Label: CUROSURF- poractant alfa suspension
- NDC Code(s): 10122-510-01, 10122-510-03
- Packager: Chiesi USA, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Biologic Licensing Application
Drug Label Information
Updated May 2, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use CUROSURF® safely and effectively. See full prescribing information for CUROSURF.
CUROSURF (poractant alfa) intratracheal suspension
Initial U.S. Approval: 1999INDICATIONS AND USAGE
CUROSURF is a surfactant indicated for the rescue treatment, including the reduction of mortality and pneumothoraces, of Respiratory Distress Syndrome (RDS) in premature infants. (1)
DOSAGE AND ADMINISTRATION
- Before administering CUROSURF, assure proper placement and patency of endotracheal tube (2.1)
- Administer intratracheally either in (2.1):
-
Two divided aliquots through a 5 French end-hole catheter; or
-
A single bolus through secondary lumen of a dual lumen endotracheal tube without interrupting mechanical ventilation
-
- Initial recommended dose is 2.5 mL/kg birth weight (2.2)
- Up to two repeat doses of 1.25 mL/kg birth weight may be administered at approximately 12-hour intervals (2.2)
- Maximum total dose (initial plus repeat doses) is 5 mL/kg (2.2)
- See Full Prescribing Information for instructions on preparation and administration of the CUROSURF suspension (2.3, 2.4)
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
None. (4)
WARNINGS AND PRECAUTIONS
- Acute Changes in Lung Compliance: Frequently assess need to modify oxygen and ventilatory support to respiratory changes (5.1)
- Administration-Related Adverse Reactions: Transient adverse effects include bradycardia, hypotension, endotracheal tube blockage, and oxygen desaturation. These events require stopping CUROSURF administration and taking appropriate measures to alleviate the condition (5.2)
ADVERSE REACTIONS
- Common adverse reactions associated with the administration of CUROSURF include bradycardia, hypotension, endotracheal tube blockage, and oxygen desaturation. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Chiesi USA, Inc. at 1-888-661-9260 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Revised: 5/2022
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Instructions
2.2 Recommended Dosage
2.3 Preparation of the CUROSURF Suspension
2.4 Administration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Acute Changes in Oxygenation and Lung Compliance
5.2 Administration-Related Adverse Reactions
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Immunogenicity
6.3 Postmarketing Experience
8 USE IN SPECIFIC POPULATIONS
8.4 Pediatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Rescue Treatment of Respiratory Distress Syndrome
16 HOW SUPPLIED/STORAGE AND HANDLING
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Instructions
For intratracheal administration only.
CUROSURF should be administered by, or under the supervision of clinicians experienced in intubation, ventilator management, and general care of premature infants. Before administering CUROSURF, assure proper placement and patency of the endotracheal tube. At the discretion of the clinician, the endotracheal tube may be suctioned before administering CUROSURF. Allow the infant to stabilize before proceeding with dosing.
Administer CUROSURF either:
- Intratracheally by instillation in two divided aliquots through a 5 French end-hole catheter or
- Intratracheally in a single bolus through the secondary lumen of a dual lumen endotracheal tube without interrupting mechanical ventilation.
2.2 Recommended Dosage
The initial recommended dose is 2.5 mL/kg birth weight, administered as one or two aliquots depending upon the instillation procedure [see Dosage and Administration (2.4)].
Up to two repeat doses of 1.25 mL/kg birth weight each may be administered at approximately 12-hour intervals in infants in whom RDS is considered responsible for their persisting or deteriorating respiratory status. The maximum recommended total dosage (sum of the initial and up to two repeat doses) is 5 mL/kg.
2.3 Preparation of the CUROSURF Suspension
- Remove the vial of CUROSURF suspension from a refrigerator at +2°C to +8°C (36°F to 46°F) and slowly warm the vial to room temperature before use.
- Visually inspect the CUROSURF suspension for discoloration prior to administration. The color of the CUROSURF suspension should be white to creamy white. Discard the CUROSURF vial if the suspension is discolored.
