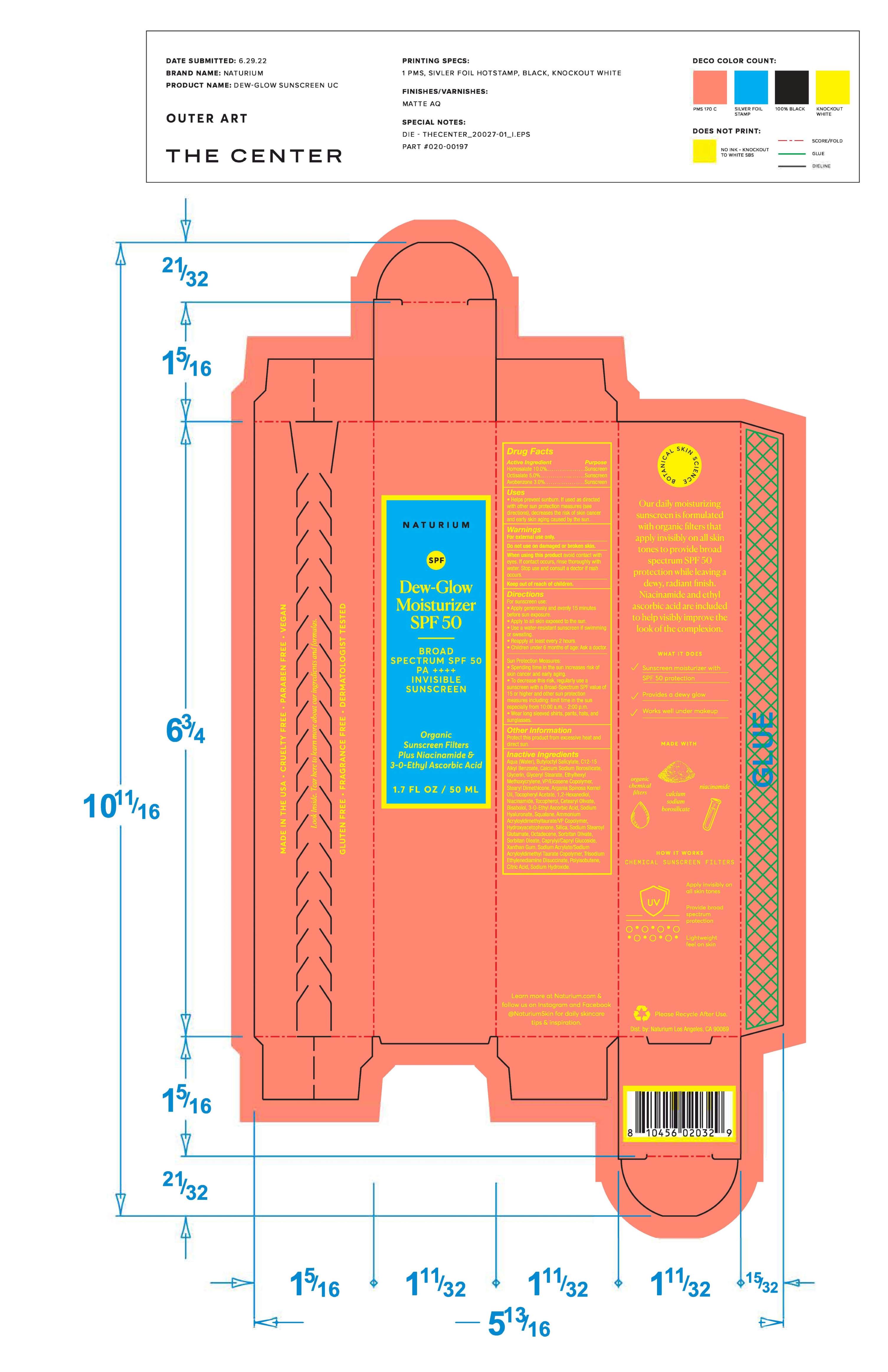
Label: NATURIUM DEW-GLOW MOISTURIZER SPF 50 cream
- NDC Code(s): 59735-338-01
- Packager: MANA Products, Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 3, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purpose
- Uses
- Warnings
- Warnings-When using
- Keep out of reach of children
-
Directions
Apply liberally 15 minutes before sun exposure. Use a water-resistant sunscreen if swimming or sweating. Reapply at least every 2 hours, Children under 6 months: ask a doctor.
