE - UNII:6MRZ85RNHQ) DIPHENYLGUANIDINE 68 ug in 48 h DITIOCARB ZINC (UNII: ICW4708Z8G) (DITIOCARB ZINC - UNII:ICW4708Z8G) DITIOCARB ZINC 68 ug in 48 h ZINC DIBUTYLDITHIOCARBAMATE (UNII: HNM5J934VP) (ZINC DIBUTYLDITHIOCARBAMATE - UNII:HNM5J934VP) ZINC DIBUTYLDITHIOCARBAMATE 68 ug in 48 h N-ISOPROPYL-N'-PHENYL-1,4-PHENYLENEDIAMINE (UNII: 0M7PSL4100) (N-ISOPROPYL-N'-PHENYL-1,4-PHENYLENEDIAMINE - UNII:0M7PSL4100) N-ISOPROPYL-N'-PHENYL-1,4-PHENYLENEDIAMINE 10 ug in 48 h N-CYCLOHEXYL-N'-PHENYL-P-PHENYLENEDIAMINE (UNII: T29JGK5V4R) (N-CYCLOHEXYL-N'-PHENYL-P-PHENYLENEDIAMINE - UNII:T29JGK5V4R) N-CYCLOHEXYL-N'-PHENYL-P-PHENYLENEDIAMINE 25 ug in 48 h N,N'-DIPHENYL-P-PHENYLENEDIAMINE (UNII: DD517SCM92) (N,N'-DIPHENYL-P-PHENYLENEDIAMINE - UNII:DD517SCM92) N,N'-DIPHENYL-P-PHENYLENEDIAMINE 25 ug in 48 h METHYLCHLOROISOTHIAZOLINONE (UNII: DEL7T5QRPN) (METHYLCHLOROISOTHIAZOLINONE - UNII:DEL7T5QRPN) METHYLCHLOROISOTHIAZOLINONE 3 ug in 48 h QUATERNIUM-15 (UNII: E40U03LEM0) (QUATERNIUM-15 - UNII:E40U03LEM0) QUATERNIUM-15 81 ug in 48 h METHYLDIBROMO GLUTARONITRILE (UNII: YX089CPS05) (METHYLDIBROMO GLUTARONITRILE - UNII:YX089CPS05) METHYLDIBROMO GLUTARONITRILE 4 ug in 48 h P-PHENYLENEDIAMINE (UNII: U770QIT64J) (P-PHENYLENEDIAMINE - UNII:U770QIT64J) P-PHENYLENEDIAMINE 65 ug in 48 h FORMALDEHYDE (UNII: 1HG84L3525) (FORMALDEHYDE - UNII:1HG84L3525) FORMALDEHYDE 146 ug in 48 h N-CYCLOHEXYL-2-BENZOTHIAZOSULFENAMIDE (UNII: UCA53G94EV) (N-CYCLOHEXYL-2-BENZOTHIAZOSULFENAMIDE - UNII:UCA53G94EV) N-CYCLOHEXYL-2-BENZOTHIAZOSULFENAMIDE 20 ug in 48 h 2,2'-DITHIOBISBENZOTHIAZOLE (UNII: 6OK753033Z) (2,2'-DITHIOBISBENZOTHIAZOLE - UNII:6OK753033Z) 2,2'-DITHIOBISBENZOTHIAZOLE 20 ug in 48 h MORPHOLINYLMERCAPTOBENZOTHIAZOLE (UNII: VCD7623F3K) (MORPHOLINYLMERCAPTOBENZOTHIAZOLE - UNII:VCD7623F3K) MORPHOLINYLMERCAPTOBENZOTHIAZOLE 20 ug in 48 h THIMEROSAL (UNII: 2225PI3MOV) (ETHYLMERCURITHIOSALICYLIC ACID - UNII:4071N4J64W) THIMEROSAL 6 ug in 48 h TETRAMETHYLTHIURAM MONOSULFIDE (UNII: 01W430XXSQ) (TETRAMETHYLTHIURAM MONOSULFIDE - UNII:01W430XXSQ) TETRAMETHYLTHIURAM MONOSULFIDE 5.5 ug in 48 h THIRAM (UNII: 0D771IS0FH) (THIRAM - UNII:0D771IS0FH) THIRAM 5.5 ug in 48 h DISULFIRAM (UNII: TR3MLJ1UAI) (DISULFIRAM - UNII:TR3MLJ1UAI) DISULFIRAM 5.5 ug in 48 h Inactive Ingredients Ingredient Name Strength HYDROXYPROPYL CELLULOSE (1600000 WAMW) (UNII: RFW2ET671P) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) SODIUM BICARBONATE (UNII: 8MDF5V39QO) SODIUM CARBONATE (UNII: 45P3261C7T) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 1 in 1 POUCH 1 48 h in 1 PATCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA103738 06/01/2013 Part 3 of 3 T.R.U.E. TEST PANEL 3.3 standardized allergenic patch Product Information Route of Administration CUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) (DIAZOLIDINYL UREA - UNII:H5RIZ3MPW4) DIAZOLIDINYL UREA 446 ug in 48 h CLIOQUINOL (UNII: 7BHQ856EJ5) (CLIOQUINOL - UNII:7BHQ856EJ5) CLIOQUINOL 77 ug in 48 h CHLORQUINALDOL (UNII: D6VHC87LLS) (CHLORQUINALDOL - UNII:D6VHC87LLS) CHLORQUINALDOL 77 ug in 48 h TIXOCORTOL PIVALATE (UNII: 6K28E35M3B) (TIXOCORTOL - UNII:ZX3KEK657Z) TIXOCORTOL PIVALATE 2 ug in 48 h GOLD SODIUM THIOSULFATE (UNII: CKS1YQ9W1J) (SODIUM CATION - UNII:LYR4M0NH37) GOLD SODIUM THIOSULFATE 23 ug in 48 h IMIDUREA (UNII: M629807ATL) (IMIDUREA - UNII:M629807ATL) IMIDUREA 486 ug in 48 h BUDESONIDE (UNII: Q3OKS62Q6X) (BUDESONIDE - UNII:Q3OKS62Q6X) BUDESONIDE 0.8 ug in 48 h HYDROCORTISONE BUTYRATE (UNII: 05RMF7YPWN) (HYDROCORTISONE - UNII:WI4X0X7BPJ) HYDROCORTISONE BUTYRATE 16 ug in 48 h 2-MERCAPTOBENZOTHIAZOLE (UNII: 