- Gently turn the vial upside-down, in order to obtain a uniform suspension. DO NOT SHAKE.
- Locate the notch (FLIP UP) on the colored plastic cap and lift the notch and pull upwards.
- Pull the plastic cap with the aluminum portion downwards.
- Remove the whole ring by pulling off the aluminum wrapper.
- Remove the rubber cap to extract content.
- Unopened, unused vials of CUROSURF suspension that have warmed to room temperature can be returned to refrigerated storage within 24 hours for future use. Do not warm to room temperature and return to refrigerated storage more than once. Protect from light.
2.4 Administration
For endotracheal tube instillation using a 5 French end-hole catheter
- Slowly withdraw the entire contents of the vial of CUROSURF suspension into a 3 or 5 mL plastic syringe through a large-gauge needle (e.g., at least 20 gauge). Enter each single-use vial only once.
- Attach the 5 French end-hole catheter of appropriate length to position the catheter tip proximal to the distal portion of the endotracheal tube, to the syringe. Fill the catheter with CUROSURF suspension. Discard excess CUROSURF through the catheter so that only the dose to be given remains in the syringe.
- For the first dose: 1.25 mL/kg (birth weight) per aliquot
- For each repeated dose: 0.625 mL/kg (birth weight) per aliquot
- First aliquot of CUROSURF suspension:
- Position the infant in a neutral position (head and body in alignment without inclination), with either the right or left side dependent.
- Immediately before CUROSURF administration, ventilate the infant with supplemental oxygen sufficient to maintain SaO2 > 92%.
- Insert the catheter into the endotracheal tube and instill the first aliquot of CUROSURF suspension.
- After the first aliquot is instilled, remove the catheter from the endotracheal tube and manually ventilate with supplemental oxygen until clinically stable.
- Second aliquot of CUROSURF suspension:
- When the infant is stable, reposition the infant such that the other side is dependent.
- Administer the remaining aliquot using the same procedures as the first aliquot.
- After completion of the dosing procedure, do not suction airways for 1 hour after surfactant instillation unless signs of significant airway obstruction occur [see Clinical Studies (14.1)].
For endotracheal tube instillation using the secondary lumen of a dual lumen endotracheal tube
- Slowly withdraw the entire contents of the vial of CUROSURF suspension into a 3 or 5 mL plastic syringe through a large-gauge needle (e.g., at least 20 gauge). Do not attach 5 French end-hole catheter. Remove the needle and discard excess CUROSURF so that only the dose to be given remains in the syringe.
- Keep the infant in a neutral position (head and body in alignment without inclination).
- Administer CUROSURF suspension through the proximal end of the secondary lumen of the endotracheal tube as a single dose, given over 1 minute, and without interrupting mechanical ventilation.
- After completion of this dosing procedure, ventilator management may require transient increases in FiO2 , ventilator rate, or PIP. Do not suction airways for 1 hour after surfactant instillation unless signs of significant airway obstruction occur.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Acute Changes in Oxygenation and Lung Compliance
The administration of exogenous surfactants, including CUROSURF, can rapidly affect oxygenation and lung compliance. Therefore, infants receiving CUROSURF should receive frequent clinical and laboratory assessments so that oxygen and ventilatory support can be modified to respond to respiratory changes. CUROSURF should only be administered by those trained and experienced in the care, resuscitation, and stabilization of pre-term infants.
5.2 Administration-Related Adverse Reactions
Transient adverse reactions associated with administration of CUROSURF include bradycardia, hypotension, endotracheal tube blockage, and oxygen desaturation. These events require stopping CUROSURF administration and taking appropriate measures to alleviate the condition. After the patient is stable, dosing may proceed with appropriate monitoring.
-
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice.
Adverse Reactions in Studies in Premature Infants with Respiratory Distress Syndrome
The safety data described below reflect exposure to CUROSURF at a single dose of 2.5 mL/kg (200 mg/kg), in 78 infants of 700-2000 grams birth weight with RDS requiring mechanical ventilation and a FiO2 ≥ 0.60 (Study 1) [see Clinical Studies (14.1)]. A total of 144 infants were studied after RDS developed and before 15 hours of age; 78 infants received CUROSURF 2.5 mL/kg single dose (200 mg/kg), and 66 infants received control treatment (disconnection from the ventilator and manual ventilation for 2 minutes).
