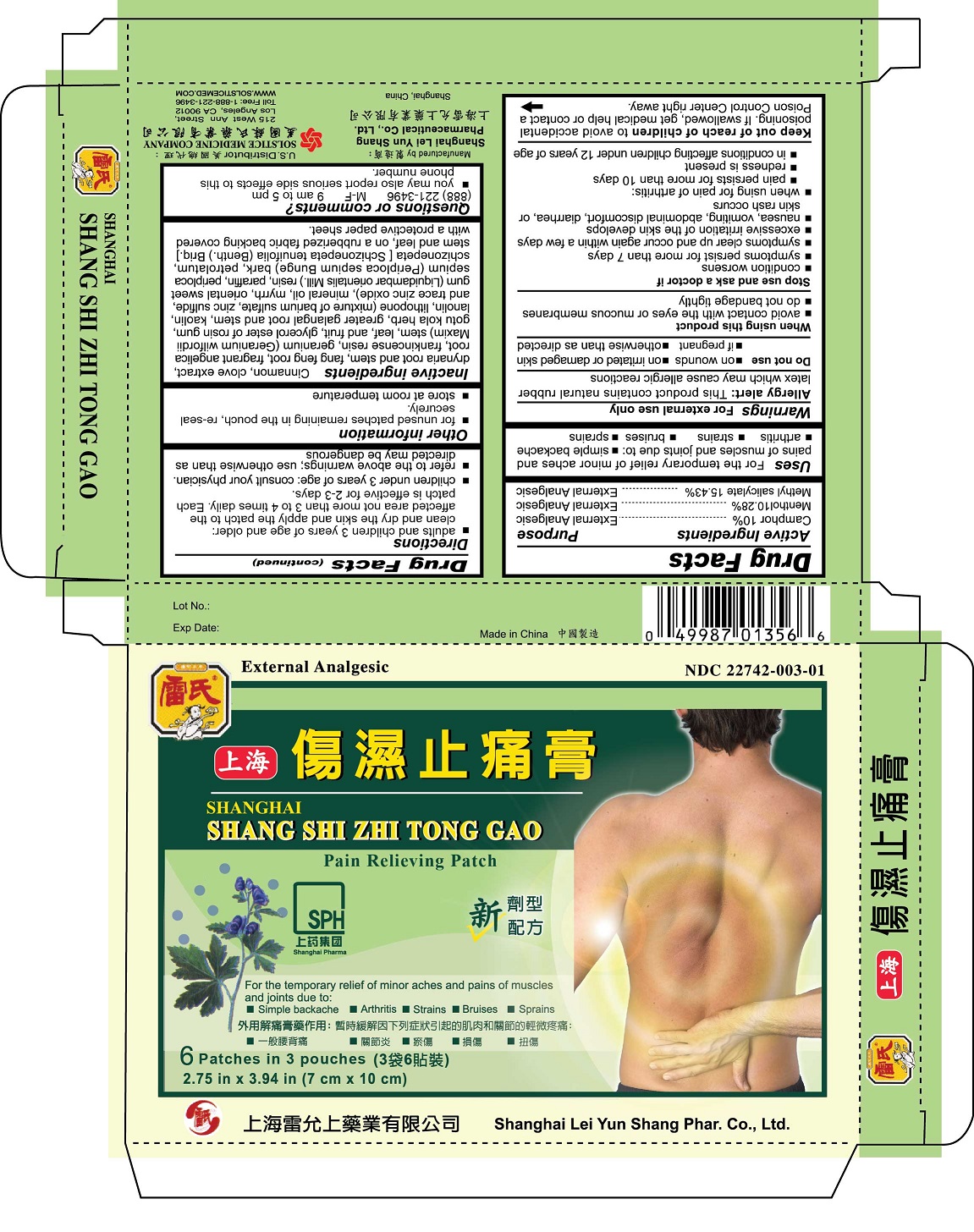
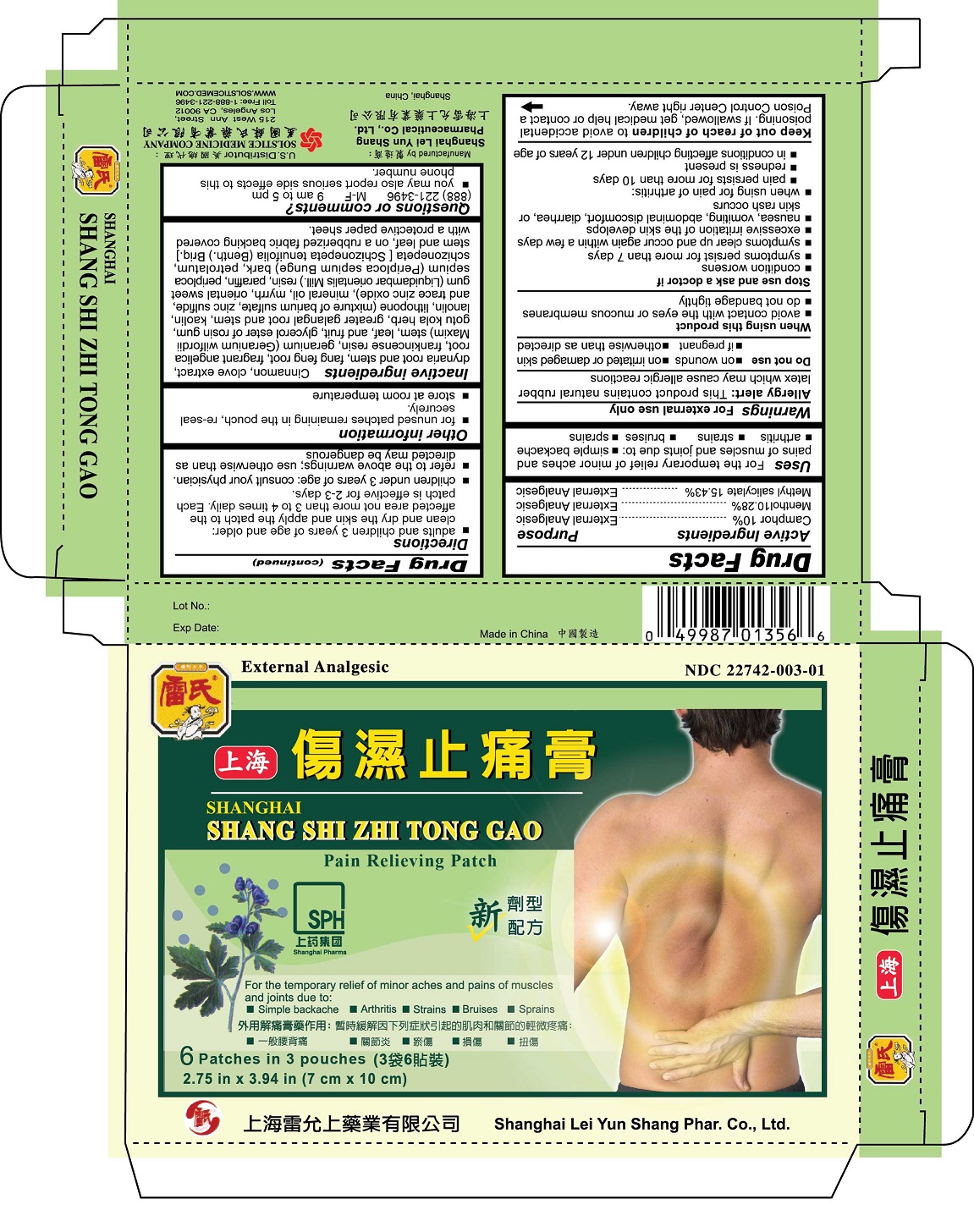
Label: SHANGHAI SHANG SHI ZHI TONG GAO PAIN RELIEVING- camphor, menthol, methyl salicylate patch
-
Contains inactivated NDC Code(s)
NDC Code(s): 22742-003-01 - Packager: SHANGHAI LEI YUN SHANG PHARMACEUTICAL CO LTD
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated November 13, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- DO NOT USE
- WHEN USING
-
STOP USE
Stop use and ask a doctor if
condition worsens
symptoms persist for more than 7 days
symptoms clear up and occur again within a few days
excessive irritation of the skin develops
nausea, vomiting, abdominal discomfort, diarrhea, or skin rash occurs
when using for pain of arthritis:
pain persists for more than 10 days
redness is present
in conditions affecting children under 12 years of age - KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
Directions
adults and children 3 years of age and older: clean and dry the skin and apply the patch to the affected area not than 3 to 4 times daily. Each patch is effective for 2-3 days.
