Label: DOTERRA SUN FACE DAILY MOISTURIZER- zinc oxide lotion
- NDC Code(s): 71630-147-15
- Packager: doTERRA International, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 26, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
-
Mineral Sunscreen Daily Moisturizer
This dermatologist-tested, mineral-based, broad-spectrum
sun protection moisturizer delivers exponential
benefits so your skin can look and act as young as
you feel. Formulated with non-nano zinc oxide,
doTERRA sun Face Mineral Sunscreen Daily Moisturizer
provides lightweight SPF 28 UVA/UVB defense
against the sun and blue light protection.
Rich in antioxidants, vitamin E, and nourishing
botanicals such as evodia, grapeseed oil, and CPTG®
essential oils, this lightweight, hydrating sunscreen
moisturizer is intended for everyday use. - INDICATIONS & USAGE
- WARNINGS
- WHEN USING
-
OTHER SAFETY INFORMATION
Sun Protection Measures. Spending time in the sun
increases your risk of skin cancer and early skin
aging. To decrease this risk, regularly use a sunscreen
with a Broad Spectrum SPF value of 15 or higher and
other sun protection measures including: • limit
time in the sun, especially from 10 a.m.–2 p.m. •
wear long-sleeved shirts, pants, hats, and sunglasses -
INACTIVE INGREDIENT
Inactive ingredients Water (Aqua), Caprylic/Capric
Triglyceride, Butyloctyl Salicylate, Propanediol, Vitis
vinifera (Grape) Seed Oil, Cetearyl Alcohol, Cetyl
Alcohol, Evodia rutaecarpa (Evodia) Fruit Extract,
Persea gratissima (Avocado) Oil, Daucus carota sativa
(Carrot) Seed Oil, Boswellia carterii (Frankincense) Oil,
Helichrysum italicum Flower/Leaf/Stem Oil,
Astrocaryum tucuma Seed Butter, Rubus idaeus
(Raspberry) Seed Oil, Sodium Stearoyl Glutamate,
Arachidyl Alcohol, Caprylyl Glycol, Coco-Glucoside,
Xanthan Gum, Helianthus annuus (Sunflower) Seed Oil,
Cocos nucifera (Coconut) Fruit Extract, Michelia alba
(Magnolia) Leaf Oil, Cymbopogon flexuosus
(Lemongrass) Oil, Cananga odorata (Ylang Ylang)
Flower Oil, Aloe barbadensis Leaf Juice, Michelia alba
(Magnolia) Flower Oil, Ethyl Ferulate, Behenyl Alcohol,
Arachidyl Glucoside, Citric Acid, Sodium Gluconate,
Bisabolol, Caprylhydroxamic Acid, Tocopherol,
Polyhydroxystearic Acid, Phytosterols, Squalene, Citrus
nobilis (Mandarin Orange) Peel Oil - STORAGE AND HANDLING
- QUESTIONS
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- doTERRA sun Face Mineral Sunscreen Daily Moisturizer
