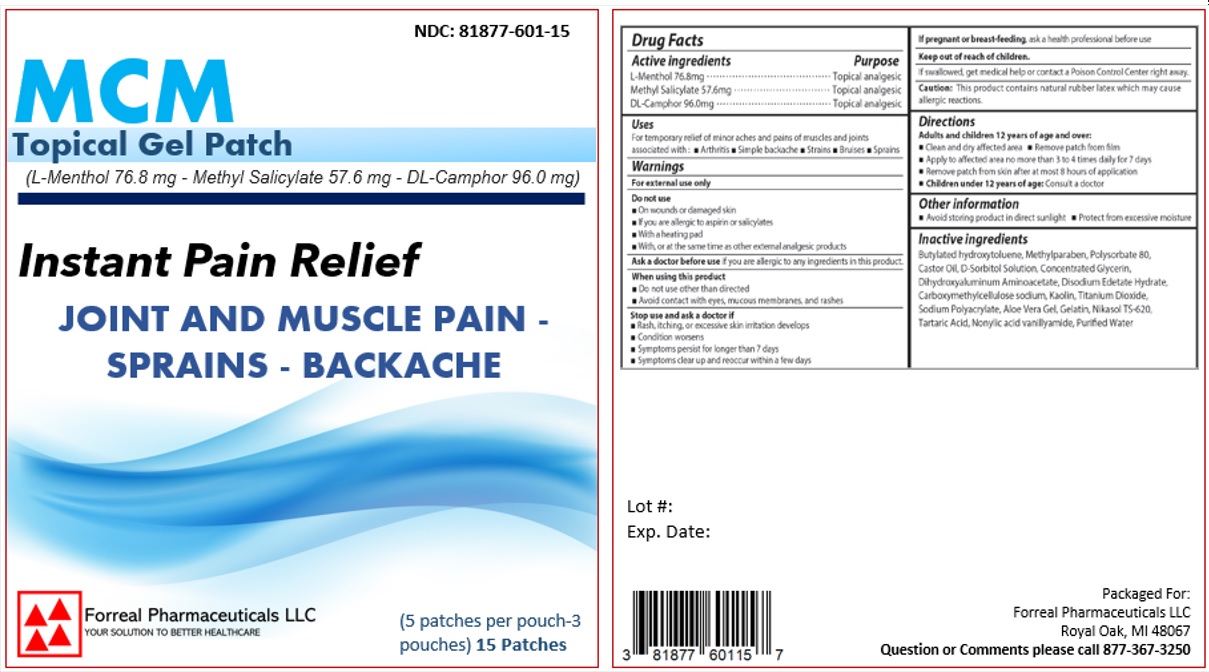
Label: MCM TOPICAL GEL PATCH- l-menthol patch
- NDC Code(s): 81877-601-15
- Packager: FORREAL PHARMACEUTICALS LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated January 23, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings
For external use only
Do not use- On wounds or damaged skin
- If you are allergic to aspirin or salicylates
- With a heating pad
- With, or at the same time as other external analgesic products
Ask a doctor before use if you are allergic to any ingredients in this product.
When using this product
- Do not use other than directed
- Avoid contact with eyes, mucous membranes, and rashes
Stop use and ask a doctor if
- Rash, itching, or excessive skin irritation develops
- Condition worsens
- Symptoms persist for longer than 7 days
- Symptoms clear up and reoccur within a few days
If pregnant or breast-feeding, ask a health professional before use
- Keep out of reach of children.
- DOSAGE & ADMINISTRATION
- OTHER SAFETY INFORMATION
-
INACTIVE INGREDIENT
Inactive Ingredients
Butylated hydroxytoluene, Methylparaben, Polysorbate 80, Castor Oil, D-Sorbitol Solution, Concentrated Glycerin, Dihydroxyaluminum Aminoacetate, Disodium Edetate Hydrate, Carboxymethylcellulose sodium, Kaolin, Titanium Dioxide, Sodium Polyacrylate, Aloe Vera Gel, Gelatin, Nikasol TS-620, Tartaric Acid, Nonylic acid vanillyamide, Purified water
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MCM TOPICAL GEL PATCH
l-menthol patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:81877-601 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEVOMENTHOL (UNII: BZ1R15MTK7) (LEVOMENTHOL - UNII:BZ1R15MTK7) LEVOMENTHOL 76.8 mg METHYL SALICYLATE (UNII: LAV5U5022Y) (SALICYLIC ACID - UNII:O414PZ4LPZ) METHYL SALICYLATE 57.6 mg CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) (CAMPHOR (SYNTHETIC) - UNII:5TJD82A1ET) CAMPHOR (SYNTHETIC) 96.0 mg Inactive Ingredients Ingredient Name Strength BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) METHYLPARABEN (UNII: A2I8C7HI9T) POLYSORBATE 80 (UNII: 6OZP39ZG8H) CASTOR OIL (UNII: D5340Y2I9G) SORBITOL SOLUTION (UNII: 8KW3E207O2) GLYCERIN (UNII: PDC6A3C0OX) DIHYDROXYALUMINUM AMINOACETATE (UNII: DO250MG0W6) EDETATE DISODIUM (UNII: 7FLD91C86K) CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) KAOLIN (UNII: 24H4NWX5CO) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) SODIUM POLYACRYLATE (8000 MW) (UNII: 285CYO341L) ALOE VERA LEAF (UNII: ZY81Z83H0X) GELATIN (UNII: 2G86QN327L) NICOTINATE ETHANOLAMINE (UNII: 71P617JXF6) TARTARIC ACID (UNII: W4888I119H) NONIVAMIDE (UNII: S846B891OR) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:81877-601-15 3 in 1 POUCH 01/23/2023 1 5 in 1 PATCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 01/23/2023 Labeler - FORREAL PHARMACEUTICALS LLC (118029197)