children under 3 years of age: consult your physician.
refer to the above warnings; use otherwise than as directed may be dangerous. - STORAGE AND HANDLING
-
INACTIVE INGREDIENT
Inactive ingredientsCinnamon, clove extract, drynaria root and stem, fang feng root, fragrant angelica root, frankincense resin, geranium (Geranium wilfordii Maxim) stem, leaf, and fruit, glycerol ester of rosin gum, gotu kola herb, greater galangal root and stem, kaolin, lanolin, lithopone (mixture of barium sulfate, zinc sulfide, and trace zinc oxide), mineral oil, myrrh, oriental sweet gum (Liquidambar orientalis Mill.) resin, paraffin, periploca sepium (Periploca sepium Bunge) bark, petrolatum, schizonepeta [Schizonepeta tenuifolia (Benth.) Briq.] stem and leaf, on a rubberized fabric backing covered with a protective paper sheet.
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SHANGHAI SHANG SHI ZHI TONG GAO PAIN RELIEVING
camphor, menthol, methyl salicylate patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:22742-003 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) (CAMPHOR (SYNTHETIC) - UNII:5TJD82A1ET) CAMPHOR (SYNTHETIC) 10 MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 10.28 METHYL SALICYLATE (UNII: LAV5U5022Y) (SALICYLIC ACID - UNII:O414PZ4LPZ) METHYL SALICYLATE 15.43 Inactive Ingredients Ingredient Name Strength CINNAMON (UNII: 5S29HWU6QB) CLOVE (UNII: K48IKT5321) DRYNARIA FORTUNEI ROOT (UNII: 731W842X8Q) SAPOSHNIKOVIA DIVARICATA ROOT (UNII: 8H84LFK2QD) ANGELICA DAHURICA ROOT (UNII: 1V63N2S972) FRANKINCENSE (UNII: R9XLF1R1WM) GERANIUM WILFORDII WHOLE (UNII: QXI974Y72W) ROSIN PARTIALLY DIMERIZED GLYCEROL ESTER (UNII: BY866DXY67) CENTELLA ASIATICA (UNII: 7M867G6T1U) ALPINIA GALANGA WHOLE (UNII: 3S1W839C0O) KAOLIN (UNII: 24H4NWX5CO) LANOLIN (UNII: 7EV65EAW6H) BARIUM SULFATE (UNII: 25BB7EKE2E) ZINC SULFIDE (UNII: KPS085631O) ZINC OXIDE (UNII: SOI2LOH54Z) MINERAL OIL (UNII: T5L8T28FGP) MYRRH (UNII: JC71GJ1F3L) LIQUIDAMBAR ORIENTALIS WHOLE (UNII: 759QNU787E) PARAFFIN (UNII: I9O0E3H2ZE) PERIPLOCA SEPIUM ROOT BARK (UNII: 638OW484M3) PETROLATUM (UNII: 4T6H12BN9U) SCHIZONEPETA TENUIFOLIA WHOLE (UNII: C1107616TR) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:22742-003-01 3 in 1 BOX 03/09/2009 1 2 in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 03/09/2009 Labeler - SHANGHAI LEI YUN SHANG PHARMACEUTICAL CO LTD (420978637) Registrant - SHANGHAI LEI YUN SHANG PHARMACEUTICAL CO LTD (420978637) Establishment Name Address ID/FEI Business Operations SHANGHAI LEI YUN SHANG PHARMACEUTICAL CO LTD 420978637 manufacture(22742-003)