Sun Protection Measures: Spending time in the sun increases risk of skin cancer and early aging. To decrease
this risk, regularly use a sunscreen with a Broad-Spectrum SPF value of 15 or higher and other sun protection measures including: limit time in the sun especially from 10:00 a.m. - 2:00 p.m. Wear long sleeved shirts, pants, hats, and sunglasses. -
Inactive Ingredients
Aqua/Water/Eau, Butyloctyl Salicylate, C12-15 Alkyl Benzoate, Calcium Sodium Borosilicate, Glycerin, Glyceryl Stearate, Ethylhexyl Methoxycrylene, VP/Eicosene Copolymer, Stearyl Dimethicone, Argania Spinosa Kernel Oil, Tocopheryl Acetate, 1,2-Hexanediol, Niacinamide, Tocopherol, Cetearyl Olivate, Ammonium Acryloyldimethyltaurate/VP Copolymer, Hydroxyacetophenone, Silica, Sodium Stearoyl Glutamate, Octadecene, Sorbitan Olivate, Xanthan Gum, Bisabolol, 3-o-ethyl ascorbic acid, Sodium Hyaluronate, Squalane, Sodium Acrylate/Sodium Acryloyldimethyl Taurate Copolymer, Trisodium Ethylenediamine Disuccinate, Polyisobutene, Citric Acid, Sodium Hydroxide, Sorbitan Oleate, Caprylyl/Capryl Glucoside.
- Other Information
- Principle Display Panel
-
INGREDIENTS AND APPEARANCE
NATURIUM DEW-GLOW MOISTURIZER SPF 50
naturium dew-glow moisturizer spf 50 creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:59735-338 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 5 g in 1 mL AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 3 g in 1 mL HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 10 g in 1 mL Inactive Ingredients Ingredient Name Strength EICOSYL POVIDONE (UNII: XQQ9MKE2BJ) STEARYL DIMETHICONE (400 MPA.S AT 50C) (UNII: R327X197HY) CETEARYL OLIVATE (UNII: 58B69Q84JO) AMMONIUM ACRYLOYLDIMETHYLTAURATE/VP COPOLYMER (UNII: W59H9296ZG) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) POLYISOBUTYLENE (65000 MW) (UNII: VC9JLP352J) CAPRYLYL/CAPRYL OLIGOGLUCOSIDE (UNII: E00JL9G9K0) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) .ALPHA.-BISABOLOL, (+)- (UNII: 105S6I733Z) TRISODIUM ETHYLENEDIAMINE DISUCCINATE (UNII: YA22H34H9Q) SORBITAN OLIVATE (UNII: MDL271E3GR) ARGAN OIL (UNII: 4V59G5UW9X) .ALPHA.-TOCOPHEROL, DL- (UNII: 7QWA1RIO01) SQUALANE (UNII: GW89575KF9) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) .ALPHA.-TOCOPHEROL ACETATE, D- (UNII: A7E6112E4N) HYDROXYETHYL ACRYLATE/SODIUM ACRYLOYLDIMETHYL TAURATE COPOLYMER (45000 MPA.S AT 1%) (UNII: 86FQE96TZ4) SODIUM HYDROXIDE (UNII: 55X04QC32I) ETHYLHEXYL METHOXYCRYLENE (UNII: S3KFG6Q5X8) HYDROXYACETOPHENONE (UNII: G1L3HT4CMH) SODIUM STEAROYL GLUTAMATE (UNII: 65A9F4P024) OCTADECENE (UNII: H5ZUQ6V4AK) XANTHAN GUM (UNII: TTV12P4NEE) 1,2-HEXANEDIOL (UNII: TR046Y3K1G) 3-O-ETHYL ASCORBIC ACID (UNII: 6MW60CB71P) HYALURONATE SODIUM (UNII: YSE9PPT4TH) NIACINAMIDE (UNII: 25X51I8RD4) SORBITAN MONOOLEATE (UNII: 06XEA2VD56) WATER (UNII: 059QF0KO0R) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) CALCIUM ALUMINUM BOROSILICATE (UNII: 3JRB8A35M0) GLYCERIN (UNII: PDC6A3C0OX) GLYCERYL STEARATE SE (UNII: FCZ5MH785I) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59735-338-01 1 in 1 CARTON 11/01/2022 1 50 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 11/01/2022 Labeler - MANA Products, Inc (078870292) Establishment Name Address ID/FEI Business Operations MANA Products, Inc 032870270 manufacture(59735-338) Establishment Name Address ID/FEI Business Operations MANA Products Inc 078870292 manufacture(59735-338)