Transient adverse reactions seen with the administration of CUROSURF included bradycardia, hypotension, endotracheal tube blockage, and oxygen desaturation. The rates of the most common serious complications associated with prematurity and RDS observed in Study 1 are shown in Table 1.
Table 1: Most Common Serious Complications Associated with Prematurity and RDS in Study 1
CUROSURF 2.5 mL/kg
n=78
CONTROL*
n=66
Acquired Pneumonia
17%
21%
Acquired Septicemia
14%
18%
Bronchopulmonary Dysplasia
18%
22%
Intracranial Hemorrhage
51%
64%
Patent Ductus Arteriosus
60%
48%
Pneumothorax
21%
36%
Pulmonary Interstitial Emphysema
21%
38%
*Control patients were disconnected from the ventilator and manually ventilated for 2 minutes. No surfactant was instilled.
Seventy-six infants (45 treated with CUROSURF) from study 1 were evaluated at 1 year of age and 73 infants (44 treated with CUROSURF) were evaluated at 2 years of age to assess for potential long-term adverse reactions. Data from follow-up evaluations for weight and length, persistent respiratory symptoms, incidence of cerebral palsy, visual impairment, or auditory impairment was similar between treatment groups. In 16 patients (10 treated with CUROSURF and 6 controls) evaluated at 5.5 years of age, the developmental quotient, derived using the Griffiths Mental Developmental Scales, was similar between groups.
-
8 USE IN SPECIFIC POPULATIONS
8.4 Pediatric Use
The safety and effectiveness of CUROSURF for the rescue treatment, including the reduction of mortality and pneumothoraces, of Respiratory Distress Syndrome (RDS) have been established in premature infants and the information on this use is discussed throughout the labeling.
The safety and effectiveness of CUROSURF in the treatment of full term infants or older pediatric patients with respiratory failure has not been established.
-
10 OVERDOSAGE
There have been no reports of overdosage following the administration of CUROSURF.
In the event of accidental overdosage, and if there are clear clinical effects on the infant's respiration, ventilation, or oxygenation, aspirate as much of the suspension as possible and provide the infant with supportive treatment, with particular attention to fluid and electrolyte balance.
-
11 DESCRIPTION
Poractant alfa is an extract of natural porcine lung (pulmonary) surfactant consisting of 99% polar lipids (mainly phospholipids) and 1% hydrophobic low molecular weight surfactant associated proteins (SP). The molecular weight of SP-B is 8.7 KDa and the molecular weight of SP-C is 3.7 KDa.
CUROSURF (poractant alfa) intratracheal suspension is a sterile, white to creamy white suspension provided in a single-dose vial for intratracheal use. Each milliliter of suspension contains 80 mg of poractant alfa (surfactant extract) that includes 76 mg of phospholipids and 1 mg of SP of which 0.45 mg is SP-B, a 79-amino acid protein and 0.59 mg is SP-C, a 35-amino acid peptide . The amount of phospholipids is calculated from the content of phosphorus and contains 55 mg of phosphatidylcholine of which 30 mg is dipalmitoylphosphatidylcholine. It is suspended in 0.9% sodium chloride solution. The pH is adjusted with sodium bicarbonate to a pH of 6.2 (5.5 to 6.5).
Curosurf contains no preservatives.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Endogenous pulmonary surfactant reduces surface tension at the air-liquid interface of the alveoli during ventilation and stabilizes the alveoli against collapse at resting transpulmonary pressures. A deficiency of pulmonary surfactant in preterm infants results in Respiratory Distress Syndrome (RDS) characterized by poor lung expansion, inadequate gas exchange, and a gradual collapse of the lungs (atelectasis).
CUROSURF compensates for the deficiency of surfactant and restores surface activity to the lungs of these infants.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies to assess potential carcinogenic effects of CUROSURF have not been conducted.
