Label: ATTRUBY- acoramidis hydrochloride tablet, film coated
- NDC Code(s): 82228-712-28
- Packager: BridgeBio Pharma, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated November 26, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use ATTRUBY safely and effectively. See full prescribing information for ATTRUBY. ATTRUBY™ (acoramidis) tablets, for oral ...
-
Table of ContentsTable of Contents
-
1 INDICATIONS AND USAGE
ATTRUBY is indicated for the treatment of the cardiomyopathy of wild-type or variant transthyretin-mediated amyloidosis (ATTR-CM) in adults to reduce cardiovascular death and ...
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage - The recommended dosage of ATTRUBY is 712 mg orally twice daily (with or without food). Swallow tablets whole; do not cut, crush, or chew.
-
3 DOSAGE FORMS AND STRENGTHS
ATTRUBY is available as 356 mg acoramidis, white, film-coated, oval tablets, printed with the BridgeBio company logo followed by “ACOR” in black ink on one side.
-
4 CONTRAINDICATIONS
None.
-
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience - Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly ...
-
7 DRUG INTERACTIONS
UDP-glucuronosyltransferases (UGT) Inducers and Strong CYP3A Inducers - Acoramidis is metabolized by UGT enzyme-mediated glucuronidation. Concomitant use of UGT inducers can potentially ...
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy - Risk Summary - Available data with acoramidis use in pregnant women are insufficient to establish a drug associated risk of major birth defects, miscarriage or other adverse ...
-
10 OVERDOSAGE
There is no clinical experience with overdose. In case of suspected overdose, treatment should be symptomatic and supportive.
-
11 DESCRIPTION
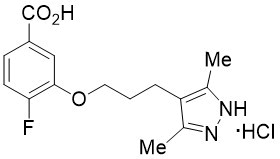
ATTRUBY contains 356 mg acoramidis equivalent to 400 mg acoramidis HCl. Acoramidis HCl is a transthyretin stabilizer. The chemical name of acoramidis HCl is ...
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action - Acoramidis is a selective stabilizer of transthyretin (TTR). Acoramidis binds TTR at thyroxine binding sites and slows dissociation of the TTR tetramer into its ...
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility - Carcinogenesis - There was no evidence of increased incidence of neoplasia in a 2-year carcinogenicity study in male rats dosed up ...
-
14 CLINICAL STUDIES
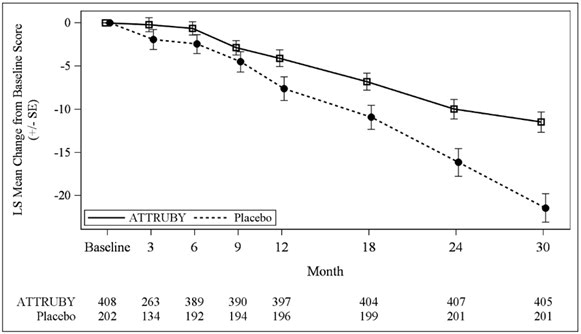
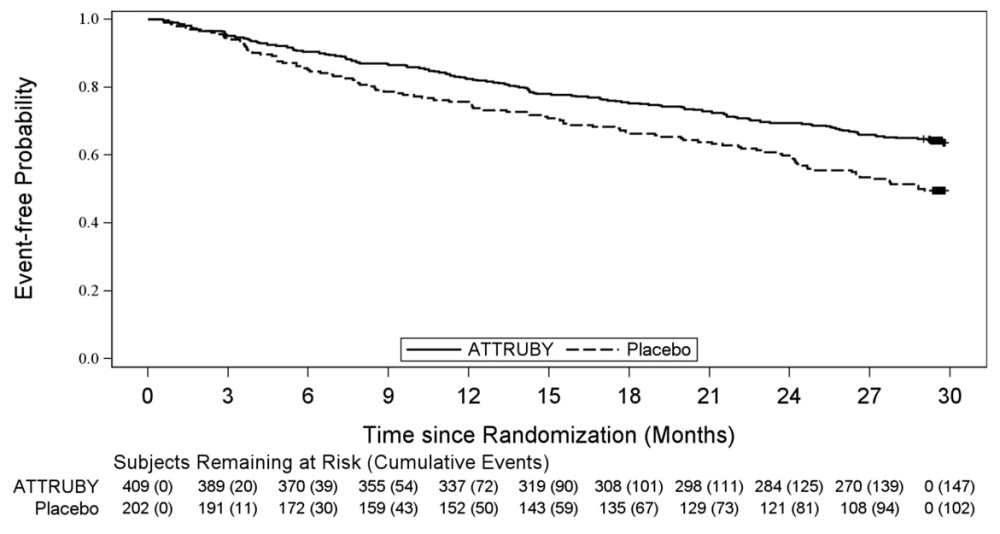
The efficacy of ATTRUBY was demonstrated in a multicenter, international, randomized, double-blind, placebo-controlled study in 611 adult patients with wild-type or variant (hereditary or de novo ...
-
16 HOW SUPPLIED/STORAGE AND HANDLING
ATTRUBY (acoramidis) tablets, 356 mg, are white, film-coated, oval tablets, printed with the BridgeBio company logo followed by “ACOR” in black ink on one side. ATTRUBY tablets are supplied as a ...
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information). Pregnancy - Advise patients who are exposed to ATTRUBY during pregnancy to contact the BridgeBio reporting ...
-
PATIENT PACKAGE INSERTThis Patient Information has been approved by the U.S. Food and Drug Administration. Issued: 11/2024 - PATIENT INFORMATION - ATTRUBYTM (ah-troo-be) (acoramidis) tablets - What is ...
-
PRINCIPAL DISPLAY PANELPrincipal Display Panel – 356 mg Carton Label - NDC 82228-712-28 - Rx only - bridgebio - Attruby™ (acoramidis) tablets - 356 mg per tablet - Take two 356 mg tablets two times a day - (712 mg dose ...
-
INGREDIENTS AND APPEARANCEProduct Information