-
INGREDIENTS AND APPEARANCE
DOTERRA SUN FACE DAILY MOISTURIZER
zinc oxide lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71630-147 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 13 g in 100 g Inactive Ingredients Ingredient Name Strength ASTROCARYUM MURUMURU SEED BUTTER (UNII: 12V64UPU6R) CAPRYLIC/CAPRIC ACID (UNII: DI775RT244) CITRUS NOBILIS WHOLE (UNII: U29I7N5FLM) CYMBOPOGON FLEXUOSUS WHOLE (UNII: 0MK18YOA5G) ALOE VERA LEAF (UNII: ZY81Z83H0X) EVODIA RUTAECARPA FRUIT, IMMATURE (UNII: F7M0RB6Y4I) MICHELIA ALBA LEAF OIL (UNII: 002RK9L1FN) POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) ARACHIDYL ALCOHOL (UNII: 1QR1QRA9BU) ARACHIDYL GLUCOSIDE (UNII: 6JVW35JOOJ) PROPANEDIOL (UNII: 5965N8W85T) BOSWELLIA SERRATA RESIN OIL (UNII: 5T1XCE6K8K) ETHYL FERULATE (UNII: 5B8915UELW) SUNFLOWER OIL (UNII: 3W1JG795YI) GLUCOSE PENTAACETATE (UNII: 3040885R4N) RASPBERRY SEED OIL (UNII: 9S8867952A) DODECANEDIOIC ACID (UNII: 978YU42Q6I) SODIUM GLUCONATE (UNII: R6Q3791S76) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) CAPRYLYL GLYCOL (UNII: 00YIU5438U) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM BENZOATE (UNII: OJ245FE5EU) CANANGA ODORATA FLOWER (UNII: 76GTF6Z97M) CARROT SEED OIL (UNII: 595AO13F11) COCOS NUCIFERA WHOLE (UNII: 245J88W96L) MICHELIA ALBA WHOLE (UNII: TKZ4AVY41O) SODIUM STEAROYL GLUTAMATE (UNII: 65A9F4P024) GRAPE SEED OIL (UNII: 930MLC8XGG) .ALPHA.-BISABOLOL, (+)- (UNII: 105S6I733Z) COCO GLUCOSIDE (UNII: ICS790225B) XANTHAN GUM (UNII: TTV12P4NEE) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) BUTYLOCTANOL (UNII: N442D9VO79) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) CETYL ALCOHOL (UNII: 936JST6JCN) CAPRYLHYDROXAMIC ACID (UNII: UPY805K99W) TOCOPHEROL (UNII: R0ZB2556P8) SQUALENE (UNII: 7QWM220FJH) WATER (UNII: 059QF0KO0R) AVOCADO OIL (UNII: 6VNO72PFC1) HELICHRYSUM ITALICUM FLOWER OIL (UNII: O97ZV7726K) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71630-147-15 50 g in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 05/01/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 05/01/2023 Labeler - doTERRA International, LLC (832274935) Establishment Name Address ID/FEI Business Operations Bell International Laboratories 967781555 manufacture(71630-147)


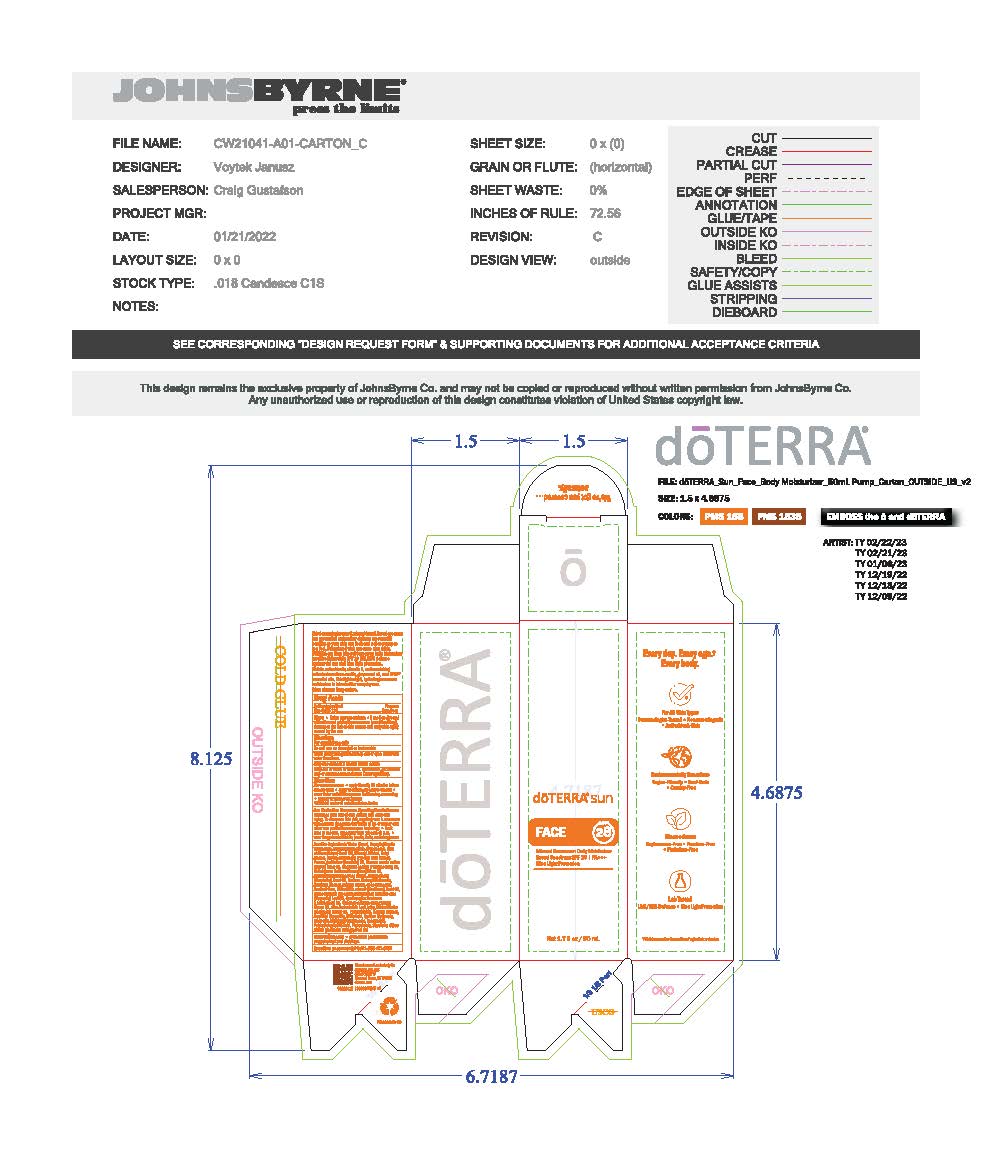

 Primary Label and Packaging
Primary Label and Packaging