5RLR54Z22K) (2-MERCAPTOBENZOTHIAZOLE - UNII:5RLR54Z22K) 2-MERCAPTOBENZOTHIAZOLE 61 ug in 48 h BACITRACIN (UNII: 58H6RWO52I) (BACITRACIN - UNII:58H6RWO52I) BACITRACIN 486 ug in 48 h PARTHENOLIDE (UNII: 2RDB26I5ZB) (PARTHENOLIDE - UNII:2RDB26I5ZB) PARTHENOLIDE 2 ug in 48 h DISPERSE BLUE 106 (UNII: C48O4QYD6N) (DISPERSE BLUE 106 - UNII:C48O4QYD6N) DISPERSE BLUE 106 41 ug in 48 h BRONOPOL (UNII: 6PU1E16C9W) (BRONOPOL - UNII:6PU1E16C9W) BRONOPOL 203 ug in 48 h Inactive Ingredients Ingredient Name Strength POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) HYDROXYPROPYL CELLULOSE (1600000 WAMW) (UNII: RFW2ET671P) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 1 in 1 POUCH 1 48 h in 1 PATCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA103738 06/01/2013 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA103738 03/01/2012
CloseLabeler - SmartPractice Denmark ApS (306216144)
Find additional resources
(also available in the left menu)Safety
Report Adverse Events, FDA Safety Recalls, Presence in Breast Milk
Related Resources
Medline Plus, Clinical Trials, PubMed, Biochemical Data Summary
More Info on this Drug
View Labeling Archives, RxNorm, Get Label RSS Feed, View NDC Code(s)NEW!
View Labeling Archives for this drug
T.R.U.E. TEST THIN-LAYER RAPID USE PATCH TEST- standardized allergenic kit
Number of versions: 9
RxNorm
T.R.U.E. TEST THIN-LAYER RAPID USE PATCH TEST- standardized allergenic kit
Out of scope - Out of scope for RxNorm and will not receive RxNorm normal forms. Out of scope information includes radiopharmaceuticals, contrast media, herbals, homeopathics, and food. Drug names that are ambiguous or not compatible with the RxNorm system, such as multivitamins with more than 4,000 characters in their names, are also out of scope.
Get Label RSS Feed for this Drug
T.R.U.E. TEST THIN-LAYER RAPID USE PATCH TEST- standardized allergenic kit
To receive this label RSS feed
Copy the URL below and paste it into your RSS Reader application.
https://dailymed.nlm.nih.gov/dailymed/labelrss.cfm?setid=2f082b68-dc74-418a-9e6f-b3c285b41d44
To receive all DailyMed Updates for the last seven days
Copy the URL below and paste it into your RSS Reader application.
https://dailymed.nlm.nih.gov/dailymed/rss.cfm
What will I get with the DailyMed RSS feed?
DailyMed will deliver notification of updates and additions to Drug Label information currently shown on this site through its RSS feed.
DailyMed will deliver this notification to your desktop, Web browser, or e-mail depending on the RSS Reader you select to use. To view updated drug label links, paste the RSS feed address (URL) shown below into a RSS reader, or use a browser which supports RSS feeds, such as Safari for Mac OS X.
How to discontinue the RSS feed
If you no longer wish to have this DailyMed RSS service, simply delete the copied URL from your RSS Reader.
More about getting RSS News & Updates from DailyMedWhy is DailyMed no longer displaying pill images on the Search Results and Drug Info pages?
Due to inconsistencies between the drug labels on DailyMed and the pill images provided by RxImage, we no longer display the RxImage pill images associated with drug labels.
We anticipate reposting the images once we are able identify and filter out images that do not match the information provided in the drug labels.
NDC Codes
T.R.U.E. TEST THIN-LAYER RAPID USE PATCH TEST- standardized allergenic kit
If this SPL contains inactivated NDCs listed by the FDA initiated compliance action, they will be specified as such.
| NDC | |
|---|---|
| 1 | 67334-0457-1 |
| 2 | 67334-0457-5 |

