Label: NOVIPET- glucosamine sulfate, chondrotin sulfate, dimethyl sulfone tablet, chewable
- NDC Code(s): 45732-013-30, 45732-013-60
- Packager: Novipet, Inc.
- Category: OTC ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated June 30, 2012
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- INACTIVE INGREDIENT
-
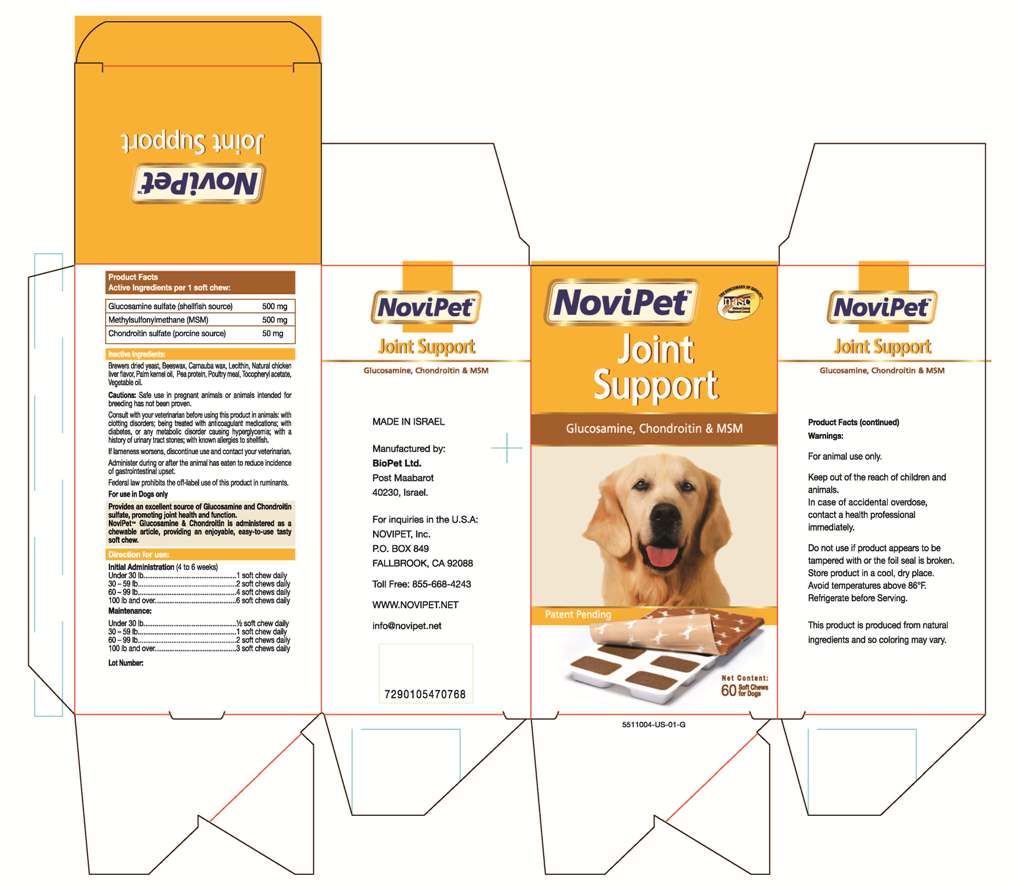
DOSAGE & ADMINISTRATION
Directions for use:
Initial Administration (4 to 6 weeks)
Under 30 lb...................1 soft chew daily
30 -59 lb.......................2 soft chews daily
60 -99 lb.......................4 soft chews daily
100 lb and over.. .......... 6 soft chews daily
Maintenance:
Under 30 lb................. 1/2 soft chew daily
30 -59 lb ......................1 soft chew daily
60 -99 lb.......................2 soft Chews daily
100 lb and over.. .......... 3 soft chews daily - WARNINGS
-
WARNINGS AND PRECAUTIONS
Cautions: Safe use in pregnant animals or animals intended for
Do not use if product appears to be tampered
breeding has not been proven.
Consult with veterinarian before using this product in animals; with
clotting disorders (or being treated with anticoagulant medications); with
diabetes, or any metabolic disorder causing hyperglycemia; with
a history of urinary tract stones; with known allergies to shellfish.
If lameness worsens, discontinue use and contact your veterinarian.
Administer during or after the animal has eaten to reduce incidence
of gastrointestinal upset.
Federal law prohibits the off-label use of this product in ruminants.
For use in Dogs only
with or the foil seal is broken.
- DESCRIPTION
- STORAGE AND HANDLING
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
NOVIPET
glucosamine sulfate, chondrotin sulfate, dimethyl sulfone tablet, chewableProduct Information Product Type OTC ANIMAL DRUG Item Code (Source) NDC:45732-013 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GLUCOSAMINE SULFATE (UNII: 1FW7WLR731) (GLUCOSAMINE - UNII:N08U5BOQ1K) GLUCOSAMINE 500 mg CHONDROITIN SULFATE SODIUM (PORCINE) (UNII: R27OH35FYQ) (CHONDROITIN SULFATE (PORCINE) - UNII:V5E8ELO4W9) CHONDROITIN SULFATE (PORCINE) 50 mg DIMETHYL SULFONE (UNII: 9H4PO4Z4FT) (DIMETHYL SULFONE - UNII:9H4PO4Z4FT) DIMETHYL SULFONE 500 mg Product Characteristics Color brown (Brown Chewable Tablet) Score no score Shape SQUARE Size 10mm Flavor Imprint Code none Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:45732-013-30 30 in 1 CARTON 2 NDC:45732-013-60 60 in 1 CARTON Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 05/15/2012 Labeler - Novipet, Inc. (078388924) Registrant - Biopet Ltd (532294121) Establishment Name Address ID/FEI Business Operations Biopet Ltd 532294121 manufacture Establishment Name Address ID/FEI Business Operations Bioiberica SA 460067325 api manufacture Establishment Name Address ID/FEI Business Operations Tianjin Zhongan Pharmaceutical Co. Ltd 544911639 manufacture Establishment Name Address ID/FEI Business Operations Maabarot Products Ltd 600472187 analysis Establishment Name Address ID/FEI Business Operations Milouda Laboratories 533761631 analysis