Poractant alfa was negative for genotoxicity in the following assays: bacterial reverse mutation assay (Ames test), gene mutation assay in Chinese hamster V79 cells, chromosomal aberration assay in Chinese hamster ovary cells, unscheduled DNA synthesis in HELA S3 cells, and in vivo mouse micronucleus assay.
No studies to assess reproductive effects of CUROSURF have been performed.
-
14 CLINICAL STUDIES
14.1 Rescue Treatment of Respiratory Distress Syndrome
The clinical efficacy of CUROSURF in the treatment of established Respiratory Distress Syndrome (RDS) in premature infants was demonstrated in one single-dose study (Study 1) and one multiple-dose study (Study 2) involving approximately 500 infants. Each study was randomized, multicenter, and controlled.
In study 1, premature infants 700 to 2000 grams birth weight with RDS requiring mechanical ventilation and a FiO2 ≥ 0.60 were enrolled. CUROSURF 2.5 mL/kg single dose (200 mg/kg) or control (disconnection from the ventilator and manual ventilation for 2 minutes) was administered after RDS developed and before 15 hours of age. The results from Study 1 are shown below in Table 2.
Table 2: Study 1 Results in Premature Infants with Respiratory Distress Syndrome
Efficacy Parameter
Single Dose
CUROSURF
n=78
Control
n=67
p-Value
Mortality at 28 Days (All Causes)
31%
48%
≤0.05
Bronchopulmonary Dysplasia*
18%
22%
N.S.
Pneumothorax
21%
36%
≤0.05
Pulmonary Interstitial Emphysema
21%
38%
≤0.05
*Bronchopulmonary dysplasia (BPD) diagnosed by positive x-ray and supplemental oxygen dependence at 28 days of life.
N.S.: not statistically significant
In Study 2, premature infants 700 to 2000 g birth weight with RDS requiring mechanical ventilation and a FiO2 ≥ 0.60 were enrolled. In this two-arm trial, CUROSURF was administered after RDS developed and before 15 hours of age, as a single-dose or as multiple doses. In the single-dose arm, infants received CUROSURF 2.5 mL/kg (200 mg/kg). In the multiple-dose arm, the initial dose of CUROSURF was 2.5 mL/kg followed by up to two 1.25 mL/kg (100 mg/kg) doses of CUROSURF. The results from Study 2 are shown below in Table 3.
Table 3: Study 2 Results in Premature Infants with Respiratory Distress Syndrome
Efficacy Parameter
Single Dose
CUROSURF
n=184
Rate (%)
Multiple Dose
CUROSURF
n=173
Rate (%)
p-Value
Mortality at 28 Days (All Causes)
21
13
0.048
Bronchopulmonary Dysplasia*
18
18
N.S.
Pneumothorax
17
9
0.03
Pulmonary Interstitial Emphysema
27
22
N.S.
*Bronchopulmonary dysplasia (BPD) diagnosed by positive x-ray and supplemental oxygen dependence at 28 days of life.
N.S.: not statistically significant
There is no controlled experience on the effects of administering initial doses of CUROSURF other than 2.5 mL/kg (200 mg/kg), subsequent doses other than 1.25 mL/kg (100 mg/kg), administration of more than three total doses, dosing more frequently than every 12 hours, or initiating therapy with CUROSURF more than 15 hours after diagnosing RDS. Adequate data are not available on the use of CUROSURF in conjunction with experimental therapies of RDS, e.g., high-frequency ventilation or extracorporeal membrane oxygenation.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
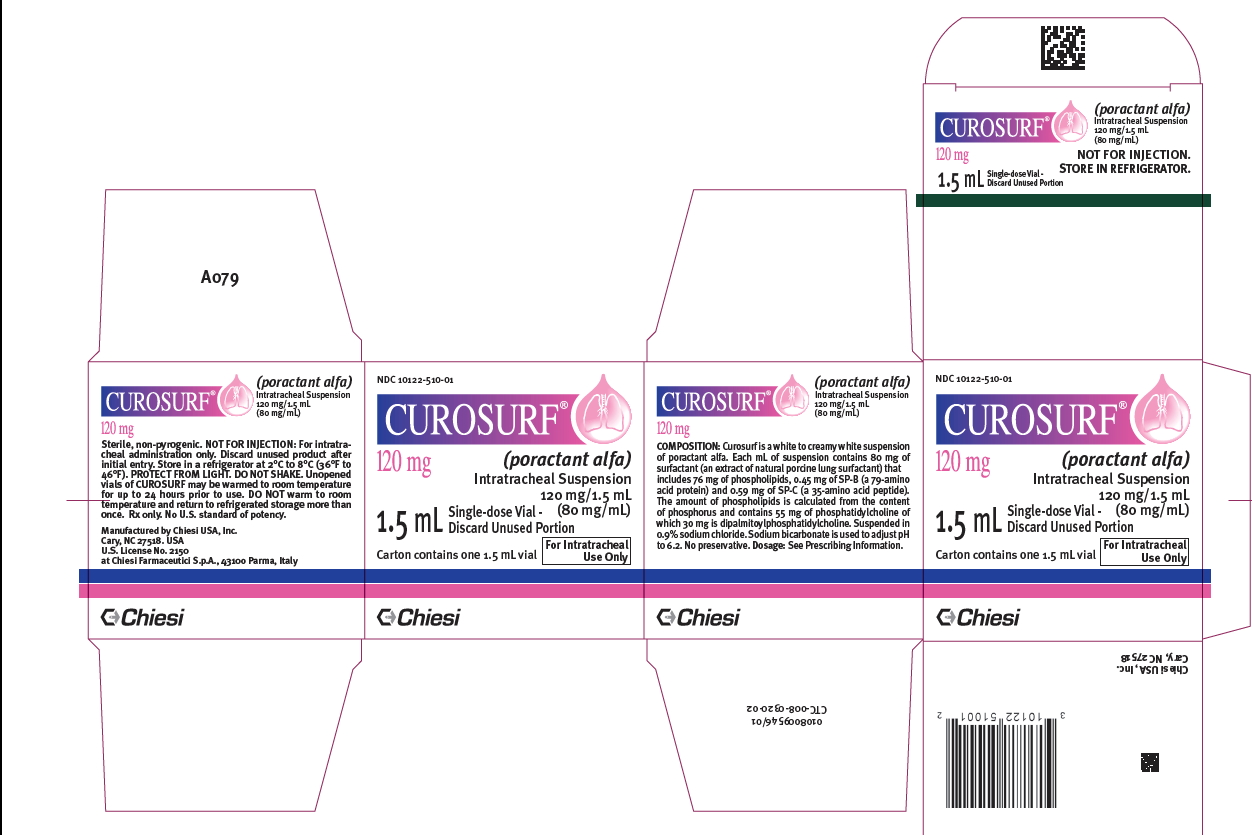
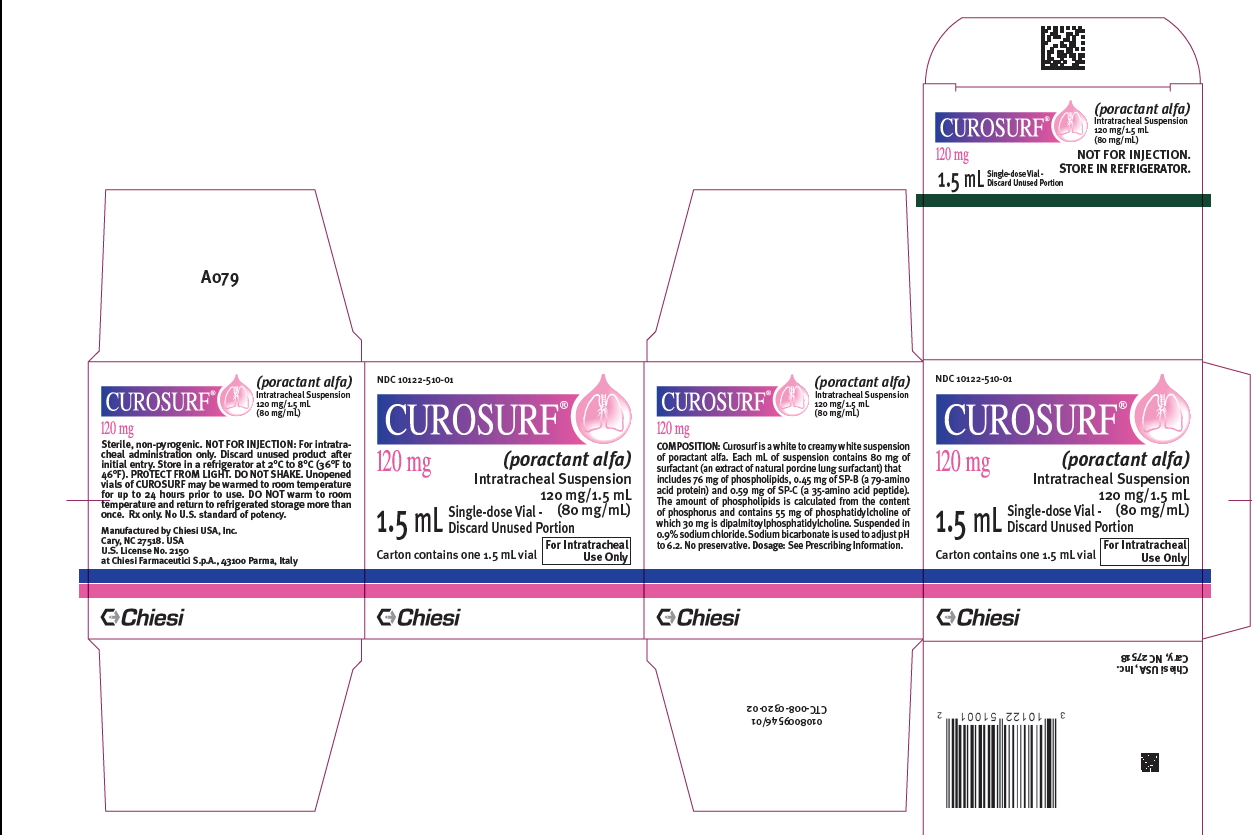
CUROSURF (poractant alfa) intratracheal suspension is a white to creamy white suspension available in sterile, rubber-stoppered clear glass vials containing (one vial per carton):
- 120 mg/1.5 mL (80 mg/mL) poractant alfa (surfactant extract) of suspension: NDC Number: 10122-510-01
- 240 mg/3 mL (80 mg/mL) poractant alfa (surfactant extract) of suspension. NDC Number: 10122-510-03
Store CUROSURF intratracheal suspension in a refrigerator at +2°C to +8°C (36°F to 46°F). PROTECT FROM LIGHT. Do not shake. Vials are for single-dose only. After opening the vial discard the unused portion [see Dosage and Administration (2.3)].
Manufactured by:
Chiesi USA, Inc.
Cary, NC 27518
U.S.License No. 2150
Manufactured at and licensed from:
Chiesi Farmaceutici, S.p.A.
Parma, Italy 43100
CTC-007-0320-03-SPL-1
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CUROSURF
poractant alfa suspensionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:10122-510 Route of Administration ENDOTRACHEAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PORACTANT ALFA (UNII: KE3U2023NP) (PORACTANT ALFA - UNII:KE3U2023NP) PORACTANT ALFA 80 mg in 1 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM BICARBONATE (UNII: 8MDF5V39QO) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10122-510-03 3.0 mL in 1 VIAL, GLASS; Type 0: Not a Combination Product 11/18/1999 2 NDC:10122-510-01 1.5 mL in 1 VIAL, GLASS; Type 0: Not a Combination Product 11/18/1999 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA020744 11/18/1999 Labeler - Chiesi USA, Inc. (088084228) Registrant - Chiesi USA, Inc. (088084228)